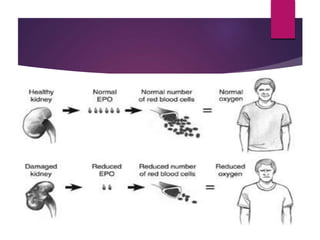
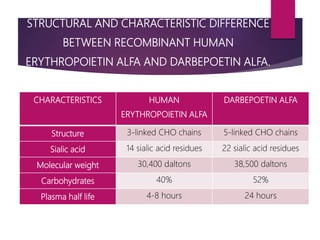
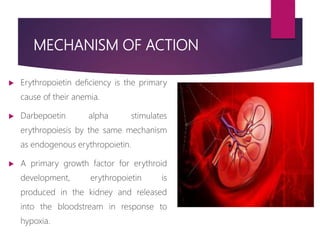
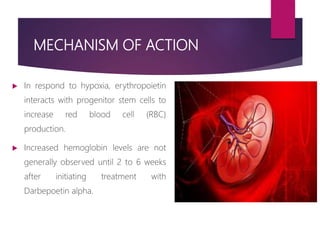
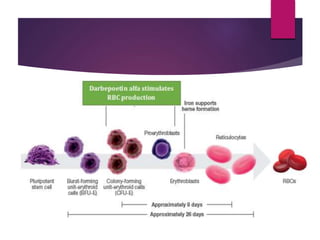
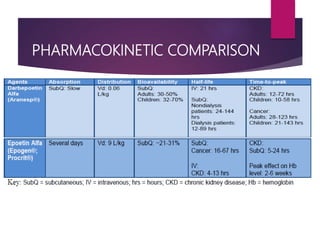
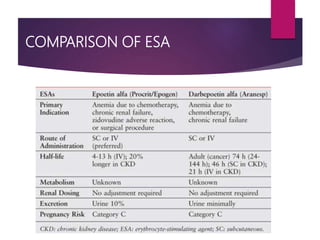
Darbepoetin alfa is an erythropoiesis-stimulating agent used to treat anemia. It is created through recombinant DNA technology and has 5 sugar chains, whereas erythropoietin has 3 chains. This longer circulating half-life allows darbepoetin alfa to be administered less frequently than erythropoietin. Clinical studies have shown darbepoetin alfa is effective at maintaining hemoglobin levels in patients with chronic kidney disease on dialysis or not on dialysis when administered weekly or every two weeks. Darbepoetin alfa is generally well-tolerated, with hypertension and dyspnea being the most common adverse reactions.






















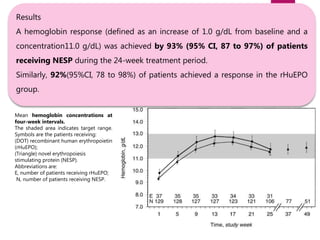

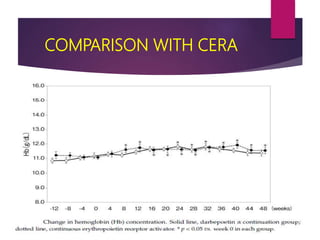
![Darbepoetin alfa administered to achieve and maintain target
hemoglobin levels for 1 year in EPO-naive patients with CKD-ND.
© Copyright
Fresenius Kabi
AG
OBJECTIVE: To assess the efficacy and safety of every-other-week darbepoetin alfa therapy in treating anemia
and maintaining hemoglobin levels for 1 year in patients with chronic kidney disease (CKD) who were not
undergoing dialysis and who had not previously received erythropoesis-stimulating proteins (ESPs).
PATIENTS AND METHODS:
• Multicenter 52-week study,
• Patients with CKD who were not receiving dialysis (creatinine
clearance < or =70 mL/min or estimated glomerular
filtration rate [GFR] < or =60 mL/min).
• Patients not receiving ESPs, had hemoglobin concentrations
<11 g/dL, and had transferrin saturation of 20% or higher
during screening.
• Patients initiated every-other-week darbepoetin alfa therapy
at 0.75 microg/kg, with the dose subsequently titrated to
maintain hemoglobin levels not to exceed 12 g/dL.
RESULTS:
• The least squares mean evaluation Hb concentration
was 11.54 g/dL and the change from baseline was 1.6
g/dL
• The mean (SD) every-other-week darbepoetin alfa dose
during evaluation was 44.5 (33.7) mcg.
• Darbepoetin alfa was well tolerated throughout the
study period
CONCLUSION: Darbepoetin alfa initiated every other week safely and effectively treated anemia and
maintained hemoglobin for 1 year in patients with CKD who were not undergoing dialysis and who were not
receiving prior ESP therapy.
Am J Nephrol. 2006;26(2):149-56.](https://image.slidesharecdn.com/darbepoetinscientificinformationandcomparison-180315055333/85/Darbepoetin-scientific-information-and-comparison-40-320.jpg)