Clinical Check in process
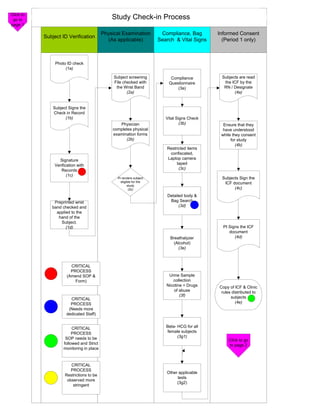
- 1. Click to go to Study Check-in Process page 1 Physical Examination Compliance, Bag Informed Consent Subject ID Verification (As applicable) Search & Vital Signs (Period 1 only) Photo ID check (1a) Subject screening Compliance Subjects are read File checked with Questionnaire the ICF by the the Wrist Band (3a) RN / Designate (2a) (4a) Subject Signs the Check in Record (1b) Vital Signs Check Physician (3b) Ensure that they completes physical have understood examination forms while they consent (2b) for study (4b) Restricted items confiscated, Signature Laptop camera Verification with taped Records (3c) (1c) PI renders subject Subjects Sign the eligible for the ICF document study (2c) (4c) Detailed body & Preprinted wrist Bag Search band checked and (3d) applied to the hand of the Subject. (1d) PI Signs the ICF document Breathalyzer (4d) (Alcohol) (3e) CRITICAL PROCESS (Amend SOP & Urine Sample Form) collection Nicotine + Drugs Copy of ICF & Clinic of abuse rules distributed to (3f) subjects CRITICAL PROCESS (4e) (Needs more dedicated Staff) CRITICAL Beta- HCG for all PROCESS female subjects SOP needs to be (3g1) Click to go followed and Strict to page 2 monitoring in place CRITICAL PROCESS Other applicable Restrictions to be tests observed more (3g2) stringent
- 2. Post Check-in Clinic Process Overnight Pre-dose Pre-dosing day Dosing day activities Post-dosing activities Check - Out activities activities * * * * * Check-in Pre-dose Scheduled Drug Meal Vitals Check Blood Draws Administration HSM, Vitals, (5a) & HSM as per (7a) ECG, (6a) protocol (8a) Biochemistry, Hematology, Sample Urine Processing (9a) Scheduled (8a1) Hands Mouth Vital checks, Restrictions Check Sleep ECG, Start (Not for (5b) Glucose & (6b) Injectibles) HSM. Blood Draw & (7b) (8b) catheter removal (9b) Subjects Suitablity for Meals as per Fluid Preparation follow posture Dosing protocol (5c) restrictions (6c) (8c) Return (7c) Confiscated Items to subjects (9c) Pre-dose Beta-HCG blood result report collection and End of Restrictions collection catheter (7d) Wallet card, (5d) insertion Snack & (6d) Compensation Results out of Range PI should (9d) be consulted High Fat Breakfast before exit. (if applicable) (6e) Subject Checks Out Release of SB’s (9e) after PI assessment (If Applicable) Subjects might return for blood draws as per study requirement *Continuous AE Monitoring & Record Click to go to Page 3
- 3. Click to go to page 2 Additional Process for Return Blood Draws Return Blood Draws Subjects attend the Clinic for Return Blood Draw (10a) Return Blood Staff verifies Draw Card ID and Signs given to the subject in subject (10b) (10b1) Signatures verified with records (10c) Return Blood Draw Compliance (10d) Staff Checks the ID and the Return Blood Draw card (10e) Sample Processing (10f1) Protocol required activities e.g. Vitals and Blood Draw (10f) Subject Compensation (If Required) (10g) Subject Leaves the Clinic with further instructions (10h) Click to go to page 1