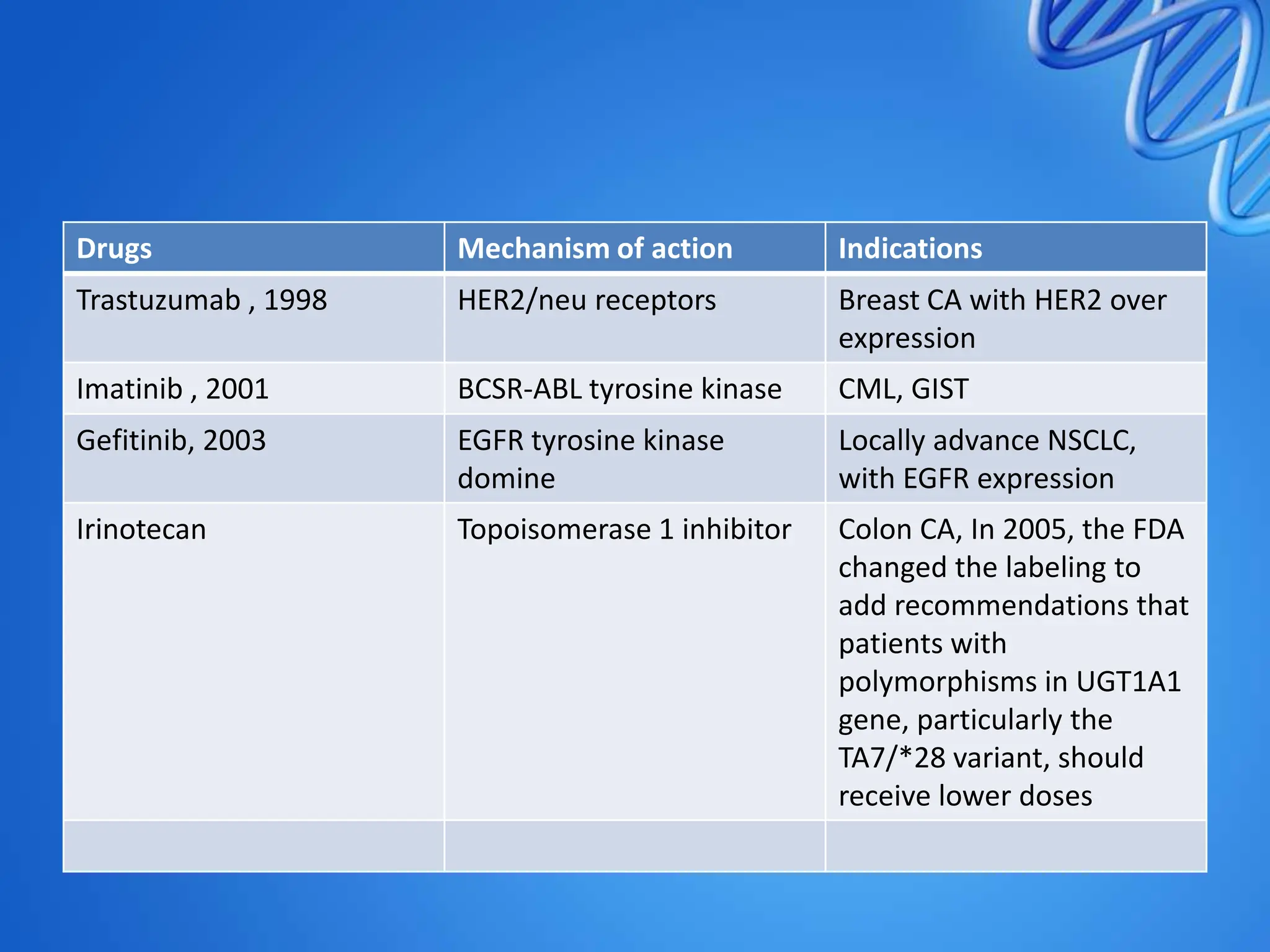
Pharmacogenomics deals with how genetic variations influence individual responses to drugs in terms of efficacy and toxicity. Key points include:
- Genetic polymorphisms, like SNPs, in drug targets, metabolizing enzymes, and transporters can impact pharmacokinetics and pharmacodynamics.
- Important drug metabolizing enzymes with genetic variants include CYP2D6, CYP2C19, CYP2C9, and TPMT. Variants in these genes can cause a patient to be a poor or ultrarapid metabolizer.
- Genetic testing can help determine the best drug and dose for a patient based on their genotype, avoiding toxic or ineffective treatment. However, widespread clinical application faces barriers like