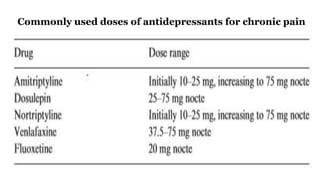
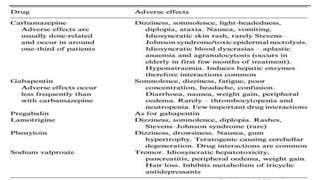
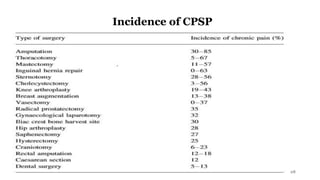
This document discusses chronic pain management and treatment options. It notes that chronic pain is influenced by many physical and psychological factors, requiring multimodal interventions. Common drug classes used include antidepressants, anticonvulsants, opioids, and local anesthetics. Antidepressants are effective for neuropathic pain and may enhance inhibitory pathways and act on other receptors. Anticonvulsants are commonly used for neuropathic pain by reducing neuronal excitability. Opioids can be effective but have side effects like nausea and constipation. Non-pharmacological therapies and more advanced interventions are also discussed.