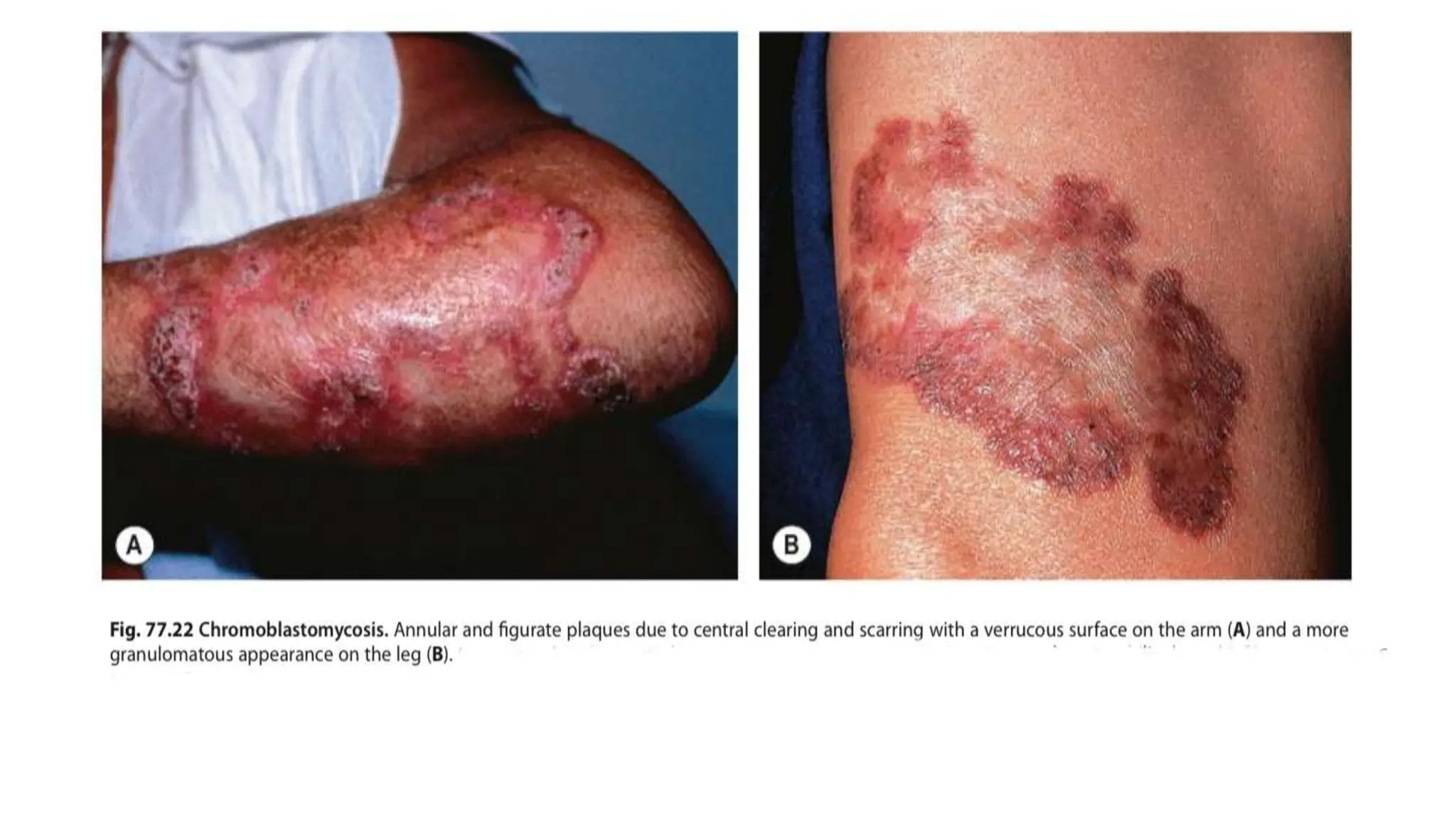
This document discusses several types of deep fungal infections:
1. Subcutaneous mycoses like sporotrichosis, chromoblastomycosis, and mycetoma which are caused by fungi entering through the skin.
2. Systemic mycoses like histoplasmosis which are acquired by inhalation and can disseminate through the bloodstream. Histoplasmosis is caused by Histoplasma capsulatum and presents as pulmonary infection, disseminated infection affecting organs, or cutaneous lesions. Treatment involves antifungals like amphotericin B or itraconazole.
3. Rarer infections like lobomycosis caused by Lacazia loboi which