Johnson & Johnson selected pipeline 2016
•
0 likes•50 views
SELECTED PHARMACEUTICALS IN LATE STAGE U.S. AND E.U. DEVELOPMENT OR REGISTRATION as of 4/19/16
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
Are you new to SlideShare? Are you looking to fine tune your channel plan? Are you using SlideShare but are looking for ways to enhance what you're doing? How can you use SlideShare for content marketing tactics such as lead generation, calls-to-action to other pieces of your content, or thought leadership? Read more from the CMI team in their latest SlideShare presentation on SlideShare.How To Get More From SlideShare - Super-Simple Tips For Content Marketing

How To Get More From SlideShare - Super-Simple Tips For Content MarketingContent Marketing Institute
Comments of Clinical and Microbiological Experience with Daptomycin in Chronic Osteomyelitis Treatment by Marcano-Lozada Marcel in Cohesive Journal of Microbiology & Infectious DiseaseComments of Clinical and Microbiological Experience with Daptomycin in Chroni...

Comments of Clinical and Microbiological Experience with Daptomycin in Chroni...CrimsonpublishersCJMI
Co-Chairs Srdan Verstovsek, MD, PhD, and Ruben A. Mesa, MD, FACP, prepared useful Practice Aids pertaining to myelofibrosis for this CME activity titled “Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on Molecular Biology, JAK Inhibitors, and Emerging Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3wzK6zG. CME credit will be available until October 9, 2022.Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, Naval Daver, MD, Courtney D. DiNardo, MD, MSCE, and Gail J. Roboz, MD prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME/MOC activity titled Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Diverse Patient Populations. For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3fsa7Jh. CME/MOC credit will be available until July 9, 2021.Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...

Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...PVI, PeerView Institute for Medical Education
The prognosis of patients bearing high grade glioma remains dismal. Epidermal Growth Factor Receptor (EGFR) is well validated as a primary contributor of glioma initiation and progression. Nimotuzumab is a humanized monoclonal antibody that recognizes the EGFR extracellular domain and reaches Central Nervous System tumors, in nonclinical and clinical setting. While it has similar activity when compared to other anti-EGFR antibodies, it does not induce skin toxicity or hypomagnesemia.Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...

Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...Enrique Moreno Gonzalez
More Related Content
Viewers also liked
Are you new to SlideShare? Are you looking to fine tune your channel plan? Are you using SlideShare but are looking for ways to enhance what you're doing? How can you use SlideShare for content marketing tactics such as lead generation, calls-to-action to other pieces of your content, or thought leadership? Read more from the CMI team in their latest SlideShare presentation on SlideShare.How To Get More From SlideShare - Super-Simple Tips For Content Marketing

How To Get More From SlideShare - Super-Simple Tips For Content MarketingContent Marketing Institute
Viewers also liked (10)
10 Ways to Win at SlideShare SEO & Presentation Optimization

10 Ways to Win at SlideShare SEO & Presentation Optimization
STOP! VIEW THIS! 10-Step Checklist When Uploading to Slideshare

STOP! VIEW THIS! 10-Step Checklist When Uploading to Slideshare
How To Get More From SlideShare - Super-Simple Tips For Content Marketing

How To Get More From SlideShare - Super-Simple Tips For Content Marketing
A Guide to SlideShare Analytics - Excerpts from Hubspot's Step by Step Guide ...

A Guide to SlideShare Analytics - Excerpts from Hubspot's Step by Step Guide ...
Similar to Johnson & Johnson selected pipeline 2016
Comments of Clinical and Microbiological Experience with Daptomycin in Chronic Osteomyelitis Treatment by Marcano-Lozada Marcel in Cohesive Journal of Microbiology & Infectious DiseaseComments of Clinical and Microbiological Experience with Daptomycin in Chroni...

Comments of Clinical and Microbiological Experience with Daptomycin in Chroni...CrimsonpublishersCJMI
Co-Chairs Srdan Verstovsek, MD, PhD, and Ruben A. Mesa, MD, FACP, prepared useful Practice Aids pertaining to myelofibrosis for this CME activity titled “Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on Molecular Biology, JAK Inhibitors, and Emerging Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3wzK6zG. CME credit will be available until October 9, 2022.Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, Naval Daver, MD, Courtney D. DiNardo, MD, MSCE, and Gail J. Roboz, MD prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME/MOC activity titled Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Diverse Patient Populations. For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3fsa7Jh. CME/MOC credit will be available until July 9, 2021.Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...

Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...PVI, PeerView Institute for Medical Education
The prognosis of patients bearing high grade glioma remains dismal. Epidermal Growth Factor Receptor (EGFR) is well validated as a primary contributor of glioma initiation and progression. Nimotuzumab is a humanized monoclonal antibody that recognizes the EGFR extracellular domain and reaches Central Nervous System tumors, in nonclinical and clinical setting. While it has similar activity when compared to other anti-EGFR antibodies, it does not induce skin toxicity or hypomagnesemia.Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...

Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...Enrique Moreno Gonzalez
Naval Daver, MD, and Joseph D. Khoury, MD, FCAP, prepared useful practice aids pertaining to acute myeloid leukemia for this CME/MOC/CC activity titled "The Pathology–Oncology Partnership in AML: Identifying and Treating the Diversity of Disease Subtypes." For the full presentation, complete CME/MOC/CC information, and to apply for credit, please visit us at https://bit.ly/32hvh7Z. CME/MOC/CC credit will be available until October 14, 2022.The Pathology–Oncology Partnership in AML: Identifying and Treating the Diver...

The Pathology–Oncology Partnership in AML: Identifying and Treating the Diver...PVI, PeerView Institute for Medical Education
Similar to Johnson & Johnson selected pipeline 2016 (20)
BCMACD3 Bispecific Antibodies for Multiple Myeloma (MM).pdf

BCMACD3 Bispecific Antibodies for Multiple Myeloma (MM).pdf
Comments of Clinical and Microbiological Experience with Daptomycin in Chroni...

Comments of Clinical and Microbiological Experience with Daptomycin in Chroni...
Advances in AML Care: Highlights From Recent Science.

Advances in AML Care: Highlights From Recent Science.
Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...
Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...

Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...
Azithromycin 250 mg film coated tablets smpc- taj pharmaceuticals

Azithromycin 250 mg film coated tablets smpc- taj pharmaceuticals
Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...

Radiotherapy plus nimotuzumab or placebo in the treatment of high grade gliom...
Imatinib, 400mg, 100mg, film coated tablets smpc- taj pharmaceuticals

Imatinib, 400mg, 100mg, film coated tablets smpc- taj pharmaceuticals
The Pathology–Oncology Partnership in AML: Identifying and Treating the Diver...

The Pathology–Oncology Partnership in AML: Identifying and Treating the Diver...
More from Deepa K
More from Deepa K (8)
Merck & Co., Inc. at 34th Annual J.P. Morgan Healthcare Conference

Merck & Co., Inc. at 34th Annual J.P. Morgan Healthcare Conference
Merck & Co., Inc. at UBS Global Healthcare Conference

Merck & Co., Inc. at UBS Global Healthcare Conference
Food Insecurity & Chronic Disease: Connecting Hunger & Health

Food Insecurity & Chronic Disease: Connecting Hunger & Health
Pfizer Analyst and Investor Call to Discuss Proposed Combination with Allergan

Pfizer Analyst and Investor Call to Discuss Proposed Combination with Allergan
Pfizer Analyst and Investor Call to Discuss Proposed Combination with Allergan

Pfizer Analyst and Investor Call to Discuss Proposed Combination with Allergan
Recently uploaded
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent High Profile Call Girl 24×7
Booking Contact Details
WhatsApp Chat: +91-9930245274
Mumbai Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts Mumbai understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
Flexibility
Choices and options
Lists of many beauty fantasies
Turn your dream into reality
Perfect companionship
Cheap and convenient
In-call and Out-call services
And many more.
29-04-24 (Smt)Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...Call Girls in Nagpur High Profile
Recently uploaded (20)
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...

The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...
Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...

Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...
Call Girls Visakhapatnam Just Call 8250077686 Top Class Call Girl Service Ava...

Call Girls Visakhapatnam Just Call 8250077686 Top Class Call Girl Service Ava...
Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...
Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Faridabad Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Faridabad Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available
Premium Call Girls Cottonpet Whatsapp 7001035870 Independent Escort Service

Premium Call Girls Cottonpet Whatsapp 7001035870 Independent Escort Service
Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...

Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...
Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...

Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...
Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...

Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...
Call Girls Ludhiana Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Ludhiana Just Call 9907093804 Top Class Call Girl Service Available
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...
Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available
Johnson & Johnson selected pipeline 2016
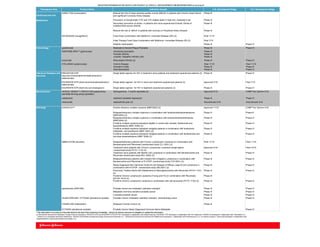
- 1. Therapeutic Area Product Name Indication Sought U.S. Development Stage E.U. Development Stage Cardiovascular and XARELTO® (rivaroxaban) Reduce the risk of major adverse cardiac events (MACE) in patients with Chronic Heart Failure and significant Coronary Artery Disease Phase III Metabolism Prevention of Symptomatic VTE and VTE-related death in high-risk, medically ill pts Phase III Secondary prevention of stroke in patients who have experienced Embolic Stroke of undetermined source (ESUS) Phase III Reduce the risk of MACE in patients with coronary or Peripheral Artery Disease Phase III INVOKANA® (canagliflozin) Fixed Dose Combination with Metformin, Extended Release (XR) (2) Filed 11/15 Initial Therapy Fixed Dose Combination with Metformin, Immediate Release (IR) (2) Filed 7/15 Diabetic nephropathy Phase III Phase III Immunology guselkumab Moderate to Severe Plaque Psoriasis Phase III Phase III SIMPONI® ARIA™ (golimumab) Ankylosing spondylitis Phase III Psoriatic arthritis Phase III Juvenile Idiopathic Arthritis (JIA) Phase III sirukumab Rheumatoid Arthritis (2) Phase III Phase III STELARA® (ustekinumab) Crohn's Disease Filed 11/15 Filed 11/15 Ulcerative Colitis Phase III Phase III Axial Spondylytis Phase III Phase III Infectious Diseases and Vaccines PREZISTA® STR (darunavir/cobicistat/emtricitabine/tenofovir alafenamide) Single tablet regimen for HIV in treatment naïve patients and treatment experienced patients (2) Phase III Phase III EDURANT® STR (rilpirivine/emtricitabine/tenofovir alafenamide) Single tablet regimen for HIV in naïve and treatment experienced patients (2) Approved 3/16 Filed 7/15 EDURANT® STR (rilpirivine and dolutegravir) Single tablet regimen for HIV in treatment experienced patients (2) Phase III Phase III Neuroscience INVEGA TRINZA™ /TREVICTA® (paliperidone palmitate IM long acting injectable) Schizophrenia - 3 month injectable (2) Approved 5/15 CHMP Pos Opinion 4/16 esketamine treatment-restistant depression Phase III Phase III fulranumab osteoarthritis pain (2) Discontinued 3/16 Discontinued 3/16 Oncology DARZALEX™ Double refractory multiple myeloma (MMY2002) (2) Approved 11/15 CHMP Pos Opinion 4/16 Relapsed/refractory multiple myeloma in combination with lenalidomide/dexamethasone (MMY3003) (2) Phase lll Phase lll Relapsed/refractory multiple myeloma in combination with bortezomib/dexamethasone (MMY3004) (2) Phase III Phase III Frontline multiple myeloma transplant eligible in combo with velcade, thalidomide and dexamethasone (MMY 3006) (2) Phase III Phase III Frontline multiple myeloma transplant ineligible patients in combination with bortezomib, melphalan, and prednisone (MMY 3007) (2) Phase III Phase III Frontline multiple myeloma transplant ineligible patients in combination with lenalidomide and low dose dexamethasone (MMY 3008) (2) Phase III Phase III IMBRUVICA® (ibrutinib) Relapsed/refractory patients with Chronic Lymphocytic Leukemia (in combination with Bendamustine and Rituximab) (randomized study CLL-3001) (2) Filed 11/15 Filed 11/15 Treatment naïve patients with Chronic Lymphocytic Leukemia (single agent) Approved 3/16 Filed 10/15 (randomized study PCYC-1115) (2) Treatment naïve patients with Mantle Cell Lymphoma in combination with Bendamustine and Rituximab (randomized study MCL-3002) (2) Phase III Phase III Relapsed/refractory patients with Indolant Non-Hodgkins Lymphoma in combination with Bendamustine and Rituximab or R-CHOP; (randomized study FLR-3001) (2) Phase III Phase III Newly Diagnosed Non-Germinal Center B-Cell Subtype of Diffuse Large B-Cell Lymphoma in combination with R-CHOP (randomized study DBL3001) (2) Phase III Phase III Previously Treated Adults with Waldenstrom's Macroglobulinemia with Rituximab (PCYC-1127) (2) Phase III Phase III Frontline Chronic Lymphocytic Leukemia (Young and Fit) (in combination with Rituximab) (ECOG 1912) (2) Phase III Frontline Chronic Lymphocitic Leukemia in combination with obinutuzumab (PCYC-1130) (2) Phase III Phase III apalutamide (ARN-509) Prostate cancer pre-metastatic castration-resistant Phase III Phase III Metastatic hormone sensitive prostate cancer Phase III Phase III Localized prostate cancer Phase III Phase III JNJ927/ARN-509 / ZYTIGA® (abiraterone acetate) Prostate Cancer metastatic castration resistant chemotherapy naïve Phase III Phase III YONDELIS® (trabectedin) Relapsed Ovarian Cancer (2) Phase III ZYTIGA® (abiraterone acetate) Prostate Cancer Newly Diagnosed Hormone Naïve Metastatic Phase III * This information is accurate as of the date hereof to the best of the Company's knowledge. Johnson & Johnson assumes no obligation to update this information. (2) INVOKANA licensed from Mitsubishi Tanabe Pharma Corporation; PREZISTA STR and EDURANT STR (R/F/TAF) developed in collaboration with Gilead Sciences; EDURANT STR developed in collaboration with ViiV Healthcare; YONDELIS developed in collaboration with PharmaMar S.A.; XARELTO co-developed with Bayer HealthCare; INVEGA SUSTENNA includes technology licensed from Alkermes, Inc.; DARZALEX licensed from Genmab A/S; IMBRUVICA devloped in collaboration with Pharmacyclics, LLC, an AbbVie company; Sirukumab developed in collaboration with GlaxoSmithKline; fulranumab licensed from Amgen, Inc. SELECTED PHARMACEUTICALS IN LATE STAGE U.S. AND E.U. DEVELOPMENT OR REGISTRATION as of 4/19/16
- 2. YONDELIS® (US) (trabectedin) 2nd Line soft tissue sarcoma DARZALEX™ (US) (daratumumab) Double Refractory multiple myeloma Daratumumab (EU)* Double Refractory multiple myeloma (* CHMP Positive Opinion) Sirukumab Rheumatoid arthritis Guselkumab Psoriasis Esketamine Treatment resistant depression JNJ-922 (Orexin-2 antagonist) Primary insomnia Neuroscience JNJ-927 (ARN-509) pre-metastatic prostate cancer Imetelstat Relapsed/refractory myelofibrosis JNJ-493 (FGFRI kinase inhibitor) Urothelial cancer Monovalent Ebola Vaccine regimen JNJ-872 Influenza A AL-8176 RSV infection 3DAA (ACH-3102 - AL-335 - SMV) HCV Infectious Diseases & Vaccines • Filings/approvals assumed to be in the US and EU unless otherwise noted. This information is accurate as of the date hereof to the best of the Company’s knowledge. Johnson & Johnson assumes no obligation to update this information. Sirukumab developed in collaboration with GlaxoSmithKline; DARZALEX licensed from Genmab A/S; YONDELIS developed in collaboration with Pharma Mar S.A.; Orexin – 2 antagonist codeveloped with Minerva Neurosciences; Imetelstat licensed from Geron; JNJ-872 licensed from Vertex Pharmaceuticals, Inc.; ACH-3102 licensed From Achillion Pharmaceuticals, Inc., Ebola vaccine codeveloped with Bavarian Nordic. Oncology APPROVED 2015/2016 IN REGISTRATION Janssen Pharmaceutical Companies of Johnson & Johnson Selected NME Pharmaceutical Pipeline - Recent Approvals/Potential Filings* Selective Highlights as of April 19, 2016 PLANNED FILINGS 2015-2019* Oncology Immunology Oncology
