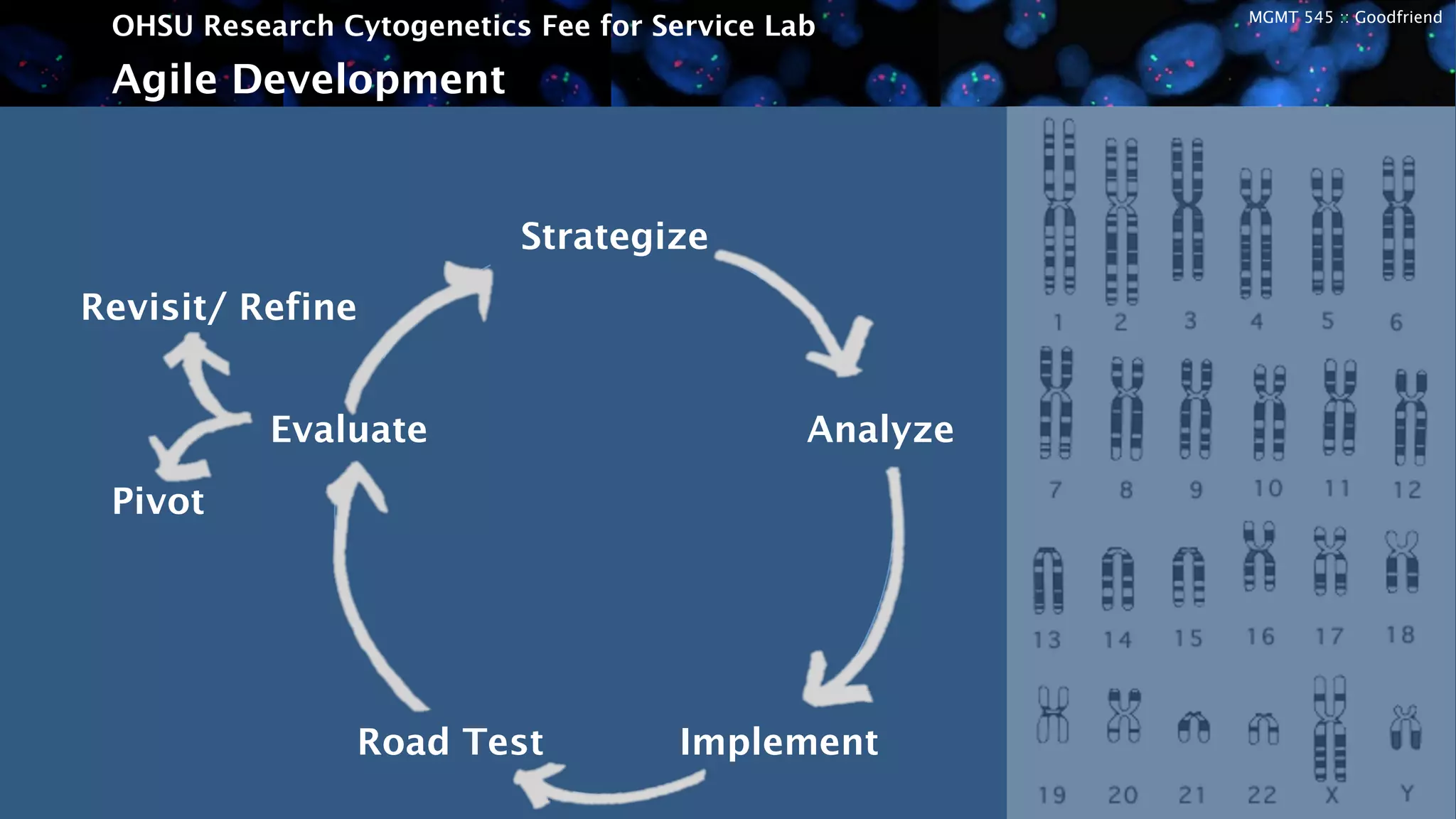
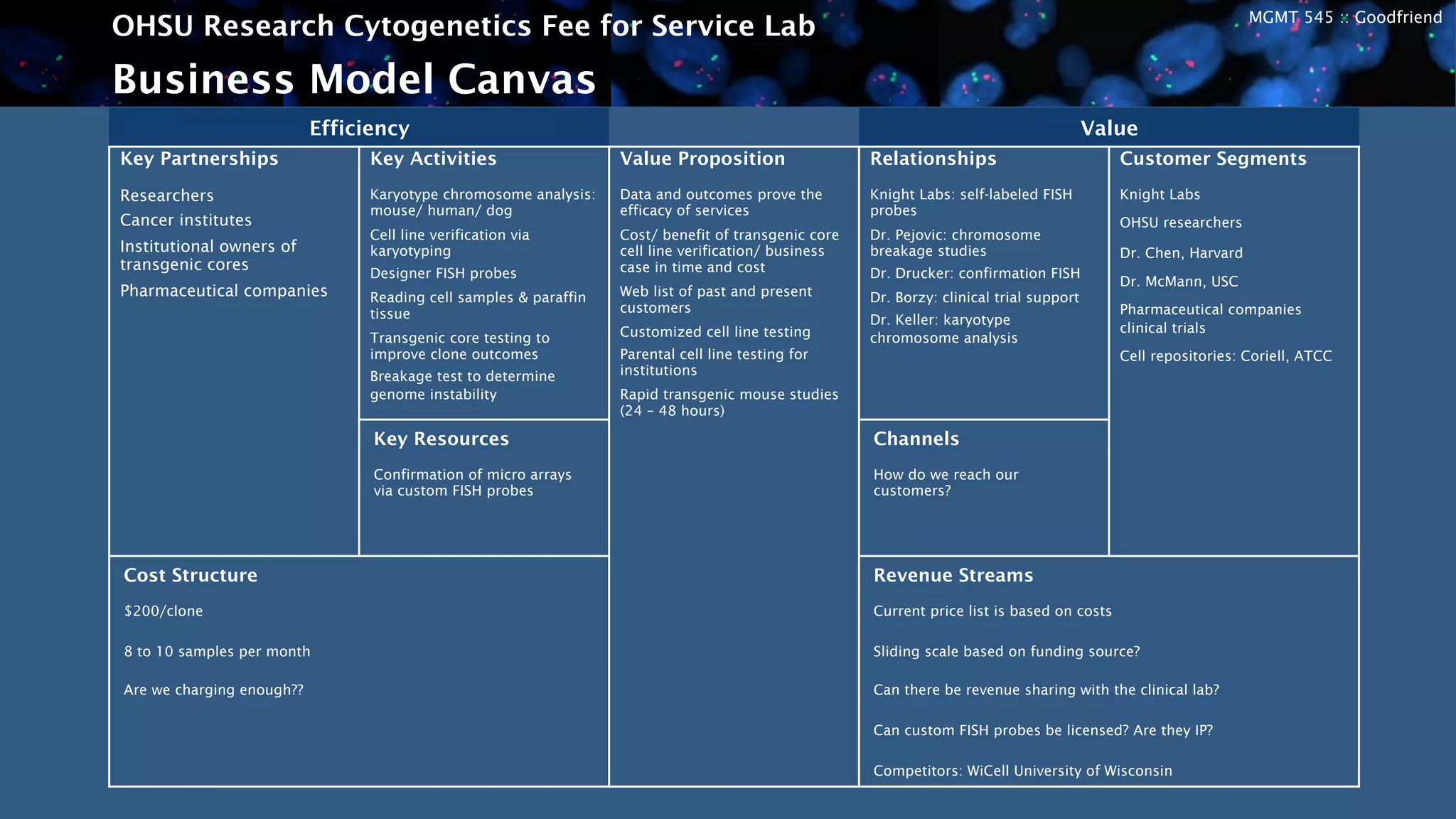
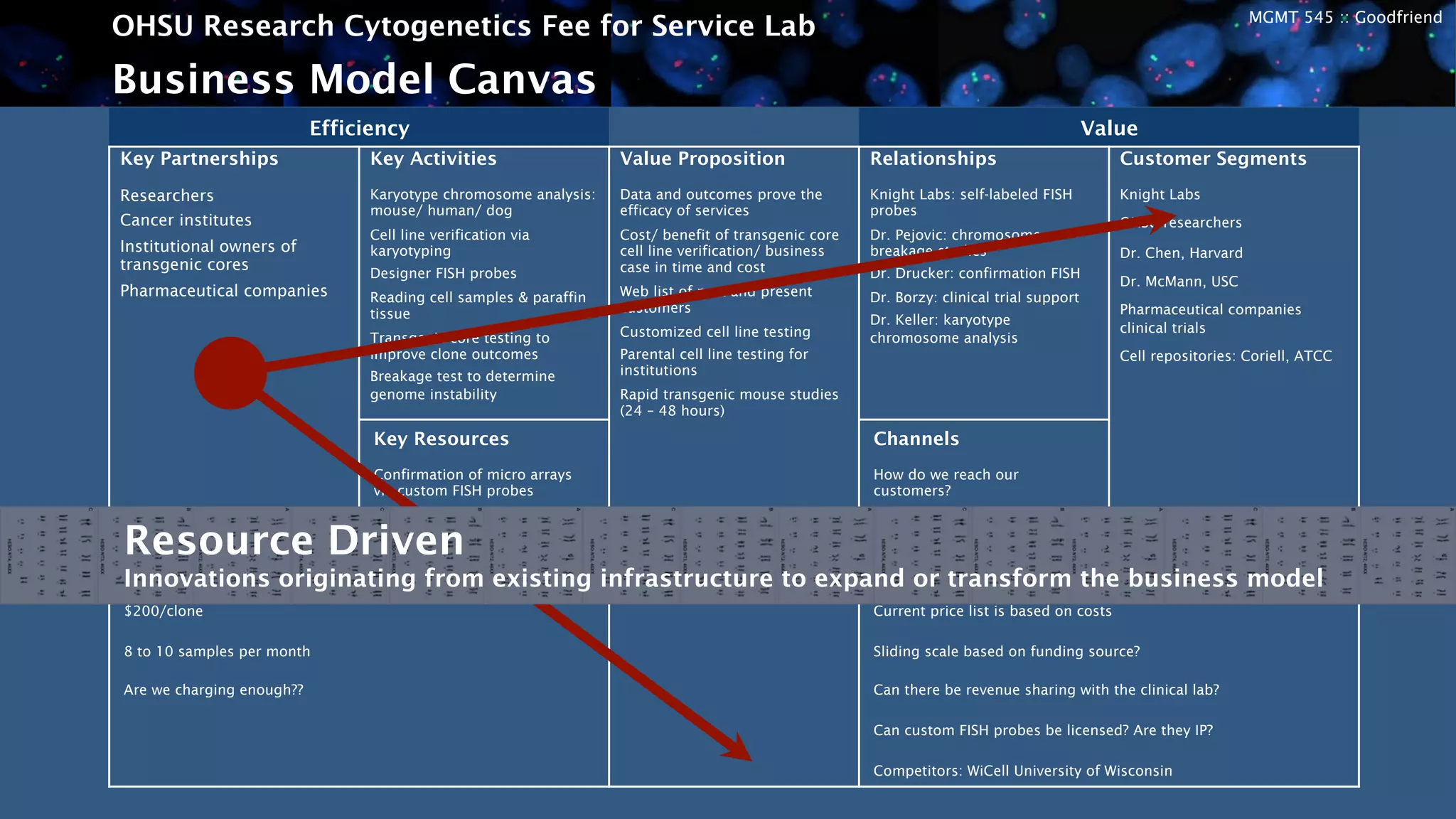
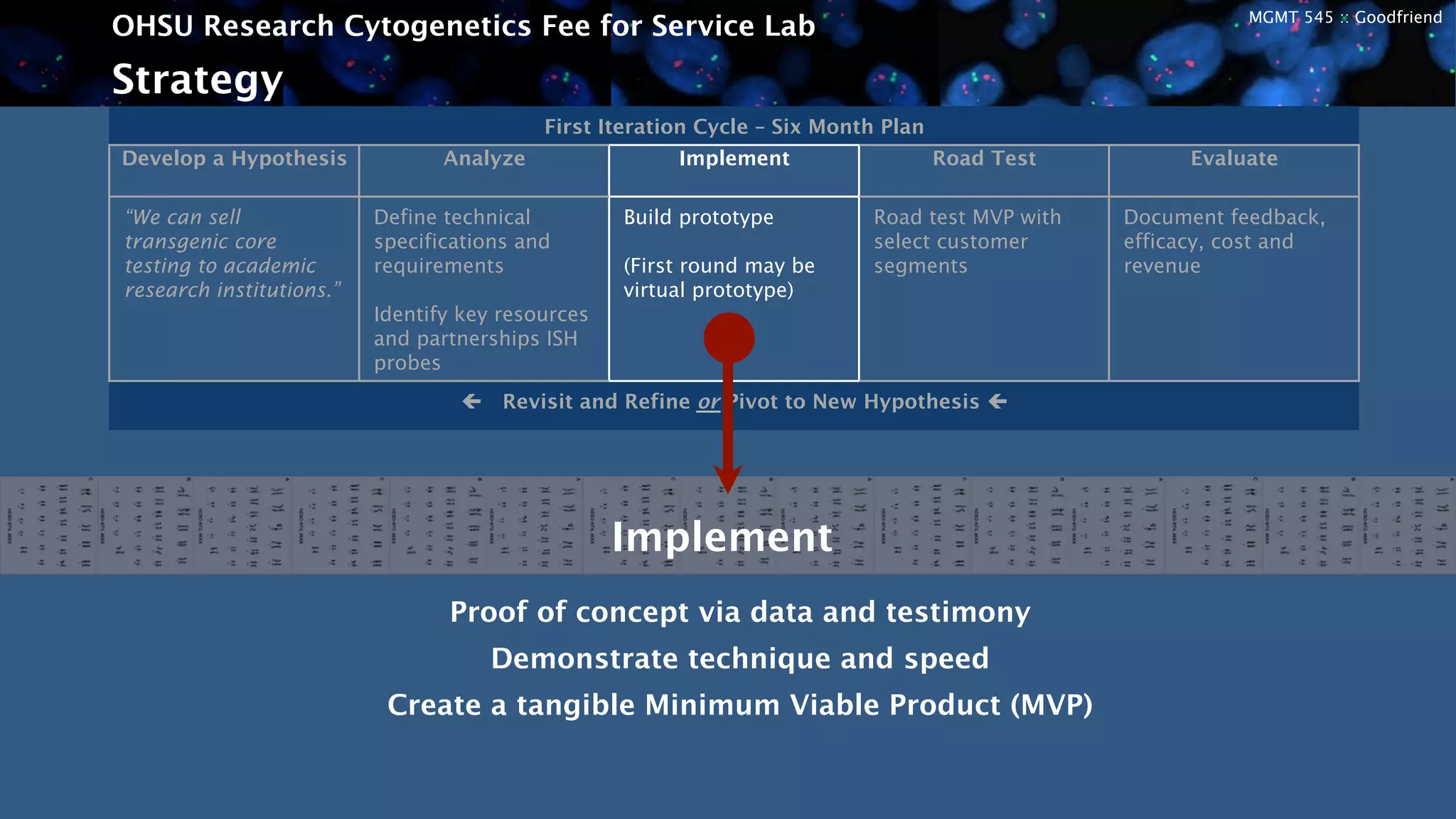
The OHSU Research Cytogenetics Fee for Service Lab is exploring opportunities to expand its business model through new services. It currently offers a range of cytogenetics services on a fee-for-service basis but sees potential to better meet unfulfilled needs. The presentation recommends taking an agile approach to iteratively develop and test new business hypotheses over six-month cycles, starting with transgenic core testing. This would involve defining specifications, building prototypes, road testing with customers, and evaluating feedback to refine or pivot the concept. The goal is to validate market interest and map a path to a proven business model that leverages existing resources to generate additional revenue streams.