Poor quality water management
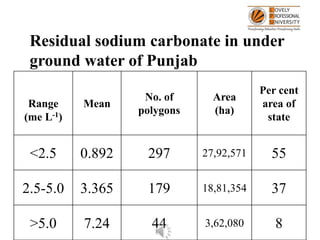
- 1. Residual sodium carbonate in under ground water of Punjab Range (me L-1) Mean No. of polygons Area (ha) Per cent area of state <2.5 0.892 297 27,92,571 55 2.5-5.0 3.365 179 18,81,354 37 >5.0 7.24 44 3,62,080 8
- 4. Simplified Quality of underground water in Punjab
- 5. Geo-statistics of generalized quality of underground water in Punjab Categories No. of polygons Area (ha) Per cent area of state Fit EC:< 2 (dS m-1) RSC: <2.5 (me L-1) 224 23,01,956 45.7 Marginal EC: 2 - 4 RSC: 2.5 - 5 245 23,19,380 46.1 Unfit EC:> 4 RSC: >5 51 4,14,664 8.2
- 6. Leaching requirement • Leaching requirement (LR) is that amount of water required to leach the salt beyond the root zone for maintaining the salt balance • Leaching requirement (LR)= EC iw • EC dw • Where: • LR=Leaching requirement • ECiw = Electrical conductivity of irrigation water • ECdw = Electrical conductivity of drainage or leaching water
- 16. Cross-Checking Chemical Analyses for Consistency and Reliability A means of locating gross errors in the chemical analyses of soils is provided by the considerable number of interrelations that exist among the values obtained for various determinations. An understanding of the principles involved in these interrelations aids in the interpretation of the analyses. ELECTRICAL CONDUCTIVITY AND TOTAL CATION and CONCENTRATION.- 1. The EC of soil solutions and saturation extracts when expressed in millimhos per centimeter at 25” C. and multiplied by 10 is approximately equal to the total soluble-cation concentration in milliequivalents per liter. 2. CATION AND ANION CONCENTRATION.-The total soluble- anion concentration or content and the total soluble cation concentration or content, expressed on an equivalent basis, are nearly equal
- 17. Cross-Checking Chemical Analyses 3. pH AND CARBONATE AND BICARBONATE CONCENTRATIONS.- If carbonate ions are present in a s&oil extract in titratable quantities, the pH reading of the extract must exceed 9. The bicarbonate concentration seldom exceeds 10 meq./l. in the absence of carbonate ions, and at pH readings of about 7 or less seldom exceeds 3 or 4 meq./l. 4.pH AND CALCIUM AND MAGNESIUM CONCENTRATIONS.- The concentration of calcium and magnesium in a saturation extract seldom exceeds 2 meq./l. at pH readings above 9. Therefore, calcium plus magnesium is low if carbonate ions are present in titratable amounts, and calcium plus magnesium is never high in the presence of a high concentration of bicarbonate ions.
- 18. Acknowledge and Thanks Author is immensely thankful to all the websites and books whom I contacted to make these slides more informative and attractive By Dr. Kamini Roy
- 19. WE SHALL OVERCOME Stay healthy By Dr. Kamini Roy