Exploring the Convergence of Advances in S1P Receptor Modulation With Progress in Understanding Brain Atrophy and Cognition Measures in Multiple Sclerosis
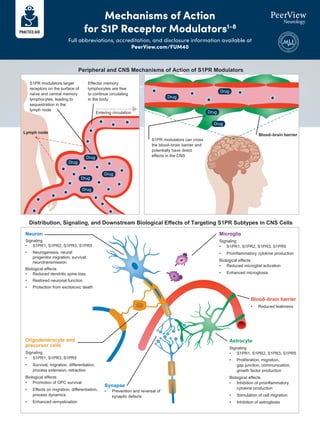
- 1. S1PR modulators target receptors on the surface of naïve and central memory lymphocytes, leading to sequestration in the lymph node Neuron Signaling • S1PR1, S1PR2, S1PR3, S1PR5 • Neurogenesis, neural progenitor migration, survival, neurotransmission Biological effects • Reduced dendritic spine loss • Restored neuronal function • Protection from excitotoxic death Peripheral and CNS Mechanisms of Action of S1PR Modulators Microglia Signaling • S1PR1, S1PR2, S1PR3, S1PR5 • Proinflammatory cytokine production Biological effects • Reduced microglial activation • Enhanced microgliosis Oligodendrocyte and precursor cells Signaling • S1PR1, S1PR3, S1PR5 • Survival, migration, differentiation, process extension, retraction Biological effects • Promotion of OPC survival • Effects on migration, differentiation, process dynamics • Enhanced remyelination Synapse • Prevention and reversal of synaptic defects Astrocyte Signaling • S1PR1, S1PR2, S1PR3, S1PR5 • Proliferation, migration, gap junction, communication, growth factor production Biological effects • Inhibition of proinflammatory cytokine production • Stimulation of cell migration • Inhibition of astrogliosis Effector memory lymphocytes are free to continue circulating in the body Entering circulation S1PR modulators can cross the blood–brain barrier and potentially have direct effects in the CNS Blood–brain barrier Lymph node Drug Drug Drug Drug Drug Drug Drug Drug Drug Blood–brain barrier • Reduced leakiness Distribution, Signaling, and Downstream Biological Effects of Targeting S1PR Subtypes in CNS Cells Mechanisms of Action for S1P Receptor Modulators1-8 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40
- 2. S1P Receptor Subtype S1P1 S1P2 S1P3 S1P4 S1P5 Localization and Function of S1PRs in the Context of MS Care CNS Neurons (function, migration), astrocytes (activation), microglia, oligodendrocytes Neurons, astrocytes, microglia, oligodendrocytes Neurons (function, migration), astrocytes (activation), microglia, oligodendrocytes Oligodendrocytes (function), astrocytes, microglia, neurons Cardiovascular Heart: HR slowing; smooth muscle: vascular tone Smooth muscle: vascular tone Heart: slowed conduction; smooth muscle: vascular tone Endothelium Permeability Permeability Permeability Immune system Lymphocytes: trafficking Lymphoid tissues, dendritic cells, mast cells NK cells (S1PR5 > S1PR1 in blood NKs) Potential adverse events Activation may contribute to HTN, pathological retinal angiogenesis (rodents), profibrotic responses (cultured fibroblasts) Activation may contribute to macular edema, decreased retinal cell death Activation may contribute to secondary heart block, profibrotic response (cultured fibroblasts) S1P receptor modulator specificity Fingolimod Siponimod Ozanimod Ponesimod Fingolimod Fingolimod Siponimod Ozanimod Fingolimod Mechanisms of Action for S1P Receptor Modulators1-8 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 1. Chun J et al. Drugs. 2021;81:207-231. 2. Chaudhry BZ et al. Neurotherapeutics. 2017;14:859-873. 3. Choi J, Chun J. Biochim Biophys Acta. 2013;1831:20-32. 4. Cohan S et al. Biomedicines. 2020;8:227. 5. Dillmann C et al. J Immunol. 2016;196:1579-1590. 6. Kulinski JM et al. Int J Mol Sci. 2018;19:1279. 7. Meissner A et al. Cardiovasc Res. 2017;113:123-133. 8. Obinata H, Hla T. Int Immunol. 2019;31:617-625.
- 3. Practical Clinical Considerations With the Use of S1P Receptor Modulators in Patients With MS1-5 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 Pharmacokinetics/Pharmacodynamics of S1P Receptor Modulators: Implications for Clinical Care Practical Aspects of Use: Monitoring and Titration Assessments • All approved S1P receptor modulators: CBC, cardiac evaluation, liver function, medications, vaccinations • Siponimod, ozanimod, ponesimod: ophthalmic assessment • Siponimod: CYP92C9 genotype Titrationc Siponimod, ozanimod, ponesimod (per prescribing information) First-Dose Monitoringc Siponimod and ponesimod (patients with certain conditionsd ), fingolimod S1P Receptor Modulators T1/2 Elimination Median Recovery Time to Normal Lymphocyte Count Fingolimod 6-9 da 1-2 months Siponimod 30 h 10 days, but up to 3-4 weeks for some patients Ozanimod 21 h 30 daysb Ponesimod 21-33 h 4 days a Increased by 50% in patients with moderate to severe heart disease. b Ninety percent recovery to baseline lymphocyte count within 3 months. c Also on reinitiation following treatment interruption. d Sinus bradycardia, Mobitz type I AV block, history of MI or HF.
- 4. Practical Clinical Considerations With the Use of S1P Receptor Modulators in Patients With MS1-5 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 1. Cohan S et al. Biomedicines. 2020;8:227. 2. Gilenya (fingolimod) Prescribing Information. https://www.novartis.us/sites/www.novartis.us/files/gilenya.pdf. 3. Mayzent (siponimod) Prescribing Information. https://www.novartis.us/sites/www.novartis.us/files/mayzent.pdf. 4. Zeposia (ozanimod) Prescribing Information. https://packageinserts.bms.com/pi/pi_zeposia.pdf. 5. Ponvory (ponesimod) Prescribing Information. https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/PONVORY-pi.pdf. Contraindications Other Than Hypersensitivity Common Adverse Reactions Transaminase increase All Headache, HTN Siponimod Headache, diarrhea, cough, influenza, sinusitis, pain (back, abdominal, extremity) Fingolimod Infection (upper respiratory, urinary tract), orthostatic hypotension, back pain, HTN Ozanimod Fingolimod, siponimod, ozanimod, ponesimod • Recent (last 6 months): MI, unstable angina, TIA, decompensated HF with hospitalization, class III/IV HF • History (fingolimod) or presence (siponimod, ozanimod) of Mobitz type 2 second- or third-degree AV block or sick sinus syndrome, unless patient has a pacemaker Fingolimod • BL QTc interval ≥500 ms • Arrhythmias needing treatment with class Ia or class III antiarrhythmic agents Ozanimod • Severe untreated sleep apnea • Concomitant MAOI use Upper respiratory tract infection, HTN Ponesimod
- 5. Imaging Outcomes of S1P Receptor Modulators in Patients With MS1-13 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 SUNBEAM RADIANCE Part B Less Brain Volume Loss With Fingolimod: Pooled Data From Phase 3 Trials Efficacy of Fingolimod, Siponimod, Ozanimod, and Ponesimod: Phase 3 Trials ns ns ns ns Cortical Gray Matter -1.77 -1.76 -1.55 -2.5 -2.0 -1.5 -1.0 -0.5 0 0.5 Baseline Month 12 Month 24 Change in Cortical Gray Matter, % b c c ns Thalamus -1.53 -1.13 -0.77 -2.5 -2.0 -1.5 -1.0 -0.5 0 0.5 Baseline a P .001. b P .05. c P .01. Month 12 Month 24 Change in Thalamus, % Fingolimod 0.5 mg Fingolimod 1.25 mg Placebo a b b ns Deep Gray Matter -2.14 -1.57 -1.83 -2.5 -2.0 -1.5 -1.0 -0.5 0 0.5 Baseline Month 12 Month 24 Change in Deep Gray Matter, % -2.5 -2.0 -1.5 -1.0 -0.5 0 0.5 Baseline Month 12 Month 24 Change in Brain Volume in the z Block, % a a a a Brain Volume -1.01 -0.69 -0.61 FREEDOMS I Fingolimod 0.5 mg/d vs placebo (24 mo) ↓ 74% (P .001) ↓ 82% (P .0001) FREEDOMS II Fingolimod 0.5 mg/d vs placebo (24 mo) ↓ 74% (P .0001) ↓ 67% (P .0001) TRANSFORMS Fingolimod 0.5 mg/d vs IFN β-1a 30 mcg (12 mo) ↓ 35% (P .001) ↓ 55% (P .001) EXPAND OPTIMUM Siponimod 2 mg/d vs placebo ↓ 81% (P .0001) ↓ 86% (P .0001) Ozanimod 0.5 mg/d vs IFN β-1a (12 mo) ↓ 25% (P = .0032) ↓ 34% (P = .0182) Ozanimod 1.0 mg/d vs IFN β-1a (12 mo) ↓ 48% (P .0001) ↓ 63% (P .0001) Ozanimod 0.5 mg/d vs IFN β-1a (24 mo) ↓ 34% (P = .0001) ↓ 47% (P = .0030) Ozanimod 1.0 mg/d vs IFN β-1a (24 mo) ↓ 42% (P .0001) ↓ 53% (P = .0006) Ponesimod 20 mg/d vs teriflunomide ↓ 56% (P .0001) ↓ 59% (P .0001) Trial NEL GEL Treatment Groups
- 6. Imaging Outcomes of S1P Receptor Modulators in Patients With MS1-13 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 Less Brain Volume Loss With Ozanimod Versus IFN β-1a: Phase 3 Clinical Trials Less Brain Volume Loss With Ponesimod Versus Teriflunomide: Phase 3 Clinical Trial Similar changes have been reported for siponimod vs placebo in SPMS (-0.71% vs -0.84% at 24 weeks; P .05) Brain Volume Changes (12 Months) -0.61 -0.49 -0.41 -1.00 -1.72 -1.12 -1.12 -0.94 -1.11 -0.50 -0.44 -1.85 -1.50 -1.40 -0.71 -0.71 -0.34 -0.16 Mean Change, % Brain Volume Changes (24 Months) -2.0 -1.5 -1.0 -0.5 0 -2.0 -1.5 -1.0 -0.5 0 Whole brain Cortical gray matter Thalamus Whole brain Cortical gray matter Thalamus Mean Change, % IFN β-1a Ozanimod 0.5 mg Ozanimod 1.0 mg IFN β-1a Ozanimod 0.5 mg Ozanimod 1.0 mg No. of patients Ponesimod 20 mg (N = 567) 403 376 Teriflunomide 14 mg (N = 566) 403 368 -0.91 -1.25 P .0001 -1.4 -1.3 -1.2 -1.1 -1.0 -0.9 -0.8 -0.7 -0.6 -0.5 -0.4 -0.3 -0.2 -0.1 0 0.1 LS Mean Change From Baseline, % (95% Cl) Ponesimod 20 mg Teriflunomide 14 mg Visit, Week
- 7. Imaging Outcomes of S1P Receptor Modulators in Patients With MS1-13 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 1. Kappos L et al. N Engl J Med. 2010;362:387-401. 2. Calabresi PA et al. Lancet Neurol. 2014;13:545-556. 3. Cohen JA et al. N Engl J Med. 2010;362:402-415. 4. Kappos L et al. Lancet. 2018;391:1263-1273. 5. Comi G et al. Lancet Neurol. 2019;18:1009-1020. 6. Cohen JA et al. Lancet Neurol. 2019;18:1021-1033. 7. Kappos L et al. JAMA Neurol. 2021;78:558-567. 8. Gaetano L et al. Neurology. 2018;0:e1-e9. 9. Kappos et al. MSVirtual 2020, 8th Joint ACTRIMS-ECTRIMS Meeting (MSVirtual 2020). Poster P0071. 10. DeLuca J et al. ECTRIMS 2018. Abstract P556. 11. DeLuca J et al. Mult Scler Rel Dis. 2021;48:102673. 12. Langdon D et al. Eur J Neurol. 2021; doi:10.1111/ENE.15081. 13. Benedict R et al. Neurology. 2021;96:e376-e386. a b Fingolimod Improved PASAT-3 Scores From Baseline Versus Placebo More Clinically Meaningful Improvement in SDMT Scores With Ozanimod Versus IFN β-1a Rate Ratios (Ozanimod vs IFN): Proportion of Patients With Clinically Meaningful Improvement in Processing Speed (Improvement in SDMT by ≥4 Points) Proportion of Patients With Clinically Meaningful Changes in SDMT Score at Months 6 and 12 (N = 1,346) More ozanimod-treated patients had improvements in SDMT score (≥4 points) at months 6 and 12 vs those receiving IFN β-1a 34.9 27 Siponimod Improved SDMT Scores in Secondary Progress Multiple Sclerosis 0 5 45 40 35 30 25 20 15 10 50 Improved (≥4 points) 40.6 41.9 No sustained change 24.6 31.1 Worsened (≥4 points) Proportion of Patients, % Siponimod (n = 1,087) Placebo (n = 540) -2.0 -1.5 1.5 1.0 0.5 0 -0.5 -1.0 2.0 Month 6 P = .0510 Month 12 Month 18 Month 24 Change From Baseline in SDMT Oral Score Siponimod Placebo IFN β-1a 30 mcg Ozanimod 0.5 mg Ozanimod 1.0 mg -1.5 12 24 36 48 60 72 84 96 108 120 -1.0 -0.5 0 0.5 1.0 1.5 2.0 2.5 3.0 Mean Change From Baseline in PASAT-3 Time, months Fingolimod 0.5 mg Placebo/fingolimod Switch from placebo to fingolimod Baseline PASAT-3 scores Fingolimod 0.5 mg = 48.3 Placebo = 47.5 a 0 1 2 Favors ozanimod Favors IFN 1.2 (1.00-1.56) Ratio (95% CI) P 1.4 (1.10-1.71) 1.2 (0.94-1.40) 1.3 (1.05-1.55) .0552 .0048 .1689 .0156 Ozanimod 0.5 mg Ozanimod 1.0 mg 24 21 23 0 70 60 50 40 30 20 10 80 Worsened (≥4 points) 53 51 48 Stable (4-point change) 22 28 30 Improved (≥4 points) 28 24 22 Worsened (≥4 points) 44 44 42 Stable (4-point change) 28 32 36 Improved (≥4 points) Patients, % Month 6 Month 12 Month 6 Month 12 a P .05. b P .01.
- 8. Screening and Monitoring for Cognitive Impairment in MS1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 • Cognitive change because of MS occurs in 34% to 65% of adults and in 33% of patients under 18 years of age • Cognitive impairment can sometimes predate the appearance of structural abnormalities on MRI and may serve as an early marker of disease activity in some individuals with MS • Information processing and memory are the most commonly affected cognitive domains in MS • Cognitive impairment predicts limitations in the workplace and in social settings independent of level of physical disability in people with MS—for example – Cognitive impairment at diagnosis predicts a change in vocational status 7 years later – People who are cognitively impaired are more likely to be unemployed and to score lower on measures of quality of life – Cognitive impairment is related to work problems such as verbal reprimands and the need for extra training in employed MS patients, representing an at-risk condition prior to job loss – Slowed information processing is associated with lower income, independent of physical dysfunction – Cognitive impairment in children and teens is associated with lack of age-appropriate cognitive development and academic difficulties, including grade retention, social challenges, depression, and behavior problems Prevalence and Impact of Cognitive Impairment in MS The National MS Society, Consortium of MS Centers, and International MS Cognition Society recommend the following cognitive screening guidelines • For adults and children (8+ years of age) with MS, when stable – At a minimum, early baseline screening with the SDMT or similarly validated test – Annual reassessment with the same instrument, or more often as needed • For adults (18+ years): more comprehensive neuropsychological assessment for anyone who tests positive on initial cognitive screening or is experiencing consequences from significant cognitive decline • For children (18 years): more comprehensive neuropsychological assessment for unexplained change in school functioning (academic or behavioral) Expert Recommendations for Cognitive Screening and Monitoring in MS
- 9. Screening and Monitoring for Cognitive Impairment in MS1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 BICAMS utilizes three tests to evaluate cognitive functioning domains most likely to be impaired in patients with MS • SDMT: information processing speed • CVLT-II: verbal memory (immediate recall trials 1-5) • BVMT-R: visual memory (immediate recall trials 1-3) BICAMS: A 15-Minute Tool to Implement in the Clinic • Health professionals should practice administering the tests • Testing should take place in a quiet room, with just the patient and assessor present • Purpose of BICAMS should be explained to the patient • BICAMS should not be used within 1 month of recovery from MS relapse or steroid therapy (or if administered to the patient within that time frame, the data should not be interpreted as indicating long-term decline), since they have a proven reversible detrimental effect on memory function • Recommended order of administration: first SDMT, second CVLT-II, third BVMT-R • In most clinical situations, yearly or biannual BICAMS evaluations will be appropriate Test Implementation • Consists of single digits paired with abstract symbols • Rows of the nine symbols are arranged pseudorandomly • The patient must say the number that corresponds with each symbol • The SDMT can be completed within 5 minutes, including instructions, practice, and testing • Reported sensitivity of 82% and specificity of 60% • Validated in several countries • High sensitivity to cognitive impairment and cognitive change in MS • Well validated against conventional brain MRI parameters (including atrophy, brain parenchymal fraction and third ventricular width, cortical lesion number, etc) • Also shown to have external clinical validity (eg, significantly linked to both current and future employment status) SDMT, Oral Form
- 10. Screening and Monitoring for Cognitive Impairment in MS1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 1. Benedict RHB et al. Lancet Neurol. 2020;19:860-871. 2. Kalb R et al. Mult Scler J. 2018;24:1665-1680. 3. Langdon DM et al. Mult Scler J. 2012;18:891-898. • Test is comprised of a 16-item word list, with four items belonging to each of four categories, arranged randomly • List is read aloud five times in the same order to the patient, at a slightly slower rate than one item per second • Patients required to recall as many items as possible, in any order, after each reading of the list • The CVLT-II trials 1-5 can be completed in 5-10 minutes, including instructions, testing, and responses • Has been validated with brain MRI total lesion area and right superior frontal atrophy, MR T1 and FLAIR lesion volume, BPF and third ventricular width, and MR diffusion measures • Full CVLT-II also has external clinical validity (eg, can differentiate employed MS patients from patients not employed because of MS) California Verbal Learning Test-II (CVLT-II) • Test requires patient to inspect a 2 × 3 stimulus array of abstract geometric figures • There are three learning trials of 10 seconds • Array is removed, and the patient is required to draw the array from memory, with the correct shapes in the correct position • Validity of BVMT-R trials 1-3 has been indicated by significant association with brain MRI total lesion area, T1 lesion and FLAIR lesion volume, BPF and third ventricular width, and right superior frontal atrophy, and correlation with atrophy of DGM nuclei, including thalamic atrophy Brief Visuospatial Memory Test Revised (BVMT-R)
- 11. Shared Treatment Decision-Making in Multiple Sclerosis1-6 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 Yes Consider different MOA and/or route of administration Change therapy? • Suboptimal response? • Intolerable adverse events? • Inadequate treatment adherence? • Disease-related factors (phenotype, activity, severity) • Patient-related factors (preference, comorbidities) • Drug-related factors (efficacy, safety, route of administration) • Relapsing or progressive • Active or not active • With or without progression or worsening Identify the multiple sclerosis phenotype • Clinical • MRI • Safety • Biomarkers Monitor No Oral Self- injection Infusion Choose therapy in collaboration with the patient
- 12. Shared Treatment Decision-Making in Multiple Sclerosis1-6 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 Please provide your answers to the following questions. 2. How do you prefer to take your medicine? 4. What are your thoughts about laboratory monitoring? 3. How much risk are you truly willing to accept? I prefer to self-inject I prefer to take a pill I prefer to have an IV infusion I have no preference of how I take my medication as long as it can be taken as infrequently as possible Risk averse: I am a safety first, benefits second type of person Moderately risk averse: I am willing to accept a little bit of risk for some extra benefit Mildly risk averse: I can accept risks for more benefit, as long as they are not serious High risk, high reward: I can accept any potential risks if they are outweighed by the potential benefits No evidence of disease activity (no attacks, no new findings on my MRI, no new disability) Preserve or improve my current level of functioning Sustain employment and normal daily activities All of the above I don’t want to be required to have any laboratory testing during treatment I am okay with laboratory testing as long I don’t have to do it every month or after I stop treatment I have no issues with the frequency or duration of laboratory testing that may be required during or after treatment 1. What are your goals for treatment?
- 13. Shared Treatment Decision-Making in Multiple Sclerosis1-6 Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUM40 1. Giovannoni G et al. Curr Opin Neurol. 2012;25(Suppl):S20-S27. 2. Rae-Grant A et al. Neurology. 2018;90:777-788. 3. Lublin FD et al. Neurology. 2014;83:278-286. 4. Gaetani L et al. J Neurol Neurosurg Psychiatry. 2019;90:870-881. 5. Hanson KA et al. Patient Prefer Adherence. 2014;8:415-422. 6. Wilson LS et al. Int J MS Care. 2015;17:74-82. The Key Components of Engaging Patients in the Decision-Making Process 2. Describe options 3. Help make decisions Determine what patient already knows List and describe options including risks and benefits Provide decision support such as decision aids, which can help patients understand their options List options again and apply teach-back method by asking patient how they would describe the options to a loved one Focus on preferences Check to see if patient is ready to make a decision or if they need more time or have additional questions Agree on treatment plan Ensure patient understands plan can be modified and decisions can be reviewed at any time Identify/summarize problem Offer choices Emphasize importance of personal preferences Discuss any uncertainties in treatment Check reaction Defer final decision until you are able to discuss options in more detail if patient asks, “What would you do?” 1. Introduce choice
