This document provides an overview of quantum mechanics topics including:
1) The Schrödinger wave equation and its time-dependent and time-independent forms.
2) Expectation values and how they are used to calculate probabilities, momentum, position, and energy.
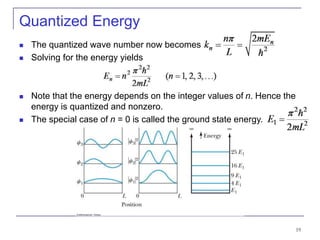
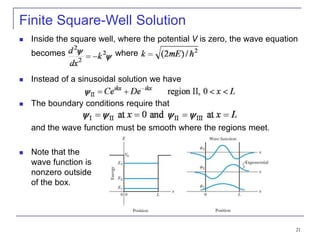
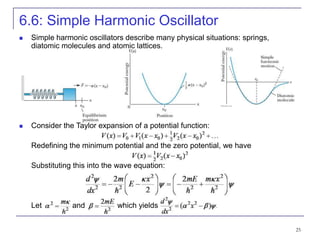
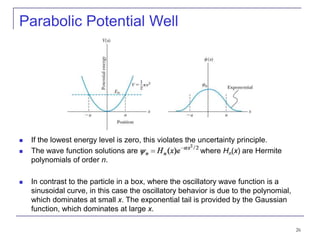
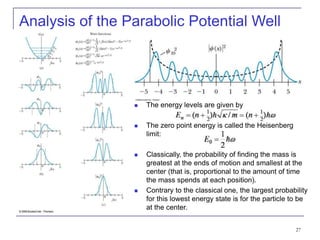
3) Specific quantum systems like infinite and finite square wells and simple harmonic oscillators. It also discusses quantization, degeneracy, and other concepts.
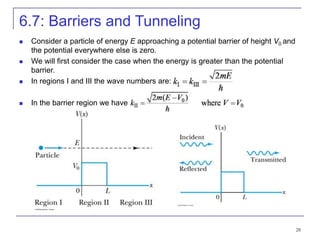
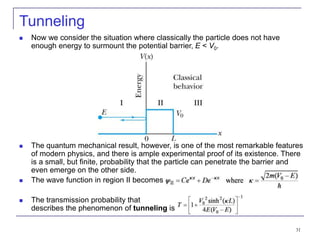
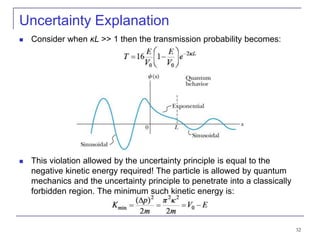
4) Barrier penetration and tunneling, where particles can pass through barriers that would be forbidden classically.
The document covers many fundamental aspects of quantum mechanics through examining various quantum systems and potentials.