Effects of Heavy Metals on Spirulina
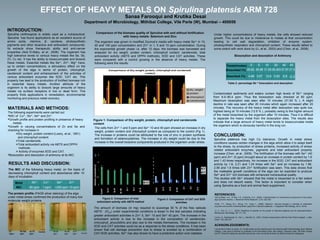
- 1. Comparioson of Dry weight, protein, chlorophyll and carotenoid content 0 2 4 6 8 10 12 14 Control Se10 Se40 Se100 Zn1 Zn5 Zn10 dry weight protein chlorophyll carotenoids Comparison of CAT and SOD activity 0 1 2 3 4 5 6 7 Control Se10 Se40 Se100 Zn1 Zn5 Zn10 CAT SOD EFFECT OF HEAVY METALS ON Spirulina platensis ARM 728 Sanaa Farooqui and Krutika Desai Department of Microbiology, Mithibai College, Vile Parle (W), Mumbai – 400056 INTRODUCTION: Spirulina (Arthrospira) is widely used as a nutraceutical. Spirulina has found applications as an excellent source of amino acids, vitamins, β- carotene, photosynthetic pigments and other bioactive and antioxidant compounds. Its extracts show therapeutic ability and anti-cancer properties (Abd El-Baky, et al., 2008). The organism has high tolerance levels to various heavy metals like Se, Sb, Zn, Cu etc. It has the ability to bioaccumulate and biosorb these metals. Essential metals like Se4+ , Zn2+ , Mg2+ have, up to certain concentrations, a stimulatory effect on the growth of the alga in terms of protein, chlorophyll, carotenoid content and enhancement of the activities of various antioxidant enzymes like SOD, CAT etc. This property has lead to the production of fortified biomass rich in essential heavy metals. Another attribute of the organism is its ability to biosorb large amounts of heavy metals via surface receptors in live or dead form. This property finds applications in remediation, environmental monitoring and precious metal recovery. MATERIALS AND METHODS: The following experiments were carried out MIC of Cu2+ , Sb3+ , Se4+ and Zn2+ . Growth profile and protein profiling in presence of heavy metals. Growth in varying concentrations of Zn and Se and checking for increase in >Dry weight, protein content (Lowry, et al., 1951) and chlorophyll content. >Total carotenoids. >Total antioxidant activity via ABTS and DPPH methods. > Activity of enzymes SOD and CAT. Biosorption and desorption of antimony at its MIC. CONCLUSION: Spirulina platensis has high Co tolerance. Growth in metal stress conditions causes certain changes in the alga which allow it to adapt itself to the stress, by production of stress proteins, increased activity of stress related antioxidant enzymes, pigments and total antioxidant property increase (Chen, et al., 2008). The fortification of the biomass with Se4+ (40 ppm) and Zn2+ (5 ppm) brought about an increase in protein content by 1.6 and 1.43 times respectively. An increase in the SOD, CAT and antioxidant activity by 1.6, 2.21 and 1.24 times with Se4+ and an increase by 1.96, 2.95 and 1.6 times with Zn2+ fortification was seen. This data shows that the malleable growth conditions of the alga can be exploited to produce Se4+ and Zn2+ rich biomass with enhanced nutraceutical quality. The studies with Sb3+ showed that the metal is biosorbed to a fair extent and does not desorb easily. This factor is important to consider when using Spirulina as a food and animal feed supplement. REFERENCES: •Abd El-Baky, H.H., El Baz, F.K., El-Baroty, G.S., (2008). Characterization of nutraceutical compounds in blue green alga Spirulina maxima. J. Medicinal Plants Research. 2(10): 292-300. •Chen, T.F., Zheng, W.J., Wong, Y.S., Yang, F., (2008). Selenium- induced changes in activities of antioxidant enzymes and content of photosynthetic pigments in Spirulina platensis. J. Integrative Plant Biology. 50(1): 40-48. •Li, Z., Guo, S., Li, L., (2003). Bioeffects of selenite on the growth of Spirulina platensis and its biotransformation. Bioresource Technology. 89: 171-176. •Lowry, O., Rosebrough, N., Farr, L., Randall, R., (1951). Protein measurement with the Folin Phenol reagent. J. Biol. Chem. 193: 265-275. ACKNOWLEDGEMENTS: I would like to thank my guide, Dr. Krutika Desai and the teaching and non-teaching staff of Microbiology Dept. Mithibai College. I also want to express my gratitude to the Pharmatech Dept. NM college, Chemistry Dept. CB Patel College and IIT, Powai for letting me use their instruments. And I would like to thank my family friends and colleagues. RESULTS AND DISCUSSION: The MIC of the following heavy metal, on the basis of decreasing chlorophyll content and absorbance after 10 days of incubation Comparison of the biomass quality of Spirulina with and without fortification with heavy metals- Selenium and Zinc The organism was grown in modified Zourruk’s media with heavy metal Se4+ in 10, 40 and 100 ppm concentration and Zn2+ in 1, 5 and 10 ppm concentration. During the exponential growth phase i.e. after 12 days, the biomass was harvested and analyzed for dry weight, protein content, chlorophyll content, carotenoids, total antioxidant activity (ABTS and DPPH methods), SOD and CAT activities. These were compared with a control growing in the absence of heavy metals. The following were the results: Figure 1: Comparison of Dry weight, protein, chlorophyll and carotenoids content The cells from Zn2+ 1 and 5 ppm and Se4+ 10 and 40 ppm showed an increase in dry weight, protein content and chlorophyll content as compared to the control (Fig 1). The increase in proteins could be attributed to the role of zinc in protein synthesis and formation of selanocysteines. The increase in dry weight could be due to the increase in the overall bioactive components produced in the organism under stress. Figure 2: Comparison of total antioxidant activity with ABTS method Figure 3: Comparison of CAT and SOD activities The amount of biomass (in mg) required to scavenge 50 % of the free radicals ABTS+ (IC50) under experimental conditions is lesser in the test samples indicating greater antioxidant activities in Zn2+ 5, Se4+ 10 and Se4+ 40 ppm. The increase in the antioxidant activity is due to the increase in the composition of carotenoids, chlorophyll, phycobillins and also due to the metals themselves. The increase in the SOD and CAT activities could be as a response to the stress conditions. It has been shown that cell damage prevention due to stress is avoided by a combination of CAT-SOD activities. Se4+ has also shown to have a protective action over catalase. Under higher concentrations of heavy metals, the cells showed reduced growth. This could be due to intolerance to metals at that concentration leading to cell wall degradation, inhibition of enzyme system, photosynthetic respiration and chlorophyll content. These results tallied to some extent with work done by (Li, et al., 2003) and (Chen, et al., 2008). Metal MIC Sb3+ Cu2+ Se4+ Zn2+ 80 ppm 1 ppm >250 ppm 10 ppm The protein profile (PAGE-silver staining) of the alga under metal stress confirmed the production of many low molecular weight proteins Cu 1ppm Se 100 ppm Zn 10 ppm Control Se 40 ppm Sb 80 ppm Zn 5 ppm Control Biosorption and desorption of Antimony Contaminated sediments and waters contain high levels of Sb3+ ranging from 8.5–90.4 ppm. Thus the biosorption was checked at 80 ppm. Maximum biosorption was seen after 10 minutes (31.93 % ). A slight decline in rate was seen after 20 minutes which again increased after 20 minutes. The recovery of the heavy metal after desorption was quite low, highest being at 10 minutes (12.6 % ), probably owing to the higher amount of the metal biosorbed by the organism after 10 minutes. Thus it is difficult to separate the heavy metal from the biosorption sites. The results also indicate that a large amount of heavy metal tends to bioaccumulate inside the organism which is obviously harmful in the long run. Time(minutes) 0 Biosorbed (%) Desorbed (%) 28.82 28.98 31.9326.40327.9727.3 10 20 40 605 4.68 3.97 12.6 0.82 8.8 Table 2: percentage Sb 3+ biosorption and desorption Comparison of the total antioxidant activity using ABTS method 0 0.5 1 1.5 2 Control Se10 Se40 Se100 Zn1 Zn5 Zn10 Inhibitionconcentration(IC50) IC50 I 12.3