Pharmacog Module V Quality Standards 2021.pptx
•Download as PPTX, PDF•
0 likes•2 views
Pharmacognosy
Report
Share
Report
Share
Recommended
Biologicals and biosimilars a review of the science and its implications

Biologicals and biosimilars a review of the science and its implicationsNational Institute of Biologics
Recommended
Biologicals and biosimilars a review of the science and its implications

Biologicals and biosimilars a review of the science and its implicationsNational Institute of Biologics
Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...

Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...Dr K SUDHEER KUMAR KANDIBANDA
Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...

Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...Rosmirella Cano Rojas
Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...

Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...Obaid Ali / Roohi B. Obaid
More Related Content
Similar to Pharmacog Module V Quality Standards 2021.pptx
Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...

Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...Dr K SUDHEER KUMAR KANDIBANDA
Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...

Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...Rosmirella Cano Rojas
Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...

Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...Obaid Ali / Roohi B. Obaid
Similar to Pharmacog Module V Quality Standards 2021.pptx (20)
Pharmacogenomics- a step to personalized medicines

Pharmacogenomics- a step to personalized medicines
Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...

Evaluation of Drugs WHO &ICH Guidelines for The Assessment of Herbal Drugs st...
Role of Markers in Standardization of Herbal Products

Role of Markers in Standardization of Herbal Products
Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...

Biosimilaridad e intercambiabilidad: principios y evidencia: una revisión sis...
Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...

Generic Drugs; Comparative Dissolution Profile & Discriminative Method for Di...
Bioavailability and bioequivalence – problems and pitfalls

Bioavailability and bioequivalence – problems and pitfalls
More from AmosWafula3
More from AmosWafula3 (6)
Recently uploaded
Recently uploaded (20)
Hi,Fi Call Girl In Mysore Road - 7001305949 | 24x7 Service Available Near Me

Hi,Fi Call Girl In Mysore Road - 7001305949 | 24x7 Service Available Near Me
College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...

College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...
Russian Call Girls Chickpet - 7001305949 Booking and charges genuine rate for...

Russian Call Girls Chickpet - 7001305949 Booking and charges genuine rate for...
VIP Call Girls Pune Vrinda 9907093804 Short 1500 Night 6000 Best call girls S...

VIP Call Girls Pune Vrinda 9907093804 Short 1500 Night 6000 Best call girls S...
Call Girl Indore Vrinda 9907093804 Independent Escort Service Indore

Call Girl Indore Vrinda 9907093804 Independent Escort Service Indore
VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...

VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...
Call Girl Service Bidadi - For 7001305949 Cheap & Best with original Photos

Call Girl Service Bidadi - For 7001305949 Cheap & Best with original Photos
Call Girls Service Jaipur Grishma WhatsApp ❤8445551418 VIP Call Girls Jaipur

Call Girls Service Jaipur Grishma WhatsApp ❤8445551418 VIP Call Girls Jaipur
Call Girls Electronic City Just Call 7001305949 Top Class Call Girl Service A...

Call Girls Electronic City Just Call 7001305949 Top Class Call Girl Service A...
Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service

Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service
Artifacts in Nuclear Medicine with Identifying and resolving artifacts.

Artifacts in Nuclear Medicine with Identifying and resolving artifacts.
High Profile Call Girls Jaipur Vani 8445551418 Independent Escort Service Jaipur

High Profile Call Girls Jaipur Vani 8445551418 Independent Escort Service Jaipur
Call Girls Jayanagar Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Jayanagar Just Call 7001305949 Top Class Call Girl Service Available
Mumbai Call Girls Service 9910780858 Real Russian Girls Looking Models

Mumbai Call Girls Service 9910780858 Real Russian Girls Looking Models
Call Girls Thane Just Call 9910780858 Get High Class Call Girls Service

Call Girls Thane Just Call 9910780858 Get High Class Call Girls Service
Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai

Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai
Call Girl Bangalore Nandini 7001305949 Independent Escort Service Bangalore

Call Girl Bangalore Nandini 7001305949 Independent Escort Service Bangalore
Kesar Bagh Call Girl Price 9548273370 , Lucknow Call Girls Service

Kesar Bagh Call Girl Price 9548273370 , Lucknow Call Girls Service
Low Rate Call Girls Mumbai Suman 9910780858 Independent Escort Service Mumbai

Low Rate Call Girls Mumbai Suman 9910780858 Independent Escort Service Mumbai
Pharmacog Module V Quality Standards 2021.pptx
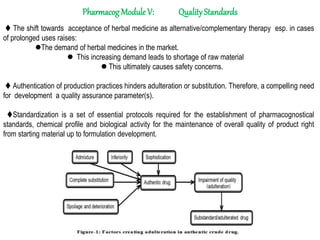
- 1. Pharmacog Module V: QualityStandards The shift towards acceptance of herbal medicine as alternative/complementary therapy esp. in cases of prolonged uses raises: The demand of herbal medicines in the market. This increasing demand leads to shortage of raw material This ultimately causes safety concerns. Authentication of production practices hinders adulteration or substitution. Therefore, a compelling need for development a quality assurance parameter(s). Standardization is a set of essential protocols required for the establishment of pharmacognostical standards, chemical profile and biological activity for the maintenance of overall quality of product right from starting material up to formulation development.
- 5. Factors affecting the quality of herbal drugs Complex nature of phytochemicals, which are responsible for holistic pharmacological response. Active principle(s) is generally unknown/uncertainty of active principle(s) Absence of a wide range of major-minor compounds Shortage of standard fingerprints of phytochemicals to record/analyse efficacy within different batches. Associated natural variability inherited with plants in wild and non-wild varieties. Variability in spectrum of pharmacological potential of species grown naturally vs. chemo varieties/cultivars. Variability in quality of the raw material, its source of origin, etc.