More Related Content
Similar to 04 ozone hole (20)
04 ozone hole
- 3. UV light and life
UV light is high energy light
When it strikes molecules it can cause
them to beak into ions or free radicals
The free radicals in turn damage large
molecules such as proteins and DNA
Damage to DNA causes mutations
It is a mutagen
Mutations can lead to cancer
© 2007 Paul Billiet ODWS
- 4. UV light and life
UV light can have beneficial effects too
It stimulates the transformation dietary
steroids into Vit D (calciferol)
UV light is used to sterilise foods and
medical equipment
Some animals can see UV light
Insects use it to guide them to nectar
sources in flowers
© 2007 Paul Billiet ODWS
- 5. UV light & DNA
UV-B has a direct effect on DNA
DNA absorbs UV light of 260nm
The action of UV forms thymine dimers
This can cause a gene mutation when the
DNA replicates
UV-B Thymine dimer
ACGCTGGCTTAGT ACGCTGGCT=TAGT
DNA
TGCGACCGAATGA TGCGACCGAATGA
© 2007 Paul Billiet ODWS
- 6. Specific damage by UV light
Sunburn
Clouding of the cornea and formation of
cataract
Skin cancer (melanoma)
Reduced rates of photosynthesis
© 2007 Paul Billiet ODWS
- 7. Ozone in the atmosphere
Ozone (O3) forms under the effect of UV
light in the stratosphere
Ozone can also form in the troposphere
Naturally it forms from volcanic activity
It is produced by motor vehicules so it is a
pollutant
Sunlight acts on NOx to produce O3
© 2007 Paul Billiet ODWS
- 8. Ozone pollution in the troposphere
Atmospheric O3 Paris 6eme arr 2006-2007
Data Airparif
160
150
140
130 Safety
120
110 limit
100
-3
O3 / µg m
90
80
70
60
50
40
30
20
10
0
02/07/2006 10/10/2006 18/01/2007 28/04/2007 06/08/2007 14/11/2007 22/02/2008
© Airparif
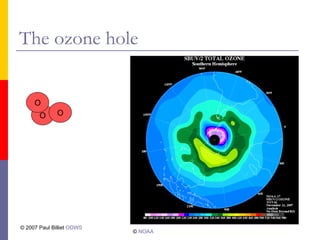
- 9. The ozone layer
Ozone is an unstable gas
It rapidly breaks down
The ozone layer is only a few cm thick
If the rate of breakdown is faster than the
rate of formation the ozone layer thins
This could develop into hole
An ozone hole was first observed over the
Antarctic in 1985
© 2007 Paul Billiet ODWS
- 10. The cause of the hole
Chlorofluorocarbons (CFCs)
The breakdown under the effect of UV
light to release chlorine radicals (Cl)
Especially under cold conditions
The chlorine radicals react with O3
converting it to O2 and more radicals ClO
© 2007 Paul Billiet ODWS
- 11. What are CFCs?
Used as propellants in aerosol spray cans
Used as refrigerants in fridges, freezers
and air conditioning units
© 2007 Paul Billiet ODWS
- 12. What can be done?
Reduce the use of CFCs
They are already banned in aerosols
(1987)
BUT they are still used as refrigerants
Recycle fridges and air conditioning plants
© 2007 Paul Billiet ODWS
- 13. The current situation
The holes developing over the pole
suggest that they may be show an
improvement
BUT CFC molecules take 30 years to rise
up to the stratosphere
The chlorine radicals last a long time
The peak ozone damage was supposed to
be in 2000
Damage could go on another 50 years
© 2007 Paul Billiet ODWS