Bringing Precision Lung Cancer Care to the Perioperative Space: How to Maximize the Impact of EGFR-Targeted Therapy in Resectable NSCLC
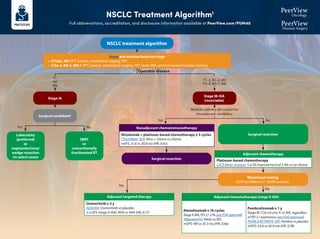
- 1. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PUM40 Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Surgical candidate? Lobectomy (preferred) or segmentectomy/ wedge resection (in select cases) SBRT or conventionally fractionated RT Surgical resection Mutational testing EGFR ex19del/ex21 L858R present? Surgical resection T1 N0 M0 Operable disease Yes Yes Yes No No No Multidisciplinary discussion for neoadjuvant candidacy T1–2, N1–2, M0 T3–4, N0–1, M0 Neoadjuvant chemoimmunotherapy Adjuvant chemotherapy Platinum-based chemotherapy LACE Meta-analysis: 5-y OS improvement of 5.4% vs no chemo Adjuvant immunotherapy (stage II-IIIA) Atezolizumab x 16 cycles StageII-IIIA,PD-L1≥1%(seeFDAapproval) IMpower010: Atezo vs BSC mDFS: NR vs 35.3 mo (HR, 0.66) Pembrolizumab x 1 y Stage IB (T2a ≥4 cm), II, or IIIA, regardless of PD-L1 expression (see FDA approval) PEARLS/KEYNOTE-091: Pembro vs placebo mDFS: 53.6 vs 42.0 mo (HR, 0.76) Adjuvant targeted therapy Osimertinib x 3 y ADAURA: Osimertinib vs placebo 2-y DFS (stage II-IIIA): 90% vs 44% (HR, 0.17) NSCLC treatment algorithm Stage IB-IIIA (resectable) Stage IA Nivolumab + platinum-based chemotherapy x 3 cycles CheckMate -816: Nivo + chemo vs chemo mEFS: 31.6 vs 20.8 mo (HR, 0.63)
- 2. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PUM40 Stage IIIA (unresectable) or IIIB/C Definitive chemoradiation → durvalumab Concurrent platinum-based chemotherapy and radiation with consolidation durvalumab PACIFIC: Durvalumab vs placebo mPFS: 16.8 vs 5.6 mo (HR, 0.52) BRAF V600E Dabrafenib + trametiniba BRF113928: Dabrafenib + trametinib single arm ORR: 64% (95% CI, 46-79) 2L: KRAS G12C Sotorasib CodeBreaK100: Sotorasib single arm ORR: 37.1% (95% CI, 29-46); mPFS: 6.8 mo ALK Alectiniba ALEX: Alectinib vs crizotinib 1-y PFS: 68.4% vs 48.7% (HR, 0.47) Brigatiniba ALTA-1L: Brigatinib vs crizotinib mPFS: 24 vs 11.1 mo (HR, 0.48) Lorlatiniba CROWN: Lorlatinib vs crizotinib mPFS: NR vs 9.3 mo, (HR, 0.28); 1-y PFS: 78% vs 39% Ceritinib ASCEND-4: Ceritinib vs chemo mPFS: 16.6 vs 8.1 mo (HR, 0.55) Crizotinib PROFILE 1007: Crizotinib vs chemo mPFS: 7.7 vs 3 mo (HR, 0.49) NTRK Larotrecteniba Entrectiniba ALKA/STARTRK: Entrectinib single arm ORR: 70% (NSCLC) RET Selpercatiniba LIBRETTO-001: Selpercatinib single arm ORR: 64%; mDOR: 17.5 mo Pralsetiniba ARROW: Pralsetinib single arm ORR: 61% (95% CI, 50–71) 2L: EGFR (ex20) Amivantamab CHRYSALIS: Amivantamab single arm CBR: 74% (95%CI, 63-83); mPFS: 8.3 mo Mobocertinib AP32788-15-101: Mobocertinib single arm DCR: 78% (95% CI, 69-85); mPFS: 7.3 mo ROS1 Crizotiniba PROFILE 1001: Crizotinib single arm ORR: 72% (95% CI, 58-84) Entrectiniba ALKA STARTRK: Entrectinib single arm ORR: 67.1%; mPFS: 19 mo Ceritinib YONSEI: Ceritinib single arm ORR: 67% (95% CI, 48-81) EGFR (ex19 del or L858R) Osimertiniba FLAURA: Osimertinib vs erlotinib/gefitinib mPFS: 18.9 vs 10.2 mo (HR, 0.46) Erlotinib EURTAC: Erlotinib vs chemo mPFS: 9.7 vs 5.2 mo (HR, 0.37) Afatinib LUX-Lung 3: Afatinib vs cis/pemetrexed mPFS: 13.6 vs 6.9 mo (HR, 0.47) Gefitinib IFUM: Gefitinib single arm mPFS: 9.7 mo Dacomitinib ARCHER 1050: Dacomitinib vs gefitinib mOS: 34.1 vs 27 mo (HR, 0.75) Erlotinib + ramucirumab RELAY: Erlotinib + ramucirumab vs erlotinib mPFS: 19.4 vs 12.4 mo (HR, 0.59) Erlotinib + bevacizumab ARTEMIS-CTONG1509: Erlotinib + bevacizumab vs erlotinib mPFS: 17.9 vs 11.2 mo (HR, 0.55) MET (exon 14) Capmatiniba GEOMETRY mono-1: Capmatinib single arm mPFS: 12.4 mo Tepotiniba VISION: Tepotinib single arm mPFS: 8.5–11 mo 2L: HER2 Trastuzumab deruxtecan DESTINY-Lung01: T-DXd single arm ORR: 55% (95% CI, 44-65); mPFS: 8.2 mo T1-2, N2–3, M0 T3, N1–3, M0 T4, N0–3, M0 Tx Nx M1 Actionable mutation detected • EGFR (ex19, ex20ins) • ALK • ROS1 • BRAF V600E • RET • MET (ex14) • HER2 • NTRK1/2/3 • KRAS G12C Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Please see the next page for recommendations if no actionable mutation is detected Stage IV Adagrasib KRYSTAL-1: Adagrasib single arm ORR: 43% (95% CI, 34-53); mDOR: 8.5 mo
- 3. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PUM40 a Denotes NCCN-preferred regimens. 1. Created by Aakash Desai, MBBS, MPH, and Matthew Ho, MD, PhD. Used with permission from the authors. PD-L1 1% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) IMMUNOTHERAPY MONOTHERAPY Pembrolizumab KEYNOTE-042: Pembro vs plat-based chemo mOS: 16.7 vs 12.1 mo (HR, 0.81) Ramucirumab + docetaxela REVEL: Ram/docetaxel vs docetaxel; mOS: 10.5 vs 9.1 mo (HR, 0.86) Docetaxela TAX320: Docetaxel vs vinorelbine/ifosfamide; 1-y OS: 32% vs 19% Gemcitabine DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo OS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) PD-L1 1%-49% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab+chemotherapya (carboplatin+paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150 : Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) • Cemiplimab + chemotherapy (carboplatin + pemetrexed) EMPOWER-Lung 3: Cemi + chemo vs chemo mOS: 21.9 vs 13 mo (HR, 0.7) PD-L1 ≥50% IMMUNOTHERAPY MONOTHERAPY Pembrolizumaba KEYNOTE-024: Pembro vs platinum-based chemo mPFS: 10.3 vs 6 mo (HR, 0.50) Atezolizumaba IMpower110: Atezo vs platinum-based chemo mOS: 20.1 vs 13.1 mo (HR, 0.59) Cemiplimaba EMPOWER-Lung1: Cemi vs platinum-based chemo mPFS: 8.2 vs 5.7 mo; mOS: NR vs 14.2 mo (HR, 0.57) IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab- paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + pemetrexed) KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) No actionable mutation detected (stratify based on PD-L1 staining %) Second-line therapy
- 4. EGFR-Targeted Therapy for Advanced- and Early-Stage EGFR-Mutated NSCLC Current Approvals and Indications1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PUM40 1. https://www.fda.gov/drugs/resources-information-approved-drugs/hematologyoncology-cancer-approvals-safety-notifications. Third-Generation Irreversible EGFR TKI Osimertinib Early Stage Third-Generation Irreversible EGFR TKI Osimertinib • 1L for EGFR exon 19 deletions or L858R mutations • Treatment of T790M-positive NSCLC with progression on or after EGFR TKI therapy EGFR/MET Bispecific Antibody Amivantamab • 2L for EGFR exon 20 insertion mutations in patients whose disease has progressed on or after platinum-based chemotherapy Irreversible EGFR/HER2 (Exon 20 Insertion) TKI Mobocertinib • 2L for EGFR exon 20 insertion mutations in patients whose disease has progressed on or after platinum-based chemotherapy EGFR TKI + VEGFR2 Antagonist Erlotinib + Ramucirumab • 1L for EGFR exon 19 deletions or L858R mutations • Adjuvant therapy after tumor resection in patients with stage IB-IIIA NSCLC whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations First-Generation Reversible EGFR TKIs Gefitinib Erlotinib • 1L for EGFR exon 19 deletions or L858R mutations • 1L for EGFR exon 19 deletions or L858R mutations Second-Generation Irreversible EGFR TKIs Afatinib Dacomitinib • 1L for EGFR exon 19 deletions or L858R, S768I, L861Q, and/or G719X mutations • 1L for EGFR exon 19 deletions or L858R mutations Metastatic
