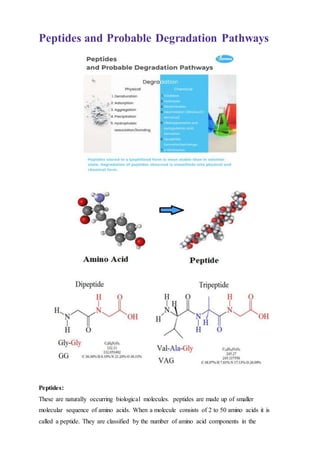
Peptides and probable degradation pathways
- 1. Peptides and Probable Degradation Pathways Peptides: These are naturally occurring biological molecules. peptides are made up of smaller molecular sequence of amino acids. When a molecule consists of 2 to 50 amino acids it is called a peptide. They are classified by the number of amino acid components in the
- 2. sequence, called amino acid residues. Tripeptides have three amino acid residues while tetrapeptides have four. A polypeptide is formed when the chain of amino acid links exceeds several dozen in length. Types of peptides depending on the number of amino acids they are characterized as: Polypeptides and Oligopeptides What are Peptides: Peptides are naturally occurring biologics around 20 naturally occurring amino acids. Peptides are safer than synthetic drugs and have a better efficacy, selectivity and specificity. Peptides can join into an enormous multiplicity of dissimilar molecules and they can have several advantages over small molecule drugs. Peptides possess bioactivities, peptide breakdowns into amino acids and manage utmost biological evolutions. Peptides are degrading into their constituent proteinogenic amino acids without generating toxic metabolites. Due to peptides impermanent nature it makes an expensive drug hence it was less accumulating in the body. Parenteral route is the most effective mode of efficient delivery of peptides. Peptides frequently act as hormones and biologic messengers carrying information through the blood from one to another tissue. Degradation Pathways: To avoid the degradation of peptides it is recommended that the peptide in lyophilized form stored below 5°C or if possible, at – 20°C or – 80 °C and solution must be refrigerated. Long term exposure to pH more than 8 and exposer to the atmospheric oxygen must be avoided. Peptides stored in a lyophilized form is most stable than in solution state. Degradation of peptides observed is classifieds into physical and chemical form is as per below: A. Physical Degradation: It involves the weak sharing of an electron pair between a hydrogen atom and another atom. It involves more dispersed variations of electromagnetic interaction between molecules or within a molecule changes in non-covalent interaction. Following are the physical degradation mechanism observed: i. Denaturation ii. Adsorption iii. Aggregation iv. Precipitation v. Hydrophobic association/bonding B. Chemical Degradation: It involves the sharing of an electron pair between two atoms are the strongest. Number of impurities were observed due to raw materials, manufacturing process, storage. Following are the chemical degradation mechanism observed:
- 3. i. Oxidation ii. Hydrolysis iii. Racemisation iv. Deamidation (Gln/Asn/C-terminus) v. Diketopiperazine and pyroglutamic acid formation vi. Disulphide formation/exchange vii. β-Elimination Peptides drugs degradation products detected and characterized by the use of systematic or complex analytical methods. Conclusion: Peptides based drug products are important remedies expected to substitute several present chemical based formulations. Peptide drugs will be produced on a large scale by biotechnology processes. Physical and chemical stability of peptide-based products is challenging. References: 1. FDA Guidance for Industry: ANDAs for certain Highly Purified Synthetic Peptide Drug Products That Refer to Listed Drugs of rDNA Origin 1. 2. ICH, Q6A, Specifications: New Chemical Drug Substances and Products, 3. ICH, Q6B, Specifications Test Procedures and Acceptance Criteria for Biotechnological/Biological Products, 4. ICH, Q5C, Stability Testing of Biotechnological/Biological Products, 5. ICH, Q1A(R2), Stability Testing of New Drug Substances and Products, 6. EMEA, Guideline on Stability Testing: Stability Testing of Existing Active Substances and Related Finished Products, 7. L. Wu , F. Chen , S. L. Lee , A. Raw and L. X. Yu , Building Parity between Brand and Generic Peptides: Regulatory and Scientific Considerations for Quality of Synthetic Peptides, Int. J. Pharm., 8. M. P. C. Ratnaparkhi and S. P. Pandya , Peptides and Proteins in Pharmaceuticals, Int. J. Curr. Pharm. Res., 9. To know more about Impurities and Pharmaceutical Drug substance read our blogs or to buy them visit Our website https://veeprho.com/
