Perfecting Precision in Modern NSCLC Practice: Are You Optimally Integrating Biomarker-Driven Therapy?
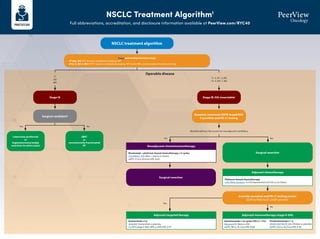
- 1. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/RYC40 NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Stage IA Surgical candidate? Lobectomy (preferred) or Segmentectomy/wedge resection (in select cases) SBRT or conventionally fractionated RT Surgical resection Consider mutation and PD-L1 testing results EGFR ex19del/ex21 L858R present? Surgical resection T1 N0 M0 Operable disease Yes Yes Yes No No No Multidisciplinary discussion for neoadjuvant candidacy Stage IB-IIIA (resectable) Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing T1–2, N1–2, M0 T3–4, N0–1, M0 Neoadjuvant chemoimmunotherapy Nivolumab + platinum-based chemotherapy x 3 cycles CheckMate -816: Nivo + chemo vs chemo mEFS: 31.6 vs 20.8 mo (HR, 0.63) Adjuvant chemotherapy Platinum-based chemotherapy LACE Meta-analysis: 5-y OS improvement of 5.4% vs no chemo Adjuvant immunotherapy (stage II-IIIA) Atezolizumab x 16 cycles (PD-L1 1%) IMpower010: Atezo vs BSC mDFS: NR vs 35.3 mo (HR, 0.66) Adjuvant targeted therapy Osimertinib x 3 y ADAURA: Osimertinib vs placebo 2-y DFS (stage II-IIIA): 90% vs 44% (HR, 0.17) Pembrolizumab x 1 y PEARLS/KEYNOTE-091: Pembro vs placebo mDFS: 53.6 vs 42.0 mo (HR, 0.76)
- 2. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/RYC40 Stage IIIA (unresectable) or IIIB/C Definitive chemoradiation → durvalumab Concurrent platinum-based chemotherapy and radiation with consolidation durvalumab PACIFIC: Durvalumab vs placebo mPFS: 16.8 vs 5.6 mo (HR, 0.52) BRAF V600E Dabrafenib + trametiniba BRF113928: Dabrafenib + trametinib single arm ORR: 64% (95% CI, 46-79) 2nd line: KRAS G12C Sotorasib CodeBreaK100: Sotorasib single arm ORR: 37.1% (95%CI, 29-46); mPFS: 6.8 mo ALK Alectiniba ALEX: Alectinib vs crizotinib 1-y PFS: 68.4% vs 48.7% (HR, 0.47) Brigatiniba ALTA-1L: Brigatinib vs crizotinib mPFS: 24 vs 11.1 mo (HR, 0.48) Lorlatiniba CROWN: Lorlatinib vs crizotinib mPFS: NR vs 9.3 mo, (HR, 0.28); 1-y PFS: 78% vs 39% Ceritinib ASCEND-4: Ceritinib vs chemo mPFS: 16.6 vs 8.1 mo (HR, 0.55) Crizotinib PROFILE 1007: Crizotinib vs chemo mPFS: 7.7 vs 3 mo (HR, 0.49) NTRK Larotrecteniba Entrectiniba ALKA/STARTRK: Entrectinib single arm ORR: 70% (NSCLC) RET Selpercatiniba LIBRETTO-001: Selpercatinib single arm ORR: 64%; mDOR: 17.5 mo Pralsetiniba ARROW: Pralsetinib single arm ORR: 61% (95% CI, 50-71) 2nd line: EGFR (ex20) Amivantamab CHRYSALIS: Amivantamab single arm CBR: 74% (95%CI, 63-83); mPFS: 8.3 mo Mobocertinib AP32788-15-101: Mobocertinib single arm DCR: 78% (95% CI, 69-85); mPFS: 7.3 mos ROS1 Crizotiniba PROFILE 1001: Crizotinib single arm ORR: 72% (95% CI, 58-84) Entrectiniba ALKA STARTRK: Entrectinib single arm ORR: 67.1%; mPFS: 19 mo Ceritinib YONSEI: Ceritinib single arm ORR: 67% (95% CI, 48-81) EGFR (ex19 del or L858R) Osimertiniba FLAURA: Osimertinib vs erlotinib/gefitinib mPFS: 18.9 vs 10.2 mo (HR, 0.46) Erlotinib EURTAC: Erlotinib vs chemo mPFS: 9.7 vs 5.2 mo (HR, 0.37) Afatinib LUX-Lung 3: Afatinib vs cis/pemetrexed mPFS: 13.6 vs 6.9 mo (HR, 0.47) Gefitinib IFUM: Gefitinib single arm mPFS: 9.7 mo Dacomitinib ARCHER 1050: Dacomitinib vs geftinib mOS: 34.1 vs 27 mo (HR, 0.75) Erlotinib + ramucirumab RELAY: Erlotinib + ramucirumab vs elotinib mPFS: 19.4 vs 12.4 mo (HR, 0.59) Erlotinib + bevacizumab ARTEMIS-CTONG1509: Erlotinib + bevacizumab vs erlotinib mPFS: 17.9 vs 11.2 mo (HR, 0.55) MET (exon 14) Capmatiniba GEOMETRY mono-1: Capmatinib single arm mPFS: 12.4 mo Tepotiniba VISION: Tepotinib single arm mPFS: 8.5-11 mo 2nd line: HER2 Trastuzumab deruxtecan DESTINY-Lung01 T-DXd single arm ORR: 55% (95% CI, 44-65); mPFS: 8.2 mo T1-2, N2–3, M0 T3, N1–3, M0 T4, N0–3, M0 Tx Nx M1 Actionable mutation detected • EGFR (ex19, ex20ins) • ALK • ROS1 • BRAF V600E • RET • MET (ex14) • HER2 • NTRK1/2/3 • KRAS G12C Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Please see the next page for recommendations if no actionable mutation is detected Stage IV KRYSTAL-1: Adagrasib single arm ORR: 43% (95% CI, 34-53); mDOR: 8.5 mo
- 3. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/RYC40 a Denotes NCCN-preferred regimens. 1. Created by Aakash Desai, MBBS, MPH, and Matthew Ho, MD, PhD. Used with permission from the authors. PD-L1 1% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52), 12-mo; OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycle) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) IMMUNOTHERAPY MONOTHERAPY Pembrolizumab KEYNOTE-042: Pembro vs plat-based chemo mOS: 16.7 vs 12.1 mo (HR, 0.81) Ramucirumab + docetaxela REVEL: Ram/docetaxel vs docetaxel; mOS: 10.5 vs 9.1 mo (HR, 0.86) Docetaxela TAX320: Docetaxel vs vinorelbine/ifosfamide; 1-y OS: 32% vs 19% Gemcitabine DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo OS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) PD-L1 1%-49% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab+chemotherapya (carboplatin+paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150 : Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) • Cemiplimab + chemotherapy (carboplatin + pemetrexed) EMPOWER-Lung 3: Cemi + chemo vs chemo mOS: 21.9 vs 13 mo (HR, 0.7) PD-L1 50% IMMUNOTHERAPY MONOTHERAPY Pembrolizumaba KEYNOTE-024: Pembro vs platinum-based chemo mPFS: 10.3 vs 6 mo (HR, 0.50) Atezolizumaba IMpower110: Atezo vs platinum-based chemo mOS: 20.1 vs 13.1 mo (HR, 0.59) Cemiplimaba EMPOWER-Lung1: Cemi vs platinum-based chemo mPFS: 8.2 vs 5.7 mo; mOS: NR vs 14.2 mo (HR, 0.57) IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + pemetrexed) KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) No actionable mutation detected (stratify based on PD-L1 staining %) Second-line therapy
