Photosurveillance of Non-FDA Approved Activity: Diabetes and Instagram
•Download as PPTX, PDF•
0 likes•156 views
Are people with diabetes wearing their continuous glucose monitor as FDA approved? And should they be? An examination of #dexcom on Instagram.
Report
Share
Report
Share
Recommended
Making an Impact in Bladder Cancer Care: Integrating the Latest Evidence and ...

Making an Impact in Bladder Cancer Care: Integrating the Latest Evidence and ...PVI, PeerView Institute for Medical Education
Recommended
Making an Impact in Bladder Cancer Care: Integrating the Latest Evidence and ...

Making an Impact in Bladder Cancer Care: Integrating the Latest Evidence and ...PVI, PeerView Institute for Medical Education
Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...PVI, PeerView Institute for Medical Education
More Related Content
Similar to Photosurveillance of Non-FDA Approved Activity: Diabetes and Instagram
Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...PVI, PeerView Institute for Medical Education
Similar to Photosurveillance of Non-FDA Approved Activity: Diabetes and Instagram (20)
Основы ведения АРТ у многократно леченных пациентов 2022 / Foundations of ART...

Основы ведения АРТ у многократно леченных пациентов 2022 / Foundations of ART...
The Trend in Body Mass Index and Clinical Stage Response in HIV Patient after...

The Trend in Body Mass Index and Clinical Stage Response in HIV Patient after...
Effect of rosuvastatin on rheumatoid arthritis clinical disease activity inde...

Effect of rosuvastatin on rheumatoid arthritis clinical disease activity inde...
Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...
Correlation of Serum Creatinine Based Calculation of Glomerular Filtration Ra...

Correlation of Serum Creatinine Based Calculation of Glomerular Filtration Ra...
Случаи и разногласия по ВИЧ в 2019 году: европейские перспективы / Cases and...

Случаи и разногласия по ВИЧ в 2019 году: европейские перспективы / Cases and...
Key Slides on Individualizing ART Management Based on Treatment Safety and To...

Key Slides on Individualizing ART Management Based on Treatment Safety and To...
Managing cirrhotic HCV patients. Whom to treat, how to treat.2014

Managing cirrhotic HCV patients. Whom to treat, how to treat.2014
Managing cirrhotic HCV patients. Whom to treat, how to treat.2014

Managing cirrhotic HCV patients. Whom to treat, how to treat.2014
The Effect of Vancomycin Doses Greater Than 2 Grams on Serum

The Effect of Vancomycin Doses Greater Than 2 Grams on Serum
Review of Diagnostic Procedures and Progress in the Management of Acute Chole...

Review of Diagnostic Procedures and Progress in the Management of Acute Chole...
Review of Diagnostic Procedures and Progress in the Management of Acute Chole...

Review of Diagnostic Procedures and Progress in the Management of Acute Chole...
HTAi 2015 - Cost-Effectiveness Analysis of Drugs Used in ObesityTreatment in ...

HTAi 2015 - Cost-Effectiveness Analysis of Drugs Used in ObesityTreatment in ...
Recently uploaded
Recently uploaded (20)
❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...

❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...
science quiz bee questions.doc FOR ELEMENTARY SCIENCE

science quiz bee questions.doc FOR ELEMENTARY SCIENCE
❤️Chandigarh Escort Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ C...

❤️Chandigarh Escort Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ C...
Call Girls Service Amritsar Just Call 9352988975 Top Class Call Girl Service ...

Call Girls Service Amritsar Just Call 9352988975 Top Class Call Girl Service ...
Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*

Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*
💸Cash Payment No Advance Call Girls Pune 🧿 9332606886 🧿 High Class Call Girl ...

💸Cash Payment No Advance Call Girls Pune 🧿 9332606886 🧿 High Class Call Girl ...
💚 Low Rate Call Girls In Chandigarh 💯Lucky 📲🔝8868886958🔝Call Girl In Chandig...

💚 Low Rate Call Girls In Chandigarh 💯Lucky 📲🔝8868886958🔝Call Girl In Chandig...
Low Rate Call Girls Pune {9xx000xx09} ❤️VVIP NISHA Call Girls in Pune Maharas...

Low Rate Call Girls Pune {9xx000xx09} ❤️VVIP NISHA Call Girls in Pune Maharas...
Low Rate Call Girls Jaipur {9521753030} ❤️VVIP NISHA CCall Girls in Jaipur Es...

Low Rate Call Girls Jaipur {9521753030} ❤️VVIP NISHA CCall Girls in Jaipur Es...
💸Cash Payment No Advance Call Girls Kanpur 🧿 9332606886 🧿 High Class Call Gir...

💸Cash Payment No Advance Call Girls Kanpur 🧿 9332606886 🧿 High Class Call Gir...
Gorgeous Call Girls In Pune {9xx000xx09} ❤️VVIP ANKITA Call Girl in Pune Maha...

Gorgeous Call Girls In Pune {9xx000xx09} ❤️VVIP ANKITA Call Girl in Pune Maha...
mental health , characteristic of mentally healthy person .pptx

mental health , characteristic of mentally healthy person .pptx
💞 Safe And Secure Call Girls chhindwara 🧿 9332606886 🧿 High Class Call Girl S...

💞 Safe And Secure Call Girls chhindwara 🧿 9332606886 🧿 High Class Call Girl S...
Delhi Call Girl Service 📞8650700400📞Just Call Divya📲 Call Girl In Delhi No💰Ad...

Delhi Call Girl Service 📞8650700400📞Just Call Divya📲 Call Girl In Delhi No💰Ad...
❤️Amritsar Escort Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amrit...

❤️Amritsar Escort Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amrit...
❤️Call Girl In Chandigarh☎️9814379184☎️ Call Girl service in Chandigarh☎️ Cha...

❤️Call Girl In Chandigarh☎️9814379184☎️ Call Girl service in Chandigarh☎️ Cha...
Call Girls In Indore 📞9235973566📞Just Call Inaaya📲 Call Girls Service In Indo...

Call Girls In Indore 📞9235973566📞Just Call Inaaya📲 Call Girls Service In Indo...
💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...

💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...
Photosurveillance of Non-FDA Approved Activity: Diabetes and Instagram
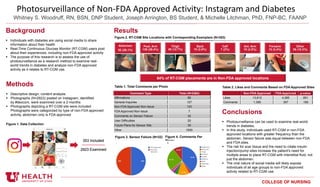
- 1. COLLEGE OF NURSING Photosurveillance of Non-FDA Approved Activity: Instagram and Diabetes Whitney S. Woodruff, RN, BSN, DNP Student, Joseph Arrington, BS Student, & Michelle Litchman, PhD, FNP-BC, FAANP Background Individuals with diabetes are using social media to share information about their health Real-Time Continuous Glucose Monitor (RT-CGM) users post about their experiences, including non-FDA approved activity The purpose of this research is to assess the use of photosurveillance as a research method to examine real- world trends in diabetes and analyze non-FDA approved activity as it relates to RT-CGM use. Methods Descriptive design; content analysis Photographs (N=2923) posted on Instagram, identified by #dexcom, were examined over a 2-months Photographs depicting a RT-CGM site were included Photographs were categorized by type of non-FDA approved activity, abdomen only is FDA approved N=147 Included Abdomen 92 (26.1%) Post. Arm 139 (39.4%) Thigh 45 (12.7%) Back 10 (2.8%) Calf 7 (2%) Ant. Arm 10 (2.8%) Forearm 12 (3.4%) Other 36 (10.2%) Results Conclusions Photosurveillance can be used to examine real-world trends in diabetes. In this study, individuals used RT-CGM in non-FDA approved locations with greater frequency than the abdomen. Sensor failure was equal between non-FDA and FDA sites. The risk for scar tissue and the need to rotate insulin injection/pump sites increase the patient's need for multiple areas to place RT-CGM with interstitial fluid, not just the abdomen. The viral nature of social media will likely expose individuals of all age groups to non-FDA approved activity related to RT-CGM use. Non-FDA Approved FDA Approved p-value Likes 13,612 4,365 .851 Comments 1,395 357 .159 Figure 2. RT-CGM Site Locations with Corresponding Exemplars (N=353) 64% of RT-CGM placements are in Non-FDA approved locations Table 2. Likes and Comments Based on FDA Approved Sites Figure 3. Sensor Failure (N=32) Comment Type Total (N=2364) Affirmations 85 General Inquiries 127 Non-FDA Approved Non-Issue 133 FDA Approved Non-Issue 7 Comments on Sensor Failure 32 User Difficulties 20 Future Plans for Sensor Site 30 Other 1930 Figure 4. Comments Per Photo Table 1. Total Comments per Photo 353 Included 2923 Examined Figure 1. Data Collection
