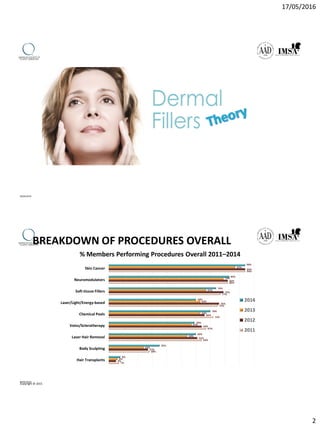
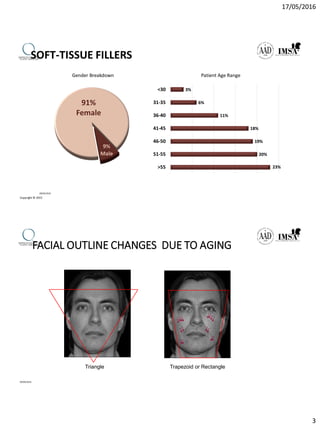
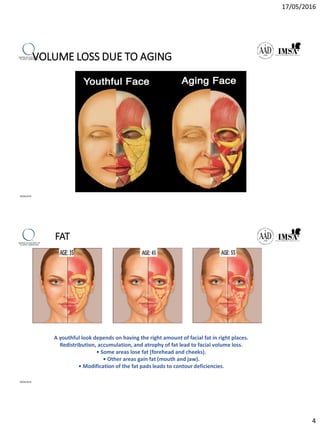
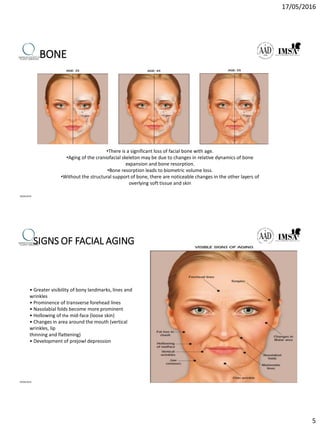
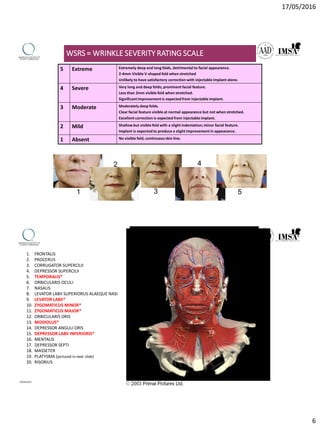
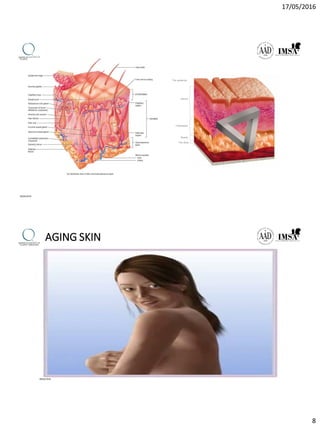
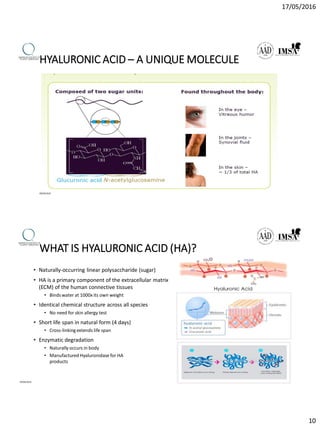
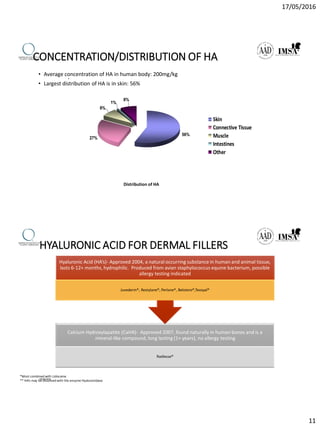
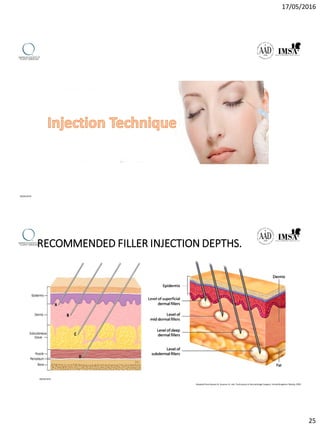
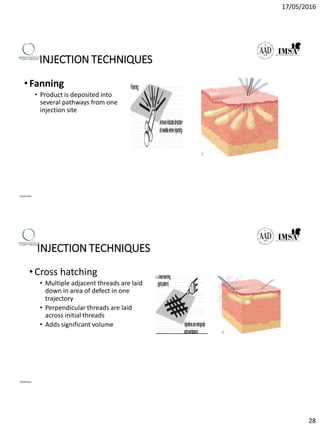
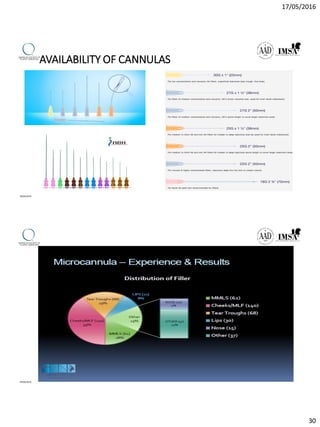
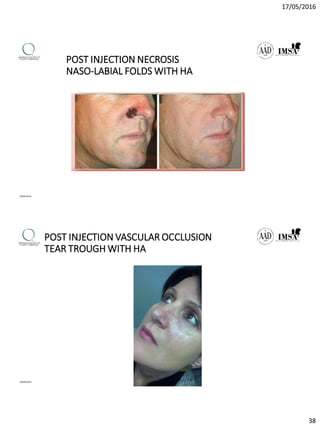
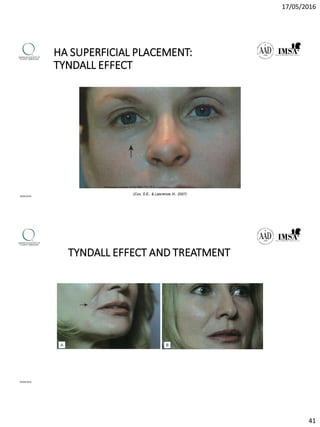
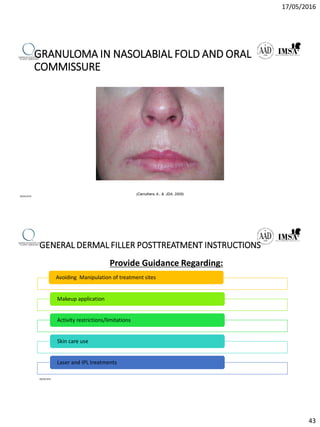
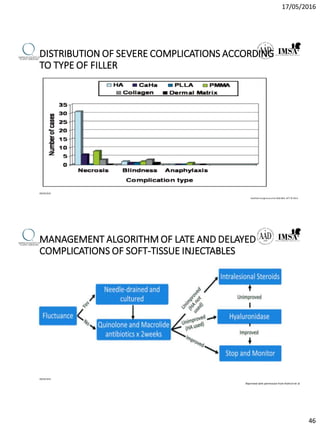
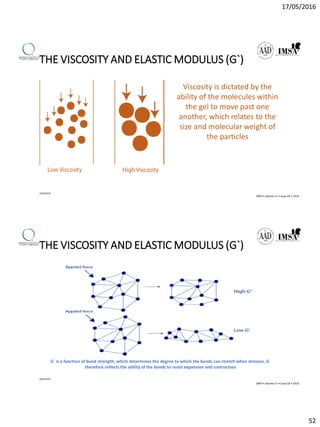
The document provides a comprehensive overview of dermal fillers, detailing their types, injection techniques, anatomical considerations, and complications related to their use. It emphasizes the aging process impacts on facial structure, the benefits of various fillers, and the techniques for optimal application. Additionally, it discusses the properties of hyaluronic acid and other filler materials, along with managing adverse reactions post-treatment.