More Related Content
Similar to Poster 7-31-16
Similar to Poster 7-31-16 (20)
Poster 7-31-16
- 1. RESEARCH POSTER PRESENTATION DESIGN © 2012
www.PosterPresentations.com
Cardiovascular diseases are the leading causes of death globally. We
have previously found that mice with smooth muscle cell (SMC)-specific
deletion of the mineralocorticoid receptor (MR-KO) lack the aging-
associated rise in cardiac hypertrophy and fibrosis. We hypothesize that
SMC-MR contributes to cardiac gene expression alterations with aging
that contribute to cardiac hypertrophy and fibrosis and to the
predisposition of the heart to atrial fibrillation (AF) with age and
propose to use a mouse in which the MR is specifically deleted from
SMCs (SMC-MR knockout mouse) to explore this hypothesis. The first aim
was to characterize alterations in cardiac gene expression with aging in
mice. Left ventricle tissue was isolated from young (3 mo.), old (9 mo.),
and elderly (19 mo.) SMC-MR knockout and MR-intact littermates. RNA
was extracted from left ventricle tissue and reverse transcribed to
cDNA. Gene expression was analyzed by q/RT-PCR to determine the fold
change in pro-fibrotic, hypertrophic, and cardiac function genes with
aging. In aim two we explore the role of SMC-MR in cardiac dysfunction
and atrial fibrillation in aged mice. Cardiac ultrasound was performed
on 19 month old SMC-MR knockout and MR-intact littermates. Heart
wall thickness, systolic and diastolic dimensions, and fractional
shortening was obtained from the images to compare heart function in
the two genotypes. Using a novel electrophysiology protocol in which we
induced atrial fibrillation in aged anaesthetized mice, we compared the
ability to induce atrial fibrillation in aged SMC-MR knockout and MR-
intact littermate controls to determine if SMC-MR plays a role in aging-
associated atrial fibrillation. These experiments have aided in the
elucidation of the role of SMC-MR in cardiac gene expression with aging
and investigated the role of SMC-MR in atrial fibrillation, which may
lead to the identification of novel therapeutic targets in the treatment
of these aging-associated cardiovascular diseases.
ABSTRACT Methods & Results
SUMMARY
Acknowledgements
Special thanks to Dr. Jen DuPont, Dr. Iris Jaffe, and everyone else at the
MCRI for their help this summer with my project and presentations. I would
also like to thank the BDBS program at Tufts University Sackler School of
Biomedical Sciences for their mentoring and accommodations this summer.
Patrick J. Donegan1,2, Jennifer J. DuPont1, Amy McCurley1, Mark Aronovitz1,
Joseph McCarthy1, Robert Blanton1, Sami Noujaim1, Iris Z. Jaffe1
1Molecular Cardiology Research Institute, Tufts Medical Center, Boston, MA, USA 2Department of Biological Sciences, University of Notre Dame, Notre Dame, IN, USA
The Role of Smooth Muscle Cell Mineralocorticoid
Receptor in Cardiac Aging & Atrial Fibrillation
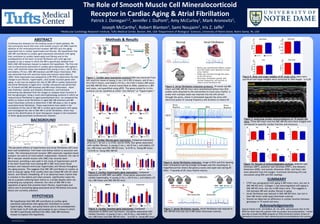
Figure 1. Cardiac gene expression protocol RNA was extracted from
left ventricle tissue of young (3 mo.) (N=2 KO/4 Intact), old (9 mo.)
(N=2 KO/2 Intact), and elderly (19 mo.) (N=2 KO/3 Intact) MR-intact
and SMC-MR-KO mice, reverse transcribed to cDNA, plated on a 384
well plate, and quantified using qPCR. The genes tested for in this
protocol can be classified as either “pro-fibrotic” or “hypertrophic”.
Figure 5. Atrial fibrillation induction protocol. 19 month old MR-
intact and SMC-MR-KO mice were anesthetized before four ECG
probes were attached to the extremities to track sinus rhythm. A
probe with multiple leads was inserted into the left atrium
through the aorta, where it stimulated (paced) the atria with
electrical pulses of varying frequency and duration to induce AF.
Figure 9. Analyzing cardiac echocardiograms of 19 month old
mice. Three MR-intact and two SMC-MR-KO mice were imaged with
ultrasound to quantify cardiac function and structure.
Figure 10. Results of echocardiography analysis. Anterior wall
thickness (AWT), posterior wall thickness (PWT), end diastolic
dimension (EDD), end systolic dimension (ESD), and heart rate
were obtained from the images. Fractional shortening (FS) was
calculated using EDD and ESD readings.
Figure 6. Atrial fibrillation induction. Image of ECG and ELG showing
heart stimulation/pacing (orange rectangle) and the resulting AF. A
magnified portion shows disrupted p waves and rapid rate during AF.
After ~7 seconds of AF, sinus rhythm returns.
BACKGROUND
HYPOTHESIS
The pervasive effects of hypertension and atrial fibrillation (AF) have
been well established, from heart and kidney failure to aneurysm and
stroke. The mineralocorticoid receptor (MR) classically regulates blood
pressure via control of renal sodium reabsorption. However, the role of
MR in vascular smooth muscle cells (SMC) has recently been
discovered, providing a new path in the study of hypertension and AF.
It has been found that mice lacking MR in SMC have lower blood
pressure and reduced ventricular hypertrophy and fibrosis throughout
the aging process. This suggests that SMC-MR contributes to cardiac as
well as vascular aging. Prior studies have also linked MR with AF, heart
failure, and fibrotic remodeling. AF is an abnormal heart rhythm that
is common in the elderly and when present, significantly raises the
risk of a patient suffering heart failure or a stroke despite often not
presenting any symptoms. However, the roles of SMC-MR in the
regulation of genes that promote heart fibrosis, hypertrophy and
failure and in promoting aging-associated atrial fibrillation and pump
failure are currently unknown.
We hypothesize that SMC-MR contributes to cardiac gene
expression alterations with aging that contribute to cardiac
hypertrophy, fibrosis, and pump failure and to the predisposition
of the heart to AF with age and propose to use a mouse in which
the MR is specifically deleted from SMCs (SMC-MR knockout
mouse) to explore this hypothesis.
Figure 2. Cardiac fibrosis gene expression. Compared expression
of A) Col 1, B) Col 3, C) CTGF, and D) TGFB, four genes associated
with cardiac fibrosis, in young (3 mo.), old (9 mo.), and elderly (19
mo.) MR-intact and SMC-MR-KO mice. *p<0.05 vs. Young MR-intact
**p<0.05 vs. Young SMC-MR-KO
Figure 3. Cardiac hypertrophy gene expression. Compared
expression of ANP, BNP, and aMHC, three genes associated with
cardiac hypertrophy, in young (3 mo.), old (9 mo.), and elderly (19
mo.) MR-intact and SMC-MR-KO mice.
Figure 4. Cardiac function gene expression. Compared
expression of SERCA and Calcineurin, two genes associated with
cardiac function, in young (3 mo.), old (9 mo.), and elderly (19
mo.) MR-intact and SMC-MR-KO mice. *p<0.05 vs. Young MR-intact
Figure 8. Body and organ weights of AF study mice. Mice were
sacrificed and organ weights were corrected to tibia length. *p<0.05
• CTGF was upregulated with aging in MR-intact mice, but not in
SMC-MR-KO mice. Collagen 3 was downregulated with aging in
SMC-MR-KO mice, but not in MR-intact mice. This suggests a
link between MR and cardiac fibrosis in aging.
• Atrial fibrillation was induced in SMC-MR-KO mice, but not in
their MR-intact (wild-type) littermate controls.
• Overall we observed no difference in cardiac function between
genotypes in 19 month old mice.
Figure 7. Atrial fibrillation results. Atrial fibrillation was induced in
SMC-MR-KO mice but not in MR-intact littermate controls.
Cardiac FibrosisBlood Pressure
%CardiacFibrosis