Conformation of Alkanes and Cycloalkanes
•
0 likes•1 view
The document discusses the conformational changes that occur in molecules. It notes that: 1) Conformations involve the rotation of bonds around single bonds, with staggered and eclipsed conformations having different energies. 2) Staggered conformations are more stable than eclipsed conformations due to interactions between hydrogen and chlorine atoms. 3) Increasing the number of chlorine substituents on an alkane increases the energy difference between staggered and eclipsed conformations.
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
Recently uploaded
Recently uploaded (20)
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
Swan(sea) Song – personal research during my six years at Swansea ... and bey...

Swan(sea) Song – personal research during my six years at Swansea ... and bey...
IAC 2024 - IA Fast Track to Search Focused AI Solutions

IAC 2024 - IA Fast Track to Search Focused AI Solutions
Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...

Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...
The Evolution of Money: Digital Transformation and CBDCs in Central Banking

The Evolution of Money: Digital Transformation and CBDCs in Central Banking
Presentation on how to chat with PDF using ChatGPT code interpreter

Presentation on how to chat with PDF using ChatGPT code interpreter
The 7 Things I Know About Cyber Security After 25 Years | April 2024

The 7 Things I Know About Cyber Security After 25 Years | April 2024
WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service

WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service
Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...

Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...
Scaling API-first – The story of a global engineering organization

Scaling API-first – The story of a global engineering organization
From Event to Action: Accelerate Your Decision Making with Real-Time Automation

From Event to Action: Accelerate Your Decision Making with Real-Time Automation
Featured
Featured (20)
Product Design Trends in 2024 | Teenage Engineerings

Product Design Trends in 2024 | Teenage Engineerings
How Race, Age and Gender Shape Attitudes Towards Mental Health

How Race, Age and Gender Shape Attitudes Towards Mental Health
AI Trends in Creative Operations 2024 by Artwork Flow.pdf

AI Trends in Creative Operations 2024 by Artwork Flow.pdf
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
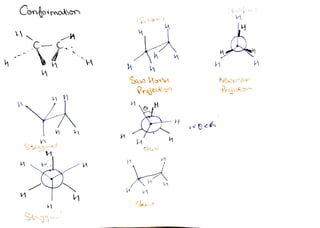
Conformation of Alkanes and Cycloalkanes
- 1. Confoimoton F i p ) Prjeie Prau - H SRL C
- 2. Eclp COn eah u t h Cnnmctian bend pou c J wil hL Monmm and o ununum stean Stga C oym pend t ut| b Dtheen ang (ang texsin Angl i two ( bonds uue ajatnt Conbon in tt pheKS i) (allust ihdhal ang *fcelipsd cmfomweien dhua ang * Stgjundcaamatn dihdal ay ail be 6 F ail sku ortomain ihdn ang l b and thn Ss TSo Stain Du t upudyien bl bcuius C-H n n Slbtensancd stein at celibsdconmotion. * valu t ch@ng nnatigm m stein ill cAcwa eusd s dncd th Vou il Ze stngans ¥ Ds Tgsana nwnathon sil Stdk ond esl incue uhn dituodnal ong incruesa) * vedu Tsnal atain ad dhdalang Ptgun sthon feltel Edbsd S R Sts 12 21 3 36
- 3. Lonfomoion of, Ppana Cl Cl2Cul CA CH M M C CW A sku staag Celtprd Elos E St St-9 300 36 Dhdea 21 G (2 In ca opana lorsiona ytcin sill be highn thun u h a Buley grtup CWz)muth Ethon eceuu CAg-M-CAL-(A2 Conpeimakan Butan c Go c cW Povniaty Anti Ecib
- 4. - J-