Animal Cell Culture Types
- 2. Introduction • Discussed already • Applications
- 5. Types of culture media
- 6. Natural Media • Obtained from natural sources • Very useful • Lack of knowledge of the exact composition of these natural media
- 7. Artificial Media • Synthetic media There are mainly four types of artificial media: 1. Serum containing media 2. Serum-free media (defined culture media) 3. Protein free media 4. Chemically defined media
- 8. 1. SERUM CONTAINING MEDIA • Definition of Serum: Yellowish liquid, clear content left over after fibrin and cells are removed the blood is known as serum. • Fetal bovin serum(FBS) is the most commonly applied supplement in animal cell culture. • Normal growth media often contain 2-10% of serum.
- 11. 2. Serum free media • Presence of serum in the media lead to mis interpretations in immunological studies • Serum free media have been developed • These media are specifically formulated to support the culture of a single cell type and incorporated defined quantities of purified components. Adv: • Consistent and chemically defined composition • Improves reproducibility of cell culture growth and product yield • Decreases potential of contamination • Reduces difficulty and cost of product purification • May provide cost saving with a low protein serum free formulation Disadv: • Requires optimization for each hybridoma line • Applicable serum-free media have not been developed for all cell lines. • May require serum hormones and growth factors which are difficult to isolate and purity • Cell growth rate, maximum cell density and cell viability are often lower • Protease inhibitors in serum may protect cells from enzymes such as trypsin
- 13. Common Cell Culture Media Examples: Leibovitz’s L-15 medium Minimal essential medium(MEM) CMRL 1066 RPMI 1640 Dulbecco’s Modified Eagle’s Medium (DMEM) • Low glucose • High glucose Iscove’sModified Dulbecco’s Medium (IMDM) Ham’s F12 Fischers, etc.
- 14. Criteria for Selecting Media 1. Immediate survival 2. Prolonged survival 3. Indefinite growth 4. Specialized functions It’s always good to start with MEM for adherent cells and RPMI-1640 for suspension cells.
- 16. Media Supplements • Serum in Media • Basic nutrients • Growth factors and hormones • Binding proteins • Promote attachment of cells to the substrate • Protease inhibitors • Provides minerals, like Na+, K+, Zn2+, Fe2+, etc • Protects cells from mechanical damages during agitation of suspension cultures • Acts a buffer • Antibiotics
- 17. Aseptic conditions 1. Switch on the laminar flow cabinet 20 mts prior to start working 2. Swab all bottle tops & necks with 70% ethanol 3. If working on the bench use a Bunsen flame 4. Flame all bottle necks & pipette by passing very quickly through the hottest part of the flame 5. Avoiding placing caps & pipettes down on the bench; practice holding bottle tops with the little finger 6. Work either left to right or vice versa, so that all material goes to one side, once finished 7. Clean up spills immediately & always leave the work place neat & tidy 8. Never use the same media bottle for different cell lines. 9. If caps are dropped or bottles touched unconditionally touched, replace them with new ones 10. Necks of glass bottles prefer heat at least for 60 secs at a temperature of 200 C 11. Never use stock of materials during handling of cells.
- 18. Types of animal cell culture Finite cell line Continuous cell line Transformed cell line Animal cell culture PRIMARY CELL CULTURE CELL LINES ORGAN CULTURE
- 19. Types of animal cell culture 1. Primary cell culture: Primary cell culture is the disassociation of cells from a parental animal or plant tissue through enzymatic(mainly Trypsin) or mechanical measures and maintaining the growth of cells in a suitable substrate in glass or plastic containers under controlled environmental conditions. Isolated and inoculated into new culture vessel with fresh medium Suitable for vaccine production
- 20. Types of animal cell culture 2. Cell lines: • A cell line is a permanently established cell culture that will proliferate indefinitely given appropriate fresh medium and space. • Developed from a single cell and therefore consisting of cells with a uniform genetic make-up • Primary cell culture may not be viable for a long time because the cells utilizes all nutrients of the medium. • Hence, cells are diluted with fresh medium and passed into fresh culture flask for maintaining the viability of cells. • When primary cell culture is sub-cultured or transferred into fresh medium then it is called cell line. • Number of times that the culture has been sub-cultured is called passage number and the number of doublings that the cell population has undergone is called generation number. • Further divided into three groups:
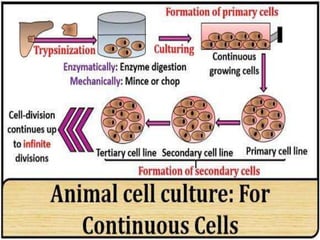
- 24. Continuous cell lines • Some cells of secondary cell culture are transformed spontaneously or chemically • Such cells lines or strain have the capacity for infinite survival (immortal) and are called continuous cell lines • Examples: Cancerous cell lines, immortal cell lines Continuous cell line may be discontinued by the effect of mutation, chemical or viruses. • The concept of change in continuous cell line is called ‘in-vitro transformation’
- 25. Characteristics of continuous cells lines • Smaller, more rounded, less adherent with a higher nucleus • Fast growth, grow in monolayer or suspension culture • Short generation time i.e. 12-24 hrs • Have aneuploidy chromosome number • Cloning efficiency is high and grow well in low concentration of serum also • Cell lines are anchorage independent (A condition in which a cell maintains its capacity to spread, divide and function despite of the absence of a stable or inert surface to anchor with)
- 27. 3. Organ culture and its culture methods The entire embryos or organs are excised from the body and culture In-vitro culture Grow organ parts Cultured organ should retain their physiological features
- 28. • Advantages: 1. Normal physiological functions are maintained 2. Cells remain fully differentiated(recognize) Disadvantages: 1. Scale-up is not recommended 2. Growth is slow 3. Fresh explantation is required for every experiment
- 29. Techniques for organ culture 1.Plasma clot or watch glass method 2.Agar gel method 3.Raft method
- 30. 1. Plasma clot or watch glass method • Developed by Dame Honor Fell and Robinson in 1963. Conditions: Petri dish should be closed with lid, sealed with parafifin wax, temperature- 37.5°C and fresh clots have been provided every 2-4 day Modified by using raft and lens paper or rayon net to place the tissue.
- 31. 2. Agar gel method • Medium is gelled with 1% agar • Gels are then kept in embtryological watch glass and explant is then transferred on surface of agar, sealed with paraffin wax • Sub-cultured into fresh agar gels every 5-7 days • Explants can be examined with stereoscopic microscope • The technique is used to study many developmental aspects of normal organs well as tumours