The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes
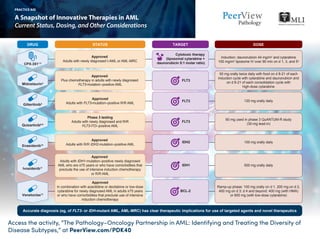
- 1. Access the activity, “The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes,” at PeerView.com/PDK40 A Snapshot of Innovative Therapies in AML Current Status, Dosing, and Other Considerations PRACTICE AID STATUS DOSEDRUG TARGET Approved Plus chemotherapy in adults with newly diagnosed FLT3-mutation–positive AML 50 mg orally twice daily with food on d 8-21 of each induction cycle with cytarabine and daunorubicin and on d 8-21 of each consolidation cycle with high-dose cytarabine Midostaurin4 FLT3 Approved Adults with R/R IDH2-mutation–positive AML 100 mg orally daily Enasidenib10 IDH2 Approved Adults with IDH1-mutation–positive newly diagnosed AML who are ≥75 years or who have comorbidities that preclude the use of intensive induction chemotherapy or R/R AML 500 mg orally daily Ivosidenib11 IDH1 Phase 3 testing Adults with newly diagnosed and R/R FLT3-ITD–positive AML 60 mg used in phase 3 QuANTUM-R study (30-mg lead-in)Quizartinib6-9 FLT3 Approved Adults with newly diagnosed t-AML or AML-MRC Induction: daunorubicin 44 mg/m2 and cytarabine 100 mg/m2 liposome IV over 90 min on d 1, 3, and 5a CPX-3511-3 Cytotoxic therapy (liposomal cytarabine + daunorubicin 5:1 molar ratio) Approved Adults with FLT3-mutation–positive R/R AML 120 mg orally daily Gilteritinib5 FLT3 Approved In combination with azacitidine or decitabine or low-dose cytarabine for newly diagnosed AML in adults ≥75 years or who have comorbidities that preclude use of intensive induction chemotherapy Ramp-up phase: 100 mg orally on d 1, 200 mg on d 2, 400 mg on d 3; d 4 and beyond: 400 mg (with HMA) or 600 mg (with low-dose cytarabine) BCL-2 Venetoclax12 Accurate diagnosis (eg, of FLT3- or IDH-mutant AML, AML-MRC) has clear therapeutic implications for use of targeted agents and novel therapeutics
- 2. a For additional induction, use d 1 and 3 for subsequent cycles, if needed; for consolidation: daunorubicin 29 mg/m2 and cytarabine 65 mg/m2 liposome IV over 90 min on d 1 and 3. AML: acute myeloid leukemia; AML-MRC: AML with myelodysplasia-related changes; BCL-2: B cell lymphoma 2; BM: bone marrow; CD: cluster of differentiation; CR: complete remission; CRi: complete remission with incomplete blood count recovery; FLT3: fms-like tyrosine kinase 3; FN: febrile neutropenia; HCT: hematopoietic cell transplantation; Hhp: hedgehog pathway; HMA: hypomethylating agent; IDH: isocitrate dehydrogenase; ITD: internal tandem duplication; mAb: monoclonal antibody; MDS: myelodysplastic syndrome; RFS: relapse-free survival; R/R: relapsed or refractory; t-AML: therapy-related acute myeloid leukemia; TLS: tumor lysis syndrome; VOD: veno-occlusive disease. 1. Lancet JE et al. 52nd Annual Meeting of the American Society of Clinical Oncology (ASCO 2016). Abstract 7000. 2. Lancet JE et al. 2017 Annual BMT Tandem Meetings (BMT Tandem 2017). Abstract 19. 3. Vyxeos (daunorubicin and cytarabine) Prescribing Information. http://pp.jazzpharma.com/pi/ vyxeos.en.USPI.pdf. 4. Rydapt (midostaurin) Prescribing Information. https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/rydapt.pdf. 5. Xospata (gilteritinib) Prescribing Information. https://astellas.us/docs/xospata.pdf. 6. https://clinicaltrials.gov/ct2/show/NCT02668653. 7. https://clinicaltrials.gov/ct2/show/NCT02039726. 8. Cortez J et al. 23rd Congress of the European Hematology Association (EHA 2018). Abstract LB2600. 9. https://pharmaphorum.com/market-access-2/fda-grants-leukaemia-drug-breakthrough-status/. 10. Idhifa (enasidenib) Prescribing Information. https://media.celgene.com/content/uploads/idhifa-pi.pdf. 11. Tibsovo (ivosidenib) Prescribing Information. https://www.tibsovo.com/pdf/prescribinginformation.pdf. 12. Venclexta (venetoclax) Prescribing Information. https://www.rxabbvie.com/pdf/venclexta.pdf. 13. Mylotarg (gemtuzumab ozogamicin) Prescribing Information. http://labeling.pfizer.com/ShowLabeling.aspx?id=9548. 14. https://www.fda.gov/drugs/fda-approves-glasdegib-aml-adults-age-75-or-older-or-who-have-comorbidities. 15. Daurismo (glasdegib) Prescribing Information. http://labeling.pfizer.com/ShowLabeling. aspx?id=11336. 16. Agura E et al. 60th American Society of Hematology Annual Meeting and Exposition (ASH 2018). Abstract 1017. 17. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-onureg-azacitidine-tablets-acute-myeloid-leukemia. Access the activity, “The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes,” at PeerView.com/PDK40 A Snapshot of Innovative Therapies in AML Current Status, Dosing, and Other Considerations PRACTICE AID STATUS DOSEDRUG TARGET Approved In combination with low-dose cytarabine for newly diagnosed AML in adults ≥75 years or who have comorbidities that preclude use of intensive induction chemotherapy 100 mg orally daily Glasdegib14,15 Hhp Approved Patients with AML who achieved first CR or CRi following intensive induction chemotherapy and are not able to complete intensive curative therapy 300 mg orally daily for 14 d of 28-d treatment cycles (QUAZAR study)Oral azacitidine17 Epigenetic modification (novel oral formulation of HMA) Accurate diagnosis (eg, of FLT3- or IDH-mutant AML, AML-MRC) has clear therapeutic implications for use of targeted agents and novel therapeutics Approved Newly diagnosed CD33+ AML in adults, R/R CD33+ AML in adults, and in pediatric patients aged ≥2 years Induction: 3 mg/m2 (up to one 4.5-mg vial) on d 1, 4, and 7 in combination with daunorubicin and cytarabine CD33Gemtuzumab ozogamicin13 Phase 3 SIERRA study Adults aged ≥55 years with active, R/R AML, adequate organ function, and related/unrelated matched donor Dosimetry directed (SIERRA study) CD45 (BC8 mAb linked to radioisotope iodine-131)Iomab-B16
- 3. Access the activity, “The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes,” at PeerView.com/PDK40 Tools for Characterizing and Diagnosing AML and Higher-Risk Subtypes PRACTICE AID AML with recurrent genetic abnormalities AML with t(8;21)(q22;q22.1);RUNX1-RUNX1T1 AML with inv(16)(p13.1q22) or t(16;16)(p13.1;q22);CBFB-MYH11 APL with PML-RARA AML with t(9;11)(p21.3;q23.3);KMT2A-MLLT3 AML with t(6;9)(p23;q34.1);DEK-NUP214 AML with inv(3)(q21.3q26.2) or t(3;3)(q21.3;q26.2); GATA2, MECOM AML (megakaryoblastic) with t(1;22)(p13.3;q13.3);RBM15-MKL1 Provisional entity: AML with BCR-ABL1 AML with mutated NPM1 AML with biallelic mutations of CEBPA Provisional entity: AML with mutated RUNX1 AML with myelodysplasia-related changes Therapy-related myeloid neoplasms AML, NOS AML with minimal differentiation AML without maturation AML with maturation Acute myelomonocytic leukemia Acute monoblastic/monocytic leukemia AML and Related Neoplasms Complex karyotype (≥3 abnormalities) Balanced abnormalities t(11;16)(q23.3;p13.3) t(3;21)(q26.2;q22.1) t(1;3)(p36.3;q21.2) t(2;11)(p21;q23.3) t(5;12)(q32;p13.2) t(5;7)(q32;q11.2) t(5;17)(q32;p13.2) t(5;10)(q32;q21.2) t(3;5)(q25.3;q35.1) Cytogenetic Abnormalities Note: these cytogenetic abnormalities are sufficient to diagnose AML-MRC when ≥20% PB or BM blasts are present and prior therapy has been excluded The WHO released an updated classification of AML in 20161 Unbalanced abnormalities –7/del(7q) del(5q)/t(5q) i(17q)/t(17p) –13/del(13q) del(11q) del(12p)/t(12p) idic(X)(q13) Pure erythroid leukemia Acute megakaryoblastic leukemia Acute basophilic leukemia Acute panmyelosis with myelofibrosis
- 4. AML: acute myeloid leukemia; AML-MRC: acute myeloid leukemia with myelodysplasia-related changes; APL: acute promyelocytic leukemia; BCR-ABL1: B-cell receptor–Abelson murine leukemia viral oncogene homolog 1; BM: bone marrow; CBFB: core-binding factor beta subunit; CD: cluster of differentiation; CEBPA: CCAAT/enhancer binding protein alpha; FLT3-ITD: FLT3-internal tandem duplication; IDH1/2: isocitrate dehydrogenase 1/2; IHC: immunohistochemistry; KMT2A-MLLT3: lysine-specific methyltransferase 2A–myeloid/lymphoid or mixed-lineage leukemia; MDS: myelodysplastic syndrome; MECOM: MDS1 and EVI1 complex locus; MPO: myeloperoxidase; MRD: minimal residual disease; MYH11: myosin heavy chain 11; NGS: next-generation sequencing; NOS: not otherwise specified; NPM1: nucleophosmin 1; PB: peripheral blood; PML-RARA: promyelocytic leukemia/retinoic acid receptor alpha; PT: prothrombin time; PTT: partial thromboplastin time; RBM15-MKL1: RNA binding motif protein 15–megakaryoblastic leukemia (translocation) 1; RUNX1: runt related transcription factor 1; sAML: secondary acute myeloid leukemia; TKD: tyrosine kinase domain; TP53: tumor protein 53. 1. Arber DA et al. Blood. 2016;127:2391. 2. NCCN Clinical Practice Guidelines in Oncology: Acute Myeloid Leukemia. Version 3.2020. https://www.nccn.org/professionals/physician_gls/pdf/aml.pdf. 3. Roloff GW, Griffiths EA. Blood Adv. 2018;2:3070-3080. Access the activity, “The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes,” at PeerView.com/PDK40 Tools for Characterizing and Diagnosing AML and Higher-Risk Subtypes PRACTICE AID Several tests should be considered to accurately diagnose AML and its subtypes2 • Including assessment for c-KIT, FLT3-ITD, FLT3-TKD, NPM1, CEBPA, IDH1/IDH2, and TP53 • Multiplex gene panels and NGS are recommended for more comprehensive assessment Although routine adoption of MRD assessment in all AML settings is not yet recommended, efforts to standardize assessment are underway. Methods include3 • Real-time PCR (dependent upon identification of a suitable abnormality) • NGS; potentially applicable to all leukemia-specific gene combinations Note: Some AML subtypes may have a distinctive diagnosis Example: AML-MRC can present with a myeloblast phenotype and key markers such as CD34 and CD117 and myeloid markers CD13, CD33, and MPO Consider ordering an MDS panel to ensure genetic abnormalities related to sAML are identified CBC, differential, platelets, chemistry profile PT and PTT BM core biopsy and aspirate analyses, including IHC and cytochemistry Cytogenetics and molecular analysis
