Application of infrared spectroscopy
•
0 likes•113 views
Application of infrared spectroscopy
Report
Share
Report
Share
Download to read offline
Recommended
Infrared-Spectroscopy (IR) in Pre-Formulations.

Infrared-Spectroscopy (IR) in Pre-Formulations.Raghavendra institute of pharmaceutical education and research .
Recommended
Infrared-Spectroscopy (IR) in Pre-Formulations.

Infrared-Spectroscopy (IR) in Pre-Formulations.Raghavendra institute of pharmaceutical education and research .
Abortion pills in Doha Qatar (+966572737505 ! Get Cytotec

Abortion pills in Doha Qatar (+966572737505 ! Get CytotecAbortion pills in Riyadh +966572737505 get cytotec
More Related Content
More from Fulchand Kajale
More from Fulchand Kajale (9)
Introduction to Biopharmaceutics and Pharmacokinetics

Introduction to Biopharmaceutics and Pharmacokinetics
Recently uploaded
Abortion pills in Doha Qatar (+966572737505 ! Get Cytotec

Abortion pills in Doha Qatar (+966572737505 ! Get CytotecAbortion pills in Riyadh +966572737505 get cytotec
Call Girls In Shalimar Bagh ( Delhi) 9953330565 Escorts Service

Call Girls In Shalimar Bagh ( Delhi) 9953330565 Escorts Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Al Barsha Escorts $#$ O565212860 $#$ Escort Service In Al Barsha

Al Barsha Escorts $#$ O565212860 $#$ Escort Service In Al Barsha
Call Girls Jalahalli Just Call 👗 7737669865 👗 Top Class Call Girl Service Ban...

Call Girls Jalahalli Just Call 👗 7737669865 👗 Top Class Call Girl Service Ban...
Call Girls In Attibele ☎ 7737669865 🥵 Book Your One night Stand

Call Girls In Attibele ☎ 7737669865 🥵 Book Your One night Stand
Call Girls Indiranagar Just Call 👗 7737669865 👗 Top Class Call Girl Service B...

Call Girls Indiranagar Just Call 👗 7737669865 👗 Top Class Call Girl Service B...
VIP Model Call Girls Hinjewadi ( Pune ) Call ON 8005736733 Starting From 5K t...

VIP Model Call Girls Hinjewadi ( Pune ) Call ON 8005736733 Starting From 5K t...
➥🔝 7737669865 🔝▻ malwa Call-girls in Women Seeking Men 🔝malwa🔝 Escorts Ser...

➥🔝 7737669865 🔝▻ malwa Call-girls in Women Seeking Men 🔝malwa🔝 Escorts Ser...
Invezz.com - Grow your wealth with trading signals

Invezz.com - Grow your wealth with trading signals
Accredited-Transport-Cooperatives-Jan-2021-Web.pdf

Accredited-Transport-Cooperatives-Jan-2021-Web.pdf
Abortion pills in Doha Qatar (+966572737505 ! Get Cytotec

Abortion pills in Doha Qatar (+966572737505 ! Get Cytotec
Cheap Rate Call girls Sarita Vihar Delhi 9205541914 shot 1500 night

Cheap Rate Call girls Sarita Vihar Delhi 9205541914 shot 1500 night
➥🔝 7737669865 🔝▻ Bangalore Call-girls in Women Seeking Men 🔝Bangalore🔝 Esc...

➥🔝 7737669865 🔝▻ Bangalore Call-girls in Women Seeking Men 🔝Bangalore🔝 Esc...
Chintamani Call Girls: 🍓 7737669865 🍓 High Profile Model Escorts | Bangalore ...

Chintamani Call Girls: 🍓 7737669865 🍓 High Profile Model Escorts | Bangalore ...
Thane Call Girls 7091864438 Call Girls in Thane Escort service book now -

Thane Call Girls 7091864438 Call Girls in Thane Escort service book now -
Call Girls In Shalimar Bagh ( Delhi) 9953330565 Escorts Service

Call Girls In Shalimar Bagh ( Delhi) 9953330565 Escorts Service
Call Girls Bommasandra Just Call 👗 7737669865 👗 Top Class Call Girl Service B...

Call Girls Bommasandra Just Call 👗 7737669865 👗 Top Class Call Girl Service B...
Call Girls Begur Just Call 👗 7737669865 👗 Top Class Call Girl Service Bangalore

Call Girls Begur Just Call 👗 7737669865 👗 Top Class Call Girl Service Bangalore
Vip Mumbai Call Girls Thane West Call On 9920725232 With Body to body massage...

Vip Mumbai Call Girls Thane West Call On 9920725232 With Body to body massage...
Application of infrared spectroscopy
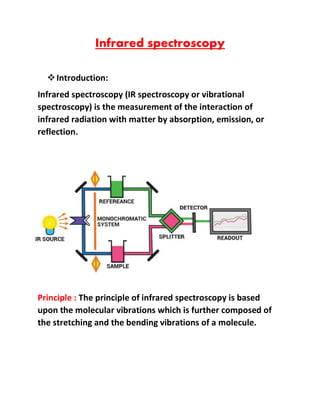
- 1. Infrared spectroscopy ❖Introduction: Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. Principle : The principle of infrared spectroscopy is based upon the molecular vibrations which is further composed of the stretching and the bending vibrations of a molecule.
- 2. ❖APPLICATIONS OF IR-SPECTROSCOPY IN THE ANALYSIS OF PHARMACEUTICAL DOSAGE FORMS ➢ Determination of Aspirin, Phenacetin and Caffeine in Tablets Carbonyl bands (c=o) at 1764, 1511 and 1665 cm–1 for aspirin, phenacetin and caffeine
- 3. ➢ Determination of Codeine Phosphate in Tablets Determined by measuring its absorption at 942 cm–1. ➢ Determination of Meprobamate in Tablets Band at 1582 cm–1 ( carbonyl) ❖APPLICATIONS OF IR-SPECTROSCOPY IN ANALYTICAL CHEMISTRY ➢ DETERMINATION OF CIS-TRANS ISOMER RATIO IN CLOMIPHENE CITRATE ➢ TO DISTINGUISH AND CHARACTERIZE THE PRI-, SEC- AND TERT-AMINE SALTS FROM ONE ANOTHER Amphetamine Sulphate: (primary amine salt ) Ephedrine Sulphate: (secondary amine salt) and Quinine Hydrochloride: tertiary amine salt. ➢ IR-SPECTROSCOPY IN THE STUDY OF COMPLEX FORMATIONS e.g Ninhydrin complex formation studied by IR spectroscopy
- 4. ➢ IR-SPECTROSCOPY IN QUANTITATIVE REACTION SEQUENCE STUDY Reduction of —NO2 group to —NH2 ; reduction of (c=o ) Carbonyl group to —CH (OH) ; oxidation of methyl-group to —COOH ➢ IR-SPECTROSCOPY IN THE IDENTIFICATION OF FUNCTIONAL GROUPS Multifunctional compound invariably exhibit separate absorption peaks due to presence of individual group e.g. Amide ➢ IR-SPECTROSCOPY : IDENTIFICATION BY FINGERPRINTING The ‘fingerprint region’ lies between 1300-400 cm–1