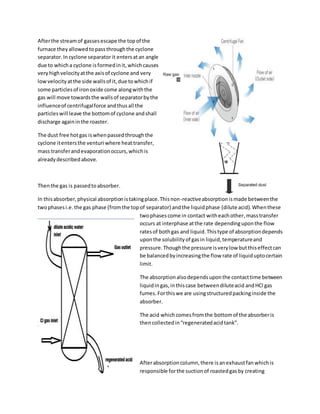
This document summarizes the processes at an acid regeneration plant and a PSA nitrogen and hydrogen gas production plant. The main objectives of the acid regeneration plant are to regenerate spent acid from steel pickling to over 99% purity and produce a valuable iron oxide byproduct. The PSA plant uses compressed air and ammonia cracking to produce high purity nitrogen and hydrogen gases for various industrial processes. Both plants utilize various unit operations like filtration, heating, cooling, absorption and adsorption to achieve the desired product specifications.