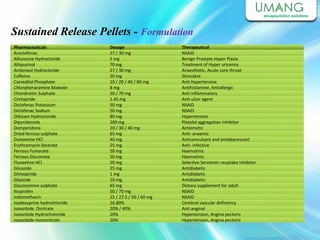
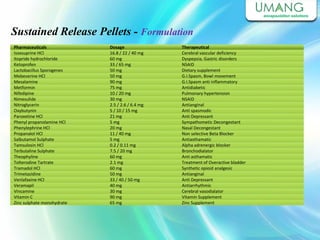
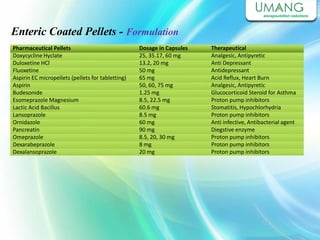
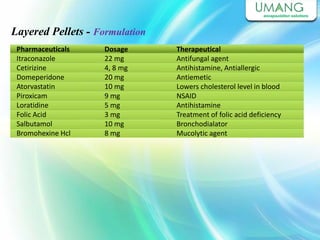
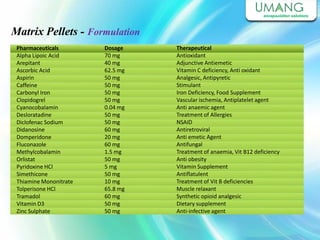
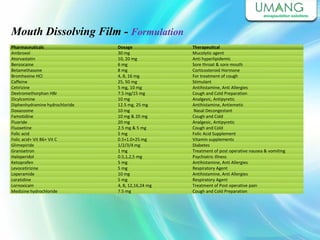
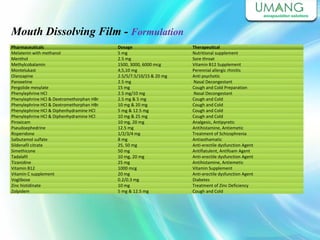
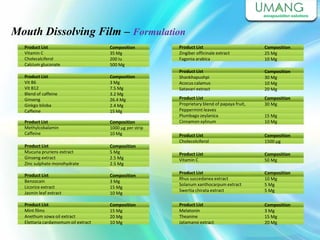
Umang Pharmatech Pvt. Ltd. is a pharmaceutical company established in 1982 that manufactures a wide range of pharmaceutical equipment and products, including various types of pellets, films, and other dosage forms. The company aims to provide high quality products that meet international standards and satisfy customer needs. It has a dedicated research and development department and experienced employees to ensure only the best products are delivered to customers on time.