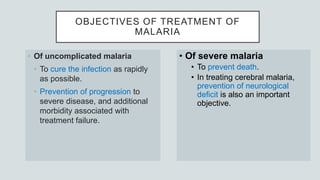
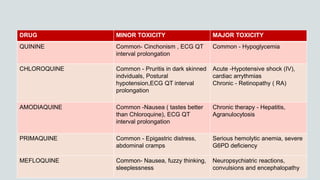
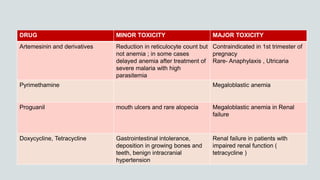
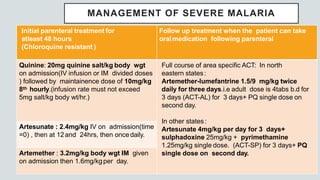
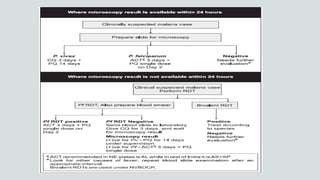
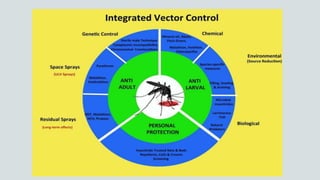
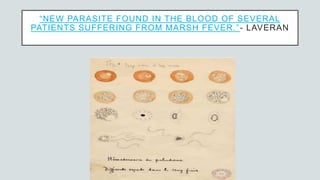
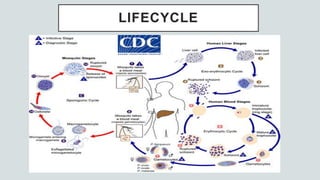
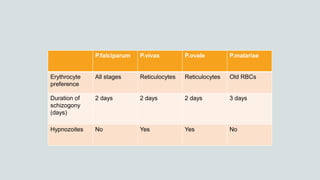
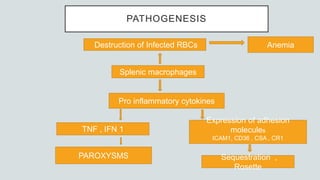
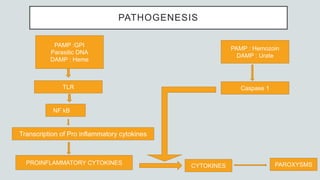
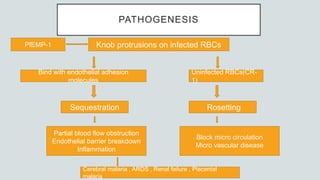
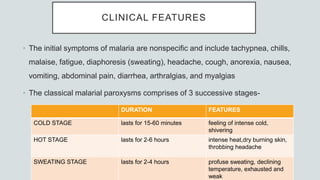
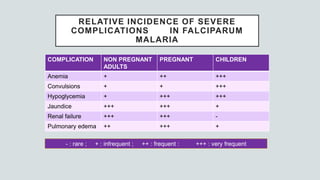
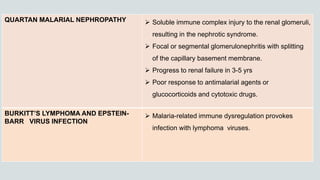
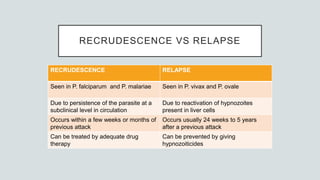
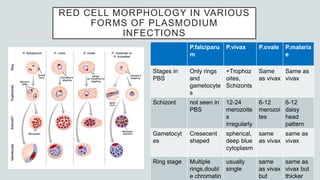
Malaria is a mosquito-borne disease caused by Plasmodium parasites. In 2018, 228 million people were infected globally resulting in over 400,000 deaths. India accounts for 3% of global malaria cases and deaths, with most cases concentrated in 7 states. Malaria is transmitted via the bites of infected female Anopheles mosquitoes. Symptoms include fever, chills, and flu-like illness, with P. falciparum infections potentially causing severe complications like cerebral malaria, severe anemia, or respiratory distress. Proper diagnosis and treatment are needed to prevent mortality from this widespread and potentially deadly disease.



















![ALGID MALARIA
• Cardiovascular abnormalities, shock (‘algid malaria’).
• The blood pressure of patients with malaria is usually at the lower end of
the normal range, although most patients are warm and well perfused.
• Severe sudden hypotension [systolic blood pressure less than 80 mmHg
in adults in the supine position] with features of circulatory failure (cold,
clammy, cyanotic skin, constricted peripheral veins, prolonged capillary
refill time)](https://image.slidesharecdn.com/malariapresentation-201020042426/85/Malaria-presentation-32-320.jpg)


















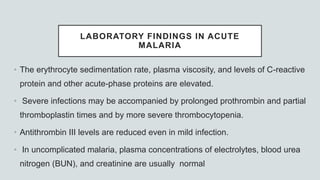

![LABORATORY FINDINGS
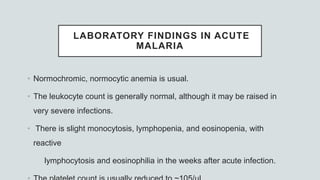
• In adults and children with cerebral malaria, CSF findings
Opening pressure at lumbar puncture is ~160mm H2O;
Slight elevation of total protein level (<200 mg/dL])
Cell count (<10/μL).
Lactate is raised
Glucose is low](https://image.slidesharecdn.com/malariapresentation-201020042426/85/Malaria-presentation-61-320.jpg)