More Related Content
Similar to Peruzzi2015 (20)
Peruzzi2015
- 1. NATURE NANOTECHNOLOGY | ADVANCE ONLINE PUBLICATION | www.nature.com/naturenanotechnology 1
news & views
T
o survive and proliferate within
an organism, tumours must evade
immune surveillance. And they do
this by expressing ligands that interact
with receptors found on the surface of
lymphocyte T cells (a type of white blood
cell that makes up the immune system), and
activating an ‘immune checkpoint’ that stops
the immune system from removing foreign
substances such as tumour antigens1
. The
understanding of this immunomodulatory
pathway has led to the development of
strategies aimed at ‘re-educating’ the
immune system against cancerous cells.
Writing in Nature Nanotechnology,
Steven Fiering and colleagues at Dartmouth
College and Case Western University now
show a new way to induce the immune
system to clear metastatic cancers by using
cowpea mosaic virus nanoparticles2
—
self-assembling protein nanoparticles
derived from a plant virus.
Over the past few years, several strategies
have been investigated to augment the
immune system to kill tumours, but each
of these approaches has strengths and
weaknesses. Vaccination against specific
tumour antigens has been developed for
multiple cancers, including melanoma,
breast, colorectal, liver and blood3
. However,
for vaccines to work, the target antigen
within the tumour must be expressed
continuously. While this approach clears
certain populations of tumour cells, it also
induces the formation of resistant clones,
which do not express that particular
antigen and are thus responsible for tumour
relapse. Another strategy entailed the use of
oncolytic viruses (viruses that preferentially
infect and kill cancer cells) to deliver pro-
immunogenic genes to tumours. Here,
tumour destruction is achieved through
the combined action of viral lysis and the
cytotoxic effects of an activated immune
system resulting from the delivery of
the genes4
.
Another approach that has gained
momentum is the inhibition of immune
checkpoints. Immune checkpoints —
the many inhibitory pathways in the
immune system — are usually activated
when a specific receptor expressed by
T lymphocytes interacts with its ligand
expressed on cancer cells. This interaction
blocks the activation of the immune system
and develops tolerance towards the cancer
cells5
. Inhibitors that block these checkpoints
are usually antibodies raised against either
the receptor or the ligand, and they work by
physically preventing this ligand–receptor
interaction. Although immune checkpoint
antibodies have undergone extensive
validation in clinical trials and have been
approved by the US Food and Drug
Administration for several cancers, they are
not universally effective in all patients. They
generally only delay tumour progression,
and have significant toxicity6
. An alternative
strategy in this quest involves the inhibition
of tumour-associated lymphocytes (known
as regulatory T cells) that are responsible for
hampering the immune response against the
tumour7
. However, pharmacologic depletion
of these regulatory T cells (for example,
using low doses of cyclophosphamide) lacks
specificity and durability, and thus far, has
yielded poor results8
.
While nanoparticles have largely
been explored as a delivery agent
for chemotherapeutics, Fiering and
co-workers found that cowpea mosaic virus
CANCER IMMUNOTHERAPY
A vaccine from plant virus proteins
Cowpea mosaic virus nanoparticles can induce the immune system to clear metastatic cancers.
Pier Paolo Peruzzi and E. Antonio Chiocca
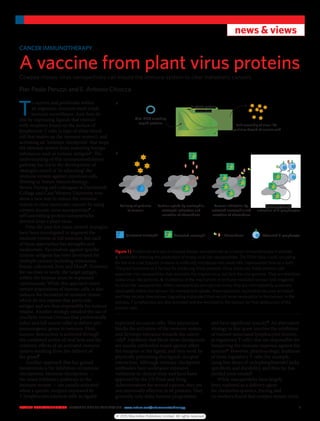
Figure 1 | Production and use of cowpea mosaic nanoparticles as a cancer immunotherapy in animals.
a, Schematic showing the production of empty viral-like nanoparticles. The DNA (blue circle) encoding
for the viral coat (capsid) proteins is artificially introduced into plant cells (represented here by a leaf).
The plant functions as a factory for producing these proteins. Once produced, these proteins self-
assemble into nanoparticles that resemble the original virus, but lack the viral genome. They are therefore
called virus-like particles. b, Illustration of the mechanism of immune-mediated tumour lysis triggered
by virus-like nanoparticles. When nanoparticles are injected in vivo, they are intercepted by quiescent
neutrophils within the tumour. On nanoparticle uptake, these quiescent neutrophils become activated
and they secrete chemokines (signalling molecules) that recruit more neutrophils to the tumour. In the
process, T lymphocytes are also activated and are recruited to the tumour for final destruction of the
tumour cells.
Viral DNA encoding
capsid proteins
In planta protein
translation
Self-assembly of virus-like
particles devoid of nucleic acid
a
Delivery of particles
to tumour
Particle uptake by neutrophils,
neutrophil activation and
secretion of chemokines
Tumour lysis and
activation of T lymphocytes
Tumour infiltration by
activated neutrophils and
secretion of chemokines
b
Quiescent neutrophil Activated neutrophil Chemokines Activated T lymphocyte
© 2015 Macmillan Publishers Limited. All rights reserved
- 2. 2 NATURE NANOTECHNOLOGY | ADVANCE ONLINE PUBLICATION | www.nature.com/naturenanotechnology
news & views
nanoparticles on their own could induce
a potent, but localized and self-contained,
activation of neutrophils (a type of immune
cell that helps fight infections) when
administered to mice either by injecting
the particles directly into the tumours or
allowing the animals to inhale them. This
activation significantly delayed the growth
of tumours and protected the animals from
tumour regrowth for a second time. These
responses were seen in a variety of tumour
models, including melanoma, and breast
and ovarian carcinoma. The results suggest
that the nanoparticles activated both innate
and adaptive immune responses against the
tumour, and a combination of proteins from
plant viruses can be possible vaccines against
tumours in animal models.
An appealing aspect of the study is
the simplicity of the structure and the
production of these nanoparticles (Fig. 1a).
The nanoparticles are 30-nm icosahedral
structures that do not contain viral DNA
and therefore would appear safe from an
infectious and genomic standpoint. They
can be produced at scale without endotoxin
contamination through molecular farming
in plants. Because of their scalability, they
can be administered in vivo via multiple
dosing regimens, in an off-the-shelf manner
that is preferable for patient treatment in an
outpatient setting. Such a dosing regimen
may be a fundamental requirement to
achieve a sustained effect over time. This
approach is also ‘antigen free’, in that it is not
based on a specific protein expressed by the
tumour. Therefore, it seems to be exempt
from the limitations that affect vaccination
strategies as discussed above.
This study does pose several questions.
Because lymphocytes are typically first
responders after a viral (or pseudo-viral)
infection, it is unclear why neutrophils
are the major players in the antitumour
response seen (Fig. 1b). Furthermore,
it is curious how such a profound and
untargeted response can be so specific to
the tumour while sparing normal tissues.
Does it suggest that the tumour maintains a
frail immunologic equilibrium with its host,
and that once the equilibrium is perturbed
(by an infection, for example), there is a
preferential immune attack that is tumour-
specific while the rest of the organism is
unaffected? If this is true, one can speculate
that possibly less immunogenic infections
would work better than highly immunogenic
challenges as they will still be able to initiate
an antitumour response, yet minimizing
the risks of immune-mediated toxicity.
Additionally, it remains an open question
whether the use of a plant virus increased
the ability to induce an antitumour response
when compared with the use of vertebrate
immune-inducing pathogens. Even after
the cowpea mosaic nanoparticle treatment,
Fiering and co-workers observed that
parental tumours eventually managed
to grow and kill a significant number of
treated mice. This suggests that in several
cases the tumours eventually managed to
escape the immune response; here, one
ponders over the mechanisms used by the
tumour to evade the activated immune
system and survive clearance. Answering
these questions will significantly advance
our understanding of the interplay between
the immune system and tumours, and
will pave the way to novel approaches in
cancer immunotherapy. ❐
Pier Paolo Peruzzi and E. Antonio Chiocca are in
the Department of Neurosurgery at the Brigham
and Women’s Hospital, 75 Francis Street, Boston,
Massachusetts 02115, USA.
e-mail: EAChiocca@partners.org
References
1. Gubin, M. M. et al. Nature 515, 577–581 (2014).
2. Lizotte, P. H. et al. Nature Nanotech.
http://dx.doi.org/10.1038/nnano.2015.292 (2015).
3. Pol, J. et al. Oncoimmunology 4, e974411 (2015).
4. Andtbacka, R. H. et al. J. Immunother. Cancer 2(Suppl. 3),
P263 (2014).
5. Pardoll, D. Nature Rev. Cancer 12, 252–264 (2012).
6. Larkin, J. et al. N. Engl. J. Med. 373, 23–34 (2015).
7. von Boehmer, H. Daniel, C. Nature Rev. Drug Discov.
12, 51–63 (2013).
8. Byrne, W. L., Mills, K. H., Lederer, J. A. O’Sullivan, G. C.
Cancer Res. 71, 6915–6920 (2011).
Published online: 21 December 2015
© 2015 Macmillan Publishers Limited. All rights reserved