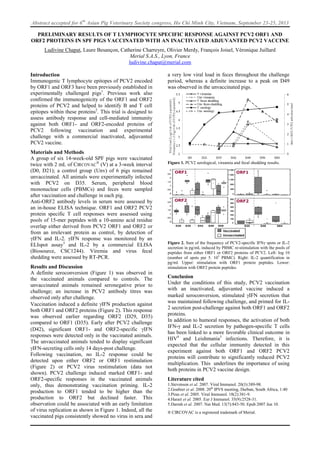
PCV2 Vaccine Cellular Immunity Against ORF1 and ORF2
- 1. Abstract accepted for 6th Asian Pig Veterinary Society congress, Ho Chi Minh City, Vietnam, September 23-25, 2013 PRELIMINARY RESULTS OF T LYMPHOCYTE SPECIFIC RESPONSE AGAINST PCV2 ORF1 AND ORF2 PROTEINS IN SPF PIGS VACCINATED WITH AN INACTIVATED ADJUVANTED PCV2 VACCINE Ludivine Chapat, Laure Besançon, Catherine Charreyre, Olivier Merdy, François Joisel, Véronique Juillard Merial S.A.S., Lyon, France ludivine.chapat@merial.com Introduction Immunogenic T lymphocyte epitopes of PCV2 encoded by ORF1 and ORF3 have been previously established in experimentally challenged pigs1. Previous work also confirmed the immunogenicity of the ORF1 and ORF2 proteins of PCV2 and helped to identify B and T cell epitopes within these proteins2. This trial is designed to assess antibody response and cell-mediated immunity against both ORF1- and ORF2-encoded proteins of PCV2 following vaccination and experimental challenge with a commercial inactivated, adjuvanted PCV2 vaccine. Materials and Methods A group of six 14-week-old SPF pigs were vaccinated twice with 2 mL of CIRCOVAC ® (V) at a 3-week interval (D0, D21); a control group (Unv) of 6 pigs remained unvaccinated. All animals were experimentally infected with PCV2 on D35. Serum, peripheral blood mononuclear cells (PBMCs) and feces were sampled after vaccination and challenge in each pig. Anti-ORF2 antibody levels in serum were assessed by an in-house ELISA technique. ORF1 and ORF2 PCV2 protein specific T cell responses were assessed using pools of 15-mer peptides with a 10-amino acid residue overlap either derived from PCV2 ORF1 and ORF2 or from an irrelevant protein as control, by detection of γIFN and IL-2. γIFN response was monitored by an ELIspot assay3 and IL-2 by a commercial ELISA (Biosource, CSC1244). Viremia and virus fecal shedding were assessed by RT-PCR. Results and Discussion A definite seroconversion (Figure 1) was observed in the vaccinated animals compared to controls. The unvaccinated animals remained seronegative prior to challenge; an increase in PCV2 antibody titres was observed only after challenge. Vaccination induced a definite IFN production against both ORF1 and ORF2 proteins (Figure 2). This response was observed earlier regarding ORF2 (D29, D35) compared to ORF1 (D35). Early after PCV2 challenge (D42), significant ORF1- and ORF2-specific IFN responses were detected only in the vaccinated animals. The unvaccinated animals tended to display significant IFN-secreting cells only 14 days-post challenge. Following vaccination, no IL-2 response could be detected upon either ORF2 or ORF1 restimulation (Figure 2) or PCV2 virus restimulation (data not shown). PCV2 challenge induced marked ORF1- and ORF2-specific responses in the vaccinated animals only, thus demonstrating vaccination priming. IL-2 production to ORF1 tended to be higher than the production to ORF2 but declined faster. This observation could be associated with an early limitation of virus replication as shown in Figure 1. Indeed, all the vaccinated pigs consistently showed no virus in sera and a very low viral load in feces throughout the challenge period, whereas a definite increase to a peak on D49 was observed in the unvaccinated pigs. Figure 1. PCV2 serological, vireamia and fecal shedding results. Figure 2. Sum of the frequency of PCV2-specific IFNγ spots or IL-2 secretion in pg/mL induced by PBMC re-stimulation with the pools of peptides from either ORF1 or ORF2 proteins of PCV2. Left: log 10 (number of spots per 5. 105 PBMC). Right: IL-2 quantification in pg/ml. Upper: stimulation with ORF1 protein peptides. Lower: stimulation with ORF2 protein peptides. Conclusion Under the conditions of this study, PCV2 vaccination with an inactivated, adjuvanted vaccine induced a marked seroconversion, stimulated IFN secretion that was maintained following challenge, and primed for IL- 2 secretion post-challenge against both ORF1 and ORF2 proteins. In addition to humoral responses, the activation of both IFN-γ and IL-2 secretion by pathogen-specific T cells has been linked to a more favorable clinical outcome in HIV4 and Leishmania5 infections. Therefore, it is expected that the cellular immunity detected in this experiment against both ORF1 and ORF2 PCV2 proteins will contribute to significantly reduced PCV2 multiplication. This underlines the importance of using both proteins in PCV2 vaccine design. Literature cited 1.Stevenson et al. 2007. Viral Immunol. 20(3):389-98. 2.Goubier et al. 2008. 20th IPVS meeting, Durban, South Africa, 1:40 3.Piras et al. 2005. Viral Immunol. 18(2):381-9. 4.Harari et al. 2005. Eur J Immunol. 35(9):2528-31. 5.Darrah et al. 2007. Nat Med. 13(7):843-50. Epub 2007 Jun 10. ® CIRCOVAC is a registered trademark of Merial.
