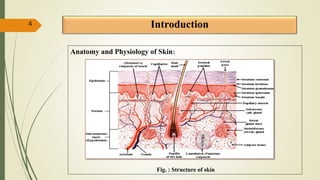
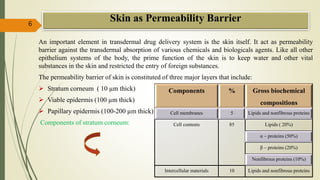
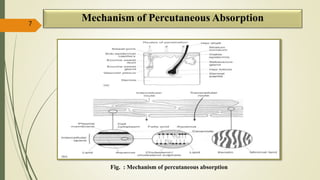
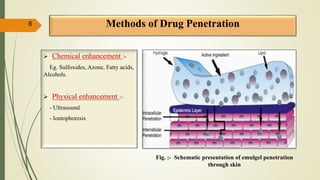
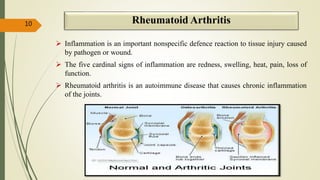
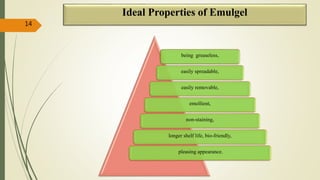
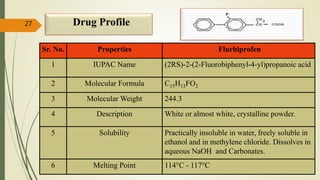
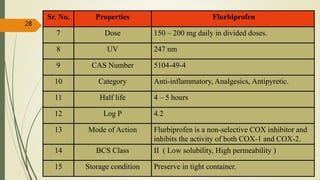
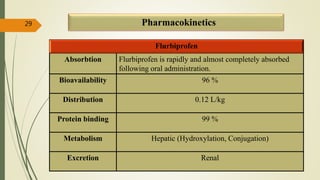
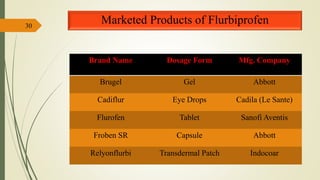
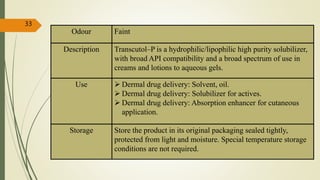
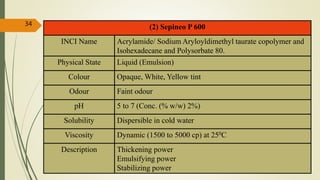
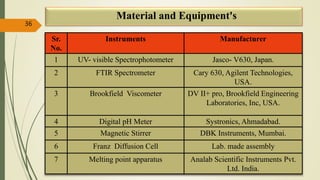
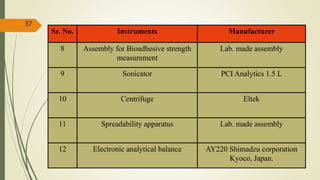
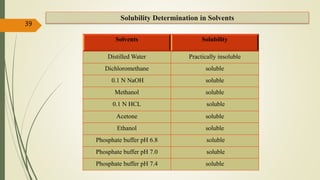
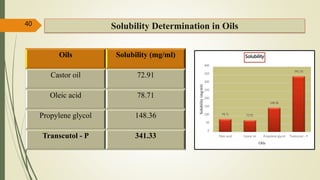
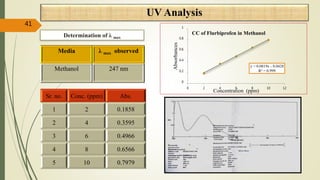
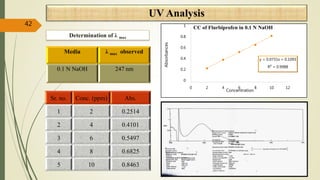
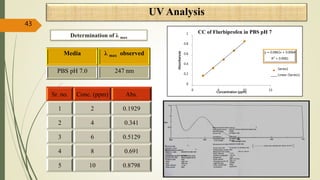
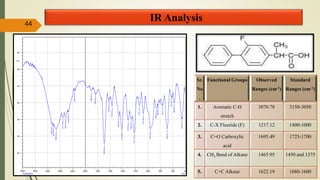
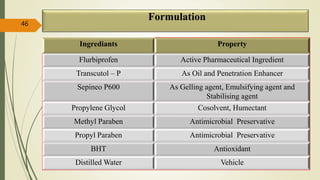
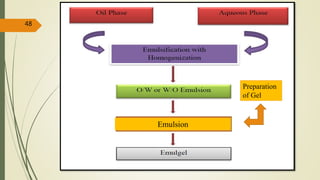
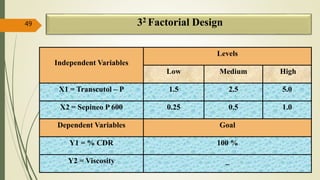
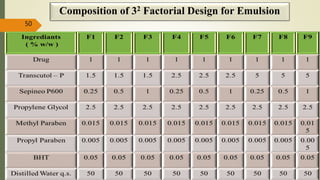
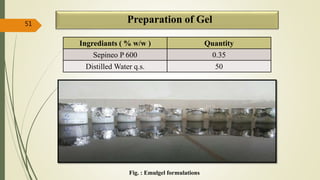
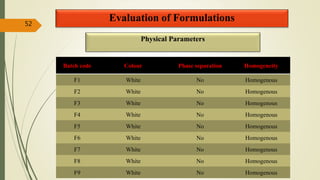
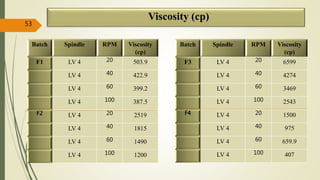
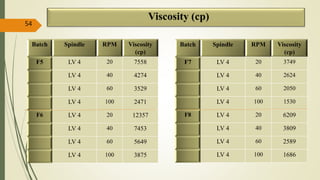
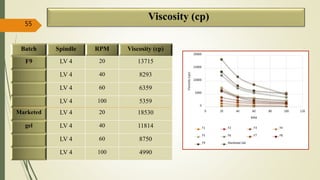
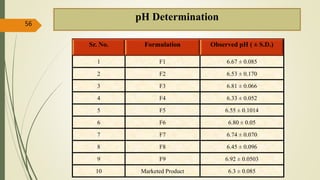
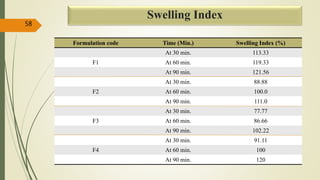
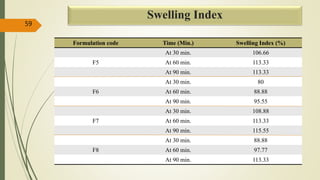
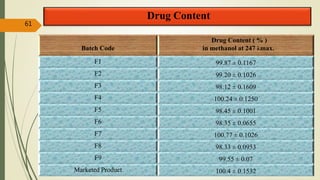
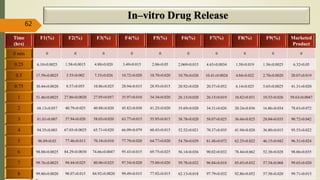
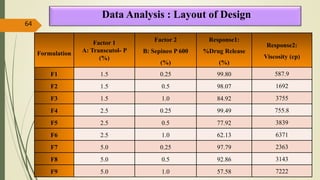
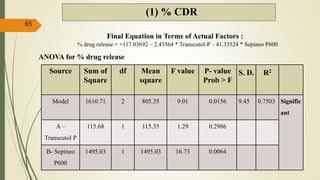
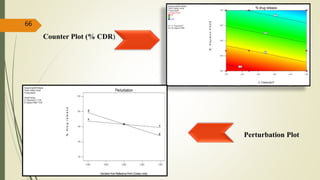
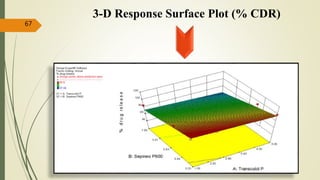
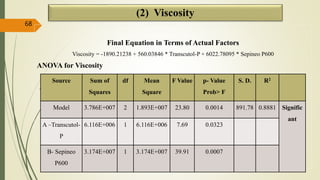
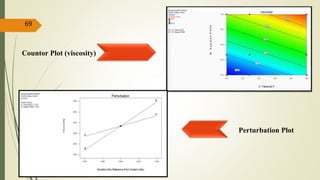
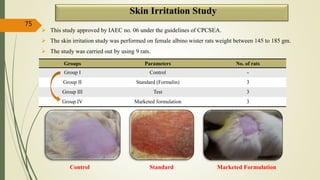
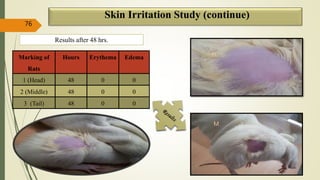
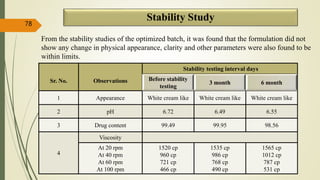
This document presents a formulation and evaluation of flurbiprofen loaded emulgel. It begins with an introduction to transdermal drug delivery systems and their advantages over other routes of administration. It then discusses skin anatomy and physiology, the mechanisms of percutaneous absorption, and factors affecting topical absorption. The document provides background on rheumatoid arthritis and defines emulgel. It discusses the ideal properties of emulgel and provides advantages and disadvantages. The objectives and plan of work are then outlined which include literature review, preformulation studies, formulation development, evaluation studies, and stability testing. Finally, the document presents a literature review of several other emulgel formulations.