Changes and Modifications
•
0 likes•3,994 views
This document summarizes regulations for changes and modifications to registered medical devices across Russia, Kazakhstan, and Belarus. It outlines three procedures - amendment, notification, and re-registration. An amendment procedure allows for name, address, or accessory changes and is fastest in Russia and Kazakhstan but requires full re-registration in Belarus. A notification procedure in Russia addresses certain indication, storage, marking, or software changes and is approved within 30 days. All other changes generally require the full re-registration process.
Report
Share
Report
Share
Recommended
Medical device approval chart for Russia - EMERGO

The regulatory process for medical devices in Russia involves classifying the device, appointing an authorized representative, submitting a registration dossier including technical documentation and clinical data to Roszdravnadzor (RZN), addressing any requests for additional information, and ultimately receiving a registration certificate to market the device in Russia. The process can take 6-12 months depending on the device class.
Taiwan medical device registration and approval chart - EMERGO

Taiwan regulates medical devices through the Pharmaceutical Affairs Act and Regulations for Governing the Management of Medical Devices. The process involves classifying the device, appointing a Taiwan agent, preparing quality system documentation for submission, and obtaining approval. Device classification and complexity of approval requirements vary, with Class I generally having the simplest process taking 1-2 months, Class II taking 10-12 months, and Class III taking 10-12 months and requiring a committee review.
How to Get a Medical Device Approved According to New Ukrainian Regulations

1. To get a medical device approved in Ukraine, manufacturers must appoint an authorized representative in Ukraine who can be a distributor or representative office. The manufacturer then identifies the appropriate technical regulations for the device and chooses a conformity assessment procedure.
2. The manufacturer prepares a technical file demonstrating compliance with the requirements in the chosen regulations. They then select a Notified Body to review the file and certify compliance with quality standards.
3. Upon approval, the authorized representative's name and address and the Ukrainian conformity assessment mark must be placed on the device labels and packaging.
Eurasian integration alexey stepanov-informa v20150619

The document discusses upcoming harmonization of medical device regulations within the Eurasian Economic Union (EEU) between Russia, Belarus, and Kazakhstan. Key points include:
- A common medical device market within the EEU is expected to launch on January 1, 2016 under new harmonized rules.
- Many regulations still need to be adopted by the end of 2015 to provide guidance for implementation.
- Transition periods and potential delays mean full harmonization may not be achieved immediately.
- Manufacturers will need to reallocate resources to adjust to the new regulatory system over time, including re-registering products under the new rules by 2022.
5 questions on safety reporting for medical devices in CIS region

This document summarizes safety reporting requirements for medical devices in CIS countries, including Russia, Belarus, Ukraine, and Kazakhstan. It outlines what information must be reported, the regulations requiring reporting, reporting deadlines, and how and where to report. The key information that must be reported includes adverse reactions, undesirable events during usage, circumstances endangering patient or health professional life or health, and device interactions. Regulations and reporting bodies differ by country but generally require reporting within 10-20 days to the relevant health ministry or inspectorate.
Medical Device Labeling in Russia

The document discusses medical device labeling requirements in Russia. It outlines several key Russian laws and regulations pertaining to medical device registration, customs clearance, advertising, sales, distribution, and use. It emphasizes that labeling must be in Russian and match the information in the registration certificate. It recommends using over-labeling with essential information in Russian as the most common way for foreign manufacturers to comply with labeling language requirements. Failure to properly translate labeling could result in seized products or additional taxes and fees.
Recommended
Medical device approval chart for Russia - EMERGO

The regulatory process for medical devices in Russia involves classifying the device, appointing an authorized representative, submitting a registration dossier including technical documentation and clinical data to Roszdravnadzor (RZN), addressing any requests for additional information, and ultimately receiving a registration certificate to market the device in Russia. The process can take 6-12 months depending on the device class.
Taiwan medical device registration and approval chart - EMERGO

Taiwan regulates medical devices through the Pharmaceutical Affairs Act and Regulations for Governing the Management of Medical Devices. The process involves classifying the device, appointing a Taiwan agent, preparing quality system documentation for submission, and obtaining approval. Device classification and complexity of approval requirements vary, with Class I generally having the simplest process taking 1-2 months, Class II taking 10-12 months, and Class III taking 10-12 months and requiring a committee review.
How to Get a Medical Device Approved According to New Ukrainian Regulations

1. To get a medical device approved in Ukraine, manufacturers must appoint an authorized representative in Ukraine who can be a distributor or representative office. The manufacturer then identifies the appropriate technical regulations for the device and chooses a conformity assessment procedure.
2. The manufacturer prepares a technical file demonstrating compliance with the requirements in the chosen regulations. They then select a Notified Body to review the file and certify compliance with quality standards.
3. Upon approval, the authorized representative's name and address and the Ukrainian conformity assessment mark must be placed on the device labels and packaging.
Eurasian integration alexey stepanov-informa v20150619

The document discusses upcoming harmonization of medical device regulations within the Eurasian Economic Union (EEU) between Russia, Belarus, and Kazakhstan. Key points include:
- A common medical device market within the EEU is expected to launch on January 1, 2016 under new harmonized rules.
- Many regulations still need to be adopted by the end of 2015 to provide guidance for implementation.
- Transition periods and potential delays mean full harmonization may not be achieved immediately.
- Manufacturers will need to reallocate resources to adjust to the new regulatory system over time, including re-registering products under the new rules by 2022.
5 questions on safety reporting for medical devices in CIS region

This document summarizes safety reporting requirements for medical devices in CIS countries, including Russia, Belarus, Ukraine, and Kazakhstan. It outlines what information must be reported, the regulations requiring reporting, reporting deadlines, and how and where to report. The key information that must be reported includes adverse reactions, undesirable events during usage, circumstances endangering patient or health professional life or health, and device interactions. Regulations and reporting bodies differ by country but generally require reporting within 10-20 days to the relevant health ministry or inspectorate.
Medical Device Labeling in Russia

The document discusses medical device labeling requirements in Russia. It outlines several key Russian laws and regulations pertaining to medical device registration, customs clearance, advertising, sales, distribution, and use. It emphasizes that labeling must be in Russian and match the information in the registration certificate. It recommends using over-labeling with essential information in Russian as the most common way for foreign manufacturers to comply with labeling language requirements. Failure to properly translate labeling could result in seized products or additional taxes and fees.
Ukraine: What information is mandatory on medical device label? 

The document outlines the mandatory information that must be included on medical device labels for products registered in Ukraine. This includes: 1) the name of the item in Ukrainian, 2) the manufacturer name and address in original language and country of origin in Ukrainian, 3) the registration certificate number and date. Optional information that can be included are main properties, sterility, guarantees, safety warnings, and manufacturer's Ukrainian representative. The label must be in Ukrainian and can include symbols for standards compliance.
Localization 2014

The Russian Ministry of Industry and Trade reconsidered restrictions on the supply of medical devices for state procurement in July 2014. The restrictions affect state and municipal procurements of medical devices from a closed list. If at least two bids with local products from Russia, Belarus or Kazakhstan are submitted for tender, buyers will be unable to procure medical devices originating from other countries. The local products must be confirmed by a certificate of origin issued by a local authorized agency. Certain categories of medical devices are subject to the restrictions, including syringes, osteosynthesis implants, ECG devices, and surgical instruments.
How Can You Group Medical Devices for Registration in Russia? 

Slide explains the principles of product grouping for submission for registration of medical device in RosZdravNadzor (Russia).
How to provide proper photos for medical device registration dossier? 

The document provides guidelines for submitting photo documentation of medical devices for registration purposes, requiring at least one high resolution photo sized 18x24cm showing the device and all accessories, with each item clearly identified either through graphic labeling or direct labeling on the photo. For similar devices of different sizes, one photo is acceptable, and for dossiers covering multiple different devices, one photo per type is sufficient along with a product catalog.
Let us put some clarity on VAT topic

This document outlines the value-added tax (VAT) rates for medical devices in Russia both past and present. It states that historically, medical equipment was subject to a 0% VAT rate, medical products had an 18% VAT rate, and the exact lists of qualifying items were defined in government orders from 2002 and 2008. As of January 1, 2013, a new law unified these terms as "medical devices" and introduced some VAT rate changes, making the VAT rules less clear. The document concludes that additional low-level legislative changes on medical device VAT rates in Russia may still be forthcoming.
Russia: What information is mandatory on medical device label? 

This document summarizes the mandatory information that must be included on medical device labels in Russia. The label must include:
1. The name of the medical device in Russian.
2. Information about the manufacturer including the word "manufacturer" in Russian, the name and address of the manufacturer in the original language, and the country of origin in Russian.
3. The registration number and date of registration for the device in Russia.
4. Information about the device's properties in Russian such as size, sterility, single-use instructions, storage conditions, and shelf life.
Simple Steps to Make Her Choose You Every Day

Simple Steps to Make Her Choose You Every Day" and unlock the secrets to building a strong, lasting relationship. This comprehensive guide takes you on a journey to self-improvement, enhancing your communication and emotional skills, ensuring that your partner chooses you without hesitation. Forget about complications and start applying easy, straightforward steps that make her see you as the ideal person she can't live without. Gain the key to her heart and enjoy a relationship filled with love and mutual respect. This isn't just a book; it's an investment in your happiness and the happiness of your partner
Sexual Disorders.gender identity disorderspptx

Gender identity disorder, paraphilias , sexual dysfunction
2024 Media Preferences of Older Adults: Consumer Survey and Marketing Implica...

When it comes to creating marketing strategies that target older adults, it is crucial to have insight into their media habits and preferences. Understanding how older adults consume and use media is key to creating acquisition and retention strategies. We recently conducted our seventh annual survey to gain insight into the media preferences of older adults in 2024. Here are the survey responses and marketing implications that stood out to us.
Emotional and Behavioural Problems in Children - Counselling and Family Thera...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
05 CLINICAL AUDIT-ORTHO done at a peripheral.pptx

Clinical audit on pain management done at peripheral health centre
一比一原版(UoA毕业证)昆士兰科技大学毕业证如何办理

UoA毕业证学历书【微信95270640】办文凭{昆士兰科技大学毕业证}Q微Q微信95270640UoA毕业证书成绩单/学历认证UoA Diploma未毕业、挂科怎么办?+QQ微信:Q微信95270640-大学Offer(申请大学)、成绩单(申请考研)、语言证书、在读证明、使馆公证、办真实留信网认证、真实大使馆认证、学历认证
办理国外昆士兰科技大学毕业证书 #成绩单改成绩 #教育部学历学位认证 #毕业证认证 #留服认证 #使馆认证(留学回国人员证明) #(证)等
真实教育部认证教育部存档中国教育部留学服务中心认证(即教育部留服认证)网站100%可查.
真实使馆认证(即留学人员回国证明)使馆存档可通过大使馆查询确认.
留信网认证国家专业人才认证中心颁发入库证书留信网永久存档可查.
昆士兰科技大学昆士兰科技大学毕业证文凭证书毕业证 #成绩单等全套材料从防伪到印刷从水印到钢印烫金跟学校原版100%相同.
国际留学归国服务中心:实体公司注册经营行业标杆精益求精!
国外毕业证学位证成绩单办理流程:
1客户提供办理昆士兰科技大学昆士兰科技大学毕业证文凭证书信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询);
2开始安排制作昆士兰科技大学毕业证成绩单电子图;
3昆士兰科技大学毕业证成绩单电子版做好以后发送给您确认;
4昆士兰科技大学毕业证成绩单电子版您确认信息无误之后安排制作成品;
5昆士兰科技大学成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快递邮寄昆士兰科技大学昆士兰科技大学毕业证文凭证书)。闷不乐的样子父亲特意带山娃去找三楼房东家的儿子小伍玩小伍比山娃小一岁虎头虎脑的很霸气父亲让山娃跟小伍去夏令营听课山娃很高兴夏令营就设在附近一所小学山娃发现那所小学比自己的学校更大更美操场上还铺有塑胶跑道呢里面很多小朋友一班一班的快快乐乐原来城里娃都藏这儿来了怪不得平时见不到他们山娃恍然大悟起来吹拉弹唱琴棋书画山娃都不懂却什么都想学山娃怨自己太笨什么都不会斟酌再三山娃终于选定了学美术当听说每月要交感
PRESSURE INJURY CARE AND MANAGEMENT FOR HCW

Cme on pressure injury care.
Credit to : PPP Abdul Manan Bin Othman.
UNIT PERKEMBANGAN PROFESYEN PPP
BAHAGIAN PEMBANGUNAN KESIHATAN KELUARGA
English Drug and Alcohol Commissioners June 2024.pptx

Presentation made by Mat Southwell to the Harm Reduction Working Group of the English Drug and Alcohol Commissioners. Discuss stimulants, OAMT, NSP coverage and community-led approach to DCRs. Focussing on active drug user perspectives and interests
THE SPECIAL SENCES- Unlocking the Wonders of the Special Senses: Sight, Sound...

Title: Unlocking the Wonders of the Special Senses: Sight, Sound, Smell, Taste, and Balance
Introduction:
Welcome to our captivating SlideShare presentation on the Special Senses, where we delve into the extraordinary capabilities that allow us to perceive and interact with the world around us. Join us on a sensory journey as we explore the intricate structures and functions of sight, sound, smell, taste, and balance.
The special senses are our primary means of experiencing and interpreting the environment, each sense providing unique and vital information that shapes our perceptions and responses. These senses are facilitated by highly specialized organs and complex neural pathways, enabling us to see a vibrant sunset, hear a symphony, savor a delicious meal, detect a fragrant flower, and maintain our equilibrium.
In this presentation, we will:
Visual System (Sight): Dive into the anatomy and physiology of the eye, exploring how light is converted into electrical signals and processed by the brain to create the images we see. Understand common vision disorders and the mechanisms behind corrective measures like glasses and contact lenses.
Auditory System (Hearing): Examine the structures of the ear and the process of sound wave transduction, from the outer ear to the cochlea and auditory nerve. Learn about hearing loss, auditory processing, and the advances in hearing aid technology.
Olfactory System (Smell): Discover the olfactory receptors and pathways that enable the detection of thousands of different odors. Explore the connection between smell and memory and the impact of olfactory disorders on quality of life.
Gustatory System (Taste): Uncover the taste buds and the five basic tastes – sweet, salty, sour, bitter, and umami. Delve into the interplay between taste and smell and the factors influencing our food preferences and eating habits.
Vestibular System (Balance): Investigate the inner ear structures responsible for balance and spatial orientation. Understand how the vestibular system helps maintain posture and coordination, and explore common vestibular disorders and their effects.
Through engaging visuals, interactive diagrams, and insightful explanations, we aim to illuminate the complexities of the special senses and their profound impact on our daily lives. Whether you're a student, educator, or simply curious about how we perceive the world, this presentation will provide valuable insights into the remarkable capabilities of the human sensory system.
Join us as we unlock the wonders of the special senses and gain a deeper appreciation for the intricate mechanisms that allow us to experience the richness of our environment.
Test bank clinical nursing skills a concept based approach 4e pearson educati...

Test bank clinical nursing skills a concept based approach 4e pearson educati...rightmanforbloodline
Test bank clinical nursing skills a concept based approach 4e pearson education
Test bank clinical nursing skills a concept based approach 4e pearson education
Test bank clinical nursing skills a concept based approach 4e pearson educationTop 5 Benefits of Cancer Registry Services

Ensure the highest quality care for your patients with Cardiac Registry Support's cancer registry services. We support accreditation efforts and quality improvement initiatives, allowing you to benchmark performance and demonstrate adherence to best practices. Confidence starts with data. Partner with Cardiac Registry Support. For more details visit https://cardiacregistrysupport.com/cancer-registry-services/
Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot

Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot
Columbia毕业证书退学办理

哥伦比亚大学毕业证成绩单(留信网认证)【176555708微信】24小时办理学位证书编号怎么查
Columbia文凭毕业证学位证(176555708微信)加急制作哥伦比亚大学毕业证认证成绩单密封邮寄|Columbia研究生毕业证书学位证书|哥伦比亚大学Office Transcript美国I20修改|Columbia Office Transcript diploma 学历认证
Columbia毕业证【微信176555708】哥伦比亚大学毕业证书原版↑制作哥伦比亚大学学历认证文凭办理哥伦比亚大学留信网认证,留学回国办理毕业证成绩单文凭学历认证【微信176555708】专业为海外学子办理毕业证成绩单、文凭制作,学历仿制,回国人员证明、做文凭,研究生、本科、硕士学历认证、留信认证、结业证、学位证书样本、美国教育部认证百分百真实存档可查】
Fit to Fly PCR Covid Testing at our Clinic Near You

A Fit-to-Fly PCR Test is a crucial service for travelers needing to meet the entry requirements of various countries or airlines. This test involves a polymerase chain reaction (PCR) test for COVID-19, which is considered the gold standard for detecting active infections. At our travel clinic in Leeds, we offer fast and reliable Fit to Fly PCR testing, providing you with an official certificate verifying your negative COVID-19 status. Our process is designed for convenience and accuracy, with quick turnaround times to ensure you receive your results and certificate in time for your departure. Trust our professional and experienced medical team to help you travel safely and compliantly, giving you peace of mind for your journey.www.nxhealthcare.co.uk
NURSING MANAGEMENT OF PATIENT WITH EMPHYSEMA .PPT

Prepared by Prof. BLESSY THOMAS, VICE PRINCIPAL, FNCON, SPN.
Emphysema is a disease condition of respiratory system.
Emphysema is an abnormal permanent enlargement of the air spaces distal to terminal bronchioles, accompanied by destruction of their walls and without obvious fibrosis.
Emphysema of lung is defined as hyper inflation of the lung ais spaces due to obstruction of non respiratory bronchioles as due to loss of elasticity of alveoli.
It is a type of chronic obstructive
pulmonary disease.
It is a progressive disease of lungs.
More Related Content
Viewers also liked
Ukraine: What information is mandatory on medical device label? 

The document outlines the mandatory information that must be included on medical device labels for products registered in Ukraine. This includes: 1) the name of the item in Ukrainian, 2) the manufacturer name and address in original language and country of origin in Ukrainian, 3) the registration certificate number and date. Optional information that can be included are main properties, sterility, guarantees, safety warnings, and manufacturer's Ukrainian representative. The label must be in Ukrainian and can include symbols for standards compliance.
Localization 2014

The Russian Ministry of Industry and Trade reconsidered restrictions on the supply of medical devices for state procurement in July 2014. The restrictions affect state and municipal procurements of medical devices from a closed list. If at least two bids with local products from Russia, Belarus or Kazakhstan are submitted for tender, buyers will be unable to procure medical devices originating from other countries. The local products must be confirmed by a certificate of origin issued by a local authorized agency. Certain categories of medical devices are subject to the restrictions, including syringes, osteosynthesis implants, ECG devices, and surgical instruments.
How Can You Group Medical Devices for Registration in Russia? 

Slide explains the principles of product grouping for submission for registration of medical device in RosZdravNadzor (Russia).
How to provide proper photos for medical device registration dossier? 

The document provides guidelines for submitting photo documentation of medical devices for registration purposes, requiring at least one high resolution photo sized 18x24cm showing the device and all accessories, with each item clearly identified either through graphic labeling or direct labeling on the photo. For similar devices of different sizes, one photo is acceptable, and for dossiers covering multiple different devices, one photo per type is sufficient along with a product catalog.
Let us put some clarity on VAT topic

This document outlines the value-added tax (VAT) rates for medical devices in Russia both past and present. It states that historically, medical equipment was subject to a 0% VAT rate, medical products had an 18% VAT rate, and the exact lists of qualifying items were defined in government orders from 2002 and 2008. As of January 1, 2013, a new law unified these terms as "medical devices" and introduced some VAT rate changes, making the VAT rules less clear. The document concludes that additional low-level legislative changes on medical device VAT rates in Russia may still be forthcoming.
Russia: What information is mandatory on medical device label? 

This document summarizes the mandatory information that must be included on medical device labels in Russia. The label must include:
1. The name of the medical device in Russian.
2. Information about the manufacturer including the word "manufacturer" in Russian, the name and address of the manufacturer in the original language, and the country of origin in Russian.
3. The registration number and date of registration for the device in Russia.
4. Information about the device's properties in Russian such as size, sterility, single-use instructions, storage conditions, and shelf life.
Viewers also liked (6)
Ukraine: What information is mandatory on medical device label? 

Ukraine: What information is mandatory on medical device label?
How Can You Group Medical Devices for Registration in Russia? 

How Can You Group Medical Devices for Registration in Russia?
How to provide proper photos for medical device registration dossier? 

How to provide proper photos for medical device registration dossier?
Russia: What information is mandatory on medical device label? 

Russia: What information is mandatory on medical device label?
Recently uploaded
Simple Steps to Make Her Choose You Every Day

Simple Steps to Make Her Choose You Every Day" and unlock the secrets to building a strong, lasting relationship. This comprehensive guide takes you on a journey to self-improvement, enhancing your communication and emotional skills, ensuring that your partner chooses you without hesitation. Forget about complications and start applying easy, straightforward steps that make her see you as the ideal person she can't live without. Gain the key to her heart and enjoy a relationship filled with love and mutual respect. This isn't just a book; it's an investment in your happiness and the happiness of your partner
Sexual Disorders.gender identity disorderspptx

Gender identity disorder, paraphilias , sexual dysfunction
2024 Media Preferences of Older Adults: Consumer Survey and Marketing Implica...

When it comes to creating marketing strategies that target older adults, it is crucial to have insight into their media habits and preferences. Understanding how older adults consume and use media is key to creating acquisition and retention strategies. We recently conducted our seventh annual survey to gain insight into the media preferences of older adults in 2024. Here are the survey responses and marketing implications that stood out to us.
Emotional and Behavioural Problems in Children - Counselling and Family Thera...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
05 CLINICAL AUDIT-ORTHO done at a peripheral.pptx

Clinical audit on pain management done at peripheral health centre
一比一原版(UoA毕业证)昆士兰科技大学毕业证如何办理

UoA毕业证学历书【微信95270640】办文凭{昆士兰科技大学毕业证}Q微Q微信95270640UoA毕业证书成绩单/学历认证UoA Diploma未毕业、挂科怎么办?+QQ微信:Q微信95270640-大学Offer(申请大学)、成绩单(申请考研)、语言证书、在读证明、使馆公证、办真实留信网认证、真实大使馆认证、学历认证
办理国外昆士兰科技大学毕业证书 #成绩单改成绩 #教育部学历学位认证 #毕业证认证 #留服认证 #使馆认证(留学回国人员证明) #(证)等
真实教育部认证教育部存档中国教育部留学服务中心认证(即教育部留服认证)网站100%可查.
真实使馆认证(即留学人员回国证明)使馆存档可通过大使馆查询确认.
留信网认证国家专业人才认证中心颁发入库证书留信网永久存档可查.
昆士兰科技大学昆士兰科技大学毕业证文凭证书毕业证 #成绩单等全套材料从防伪到印刷从水印到钢印烫金跟学校原版100%相同.
国际留学归国服务中心:实体公司注册经营行业标杆精益求精!
国外毕业证学位证成绩单办理流程:
1客户提供办理昆士兰科技大学昆士兰科技大学毕业证文凭证书信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询);
2开始安排制作昆士兰科技大学毕业证成绩单电子图;
3昆士兰科技大学毕业证成绩单电子版做好以后发送给您确认;
4昆士兰科技大学毕业证成绩单电子版您确认信息无误之后安排制作成品;
5昆士兰科技大学成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快递邮寄昆士兰科技大学昆士兰科技大学毕业证文凭证书)。闷不乐的样子父亲特意带山娃去找三楼房东家的儿子小伍玩小伍比山娃小一岁虎头虎脑的很霸气父亲让山娃跟小伍去夏令营听课山娃很高兴夏令营就设在附近一所小学山娃发现那所小学比自己的学校更大更美操场上还铺有塑胶跑道呢里面很多小朋友一班一班的快快乐乐原来城里娃都藏这儿来了怪不得平时见不到他们山娃恍然大悟起来吹拉弹唱琴棋书画山娃都不懂却什么都想学山娃怨自己太笨什么都不会斟酌再三山娃终于选定了学美术当听说每月要交感
PRESSURE INJURY CARE AND MANAGEMENT FOR HCW

Cme on pressure injury care.
Credit to : PPP Abdul Manan Bin Othman.
UNIT PERKEMBANGAN PROFESYEN PPP
BAHAGIAN PEMBANGUNAN KESIHATAN KELUARGA
English Drug and Alcohol Commissioners June 2024.pptx

Presentation made by Mat Southwell to the Harm Reduction Working Group of the English Drug and Alcohol Commissioners. Discuss stimulants, OAMT, NSP coverage and community-led approach to DCRs. Focussing on active drug user perspectives and interests
THE SPECIAL SENCES- Unlocking the Wonders of the Special Senses: Sight, Sound...

Title: Unlocking the Wonders of the Special Senses: Sight, Sound, Smell, Taste, and Balance
Introduction:
Welcome to our captivating SlideShare presentation on the Special Senses, where we delve into the extraordinary capabilities that allow us to perceive and interact with the world around us. Join us on a sensory journey as we explore the intricate structures and functions of sight, sound, smell, taste, and balance.
The special senses are our primary means of experiencing and interpreting the environment, each sense providing unique and vital information that shapes our perceptions and responses. These senses are facilitated by highly specialized organs and complex neural pathways, enabling us to see a vibrant sunset, hear a symphony, savor a delicious meal, detect a fragrant flower, and maintain our equilibrium.
In this presentation, we will:
Visual System (Sight): Dive into the anatomy and physiology of the eye, exploring how light is converted into electrical signals and processed by the brain to create the images we see. Understand common vision disorders and the mechanisms behind corrective measures like glasses and contact lenses.
Auditory System (Hearing): Examine the structures of the ear and the process of sound wave transduction, from the outer ear to the cochlea and auditory nerve. Learn about hearing loss, auditory processing, and the advances in hearing aid technology.
Olfactory System (Smell): Discover the olfactory receptors and pathways that enable the detection of thousands of different odors. Explore the connection between smell and memory and the impact of olfactory disorders on quality of life.
Gustatory System (Taste): Uncover the taste buds and the five basic tastes – sweet, salty, sour, bitter, and umami. Delve into the interplay between taste and smell and the factors influencing our food preferences and eating habits.
Vestibular System (Balance): Investigate the inner ear structures responsible for balance and spatial orientation. Understand how the vestibular system helps maintain posture and coordination, and explore common vestibular disorders and their effects.
Through engaging visuals, interactive diagrams, and insightful explanations, we aim to illuminate the complexities of the special senses and their profound impact on our daily lives. Whether you're a student, educator, or simply curious about how we perceive the world, this presentation will provide valuable insights into the remarkable capabilities of the human sensory system.
Join us as we unlock the wonders of the special senses and gain a deeper appreciation for the intricate mechanisms that allow us to experience the richness of our environment.
Test bank clinical nursing skills a concept based approach 4e pearson educati...

Test bank clinical nursing skills a concept based approach 4e pearson educati...rightmanforbloodline
Test bank clinical nursing skills a concept based approach 4e pearson education
Test bank clinical nursing skills a concept based approach 4e pearson education
Test bank clinical nursing skills a concept based approach 4e pearson educationTop 5 Benefits of Cancer Registry Services

Ensure the highest quality care for your patients with Cardiac Registry Support's cancer registry services. We support accreditation efforts and quality improvement initiatives, allowing you to benchmark performance and demonstrate adherence to best practices. Confidence starts with data. Partner with Cardiac Registry Support. For more details visit https://cardiacregistrysupport.com/cancer-registry-services/
Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot

Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot
Columbia毕业证书退学办理

哥伦比亚大学毕业证成绩单(留信网认证)【176555708微信】24小时办理学位证书编号怎么查
Columbia文凭毕业证学位证(176555708微信)加急制作哥伦比亚大学毕业证认证成绩单密封邮寄|Columbia研究生毕业证书学位证书|哥伦比亚大学Office Transcript美国I20修改|Columbia Office Transcript diploma 学历认证
Columbia毕业证【微信176555708】哥伦比亚大学毕业证书原版↑制作哥伦比亚大学学历认证文凭办理哥伦比亚大学留信网认证,留学回国办理毕业证成绩单文凭学历认证【微信176555708】专业为海外学子办理毕业证成绩单、文凭制作,学历仿制,回国人员证明、做文凭,研究生、本科、硕士学历认证、留信认证、结业证、学位证书样本、美国教育部认证百分百真实存档可查】
Fit to Fly PCR Covid Testing at our Clinic Near You

A Fit-to-Fly PCR Test is a crucial service for travelers needing to meet the entry requirements of various countries or airlines. This test involves a polymerase chain reaction (PCR) test for COVID-19, which is considered the gold standard for detecting active infections. At our travel clinic in Leeds, we offer fast and reliable Fit to Fly PCR testing, providing you with an official certificate verifying your negative COVID-19 status. Our process is designed for convenience and accuracy, with quick turnaround times to ensure you receive your results and certificate in time for your departure. Trust our professional and experienced medical team to help you travel safely and compliantly, giving you peace of mind for your journey.www.nxhealthcare.co.uk
NURSING MANAGEMENT OF PATIENT WITH EMPHYSEMA .PPT

Prepared by Prof. BLESSY THOMAS, VICE PRINCIPAL, FNCON, SPN.
Emphysema is a disease condition of respiratory system.
Emphysema is an abnormal permanent enlargement of the air spaces distal to terminal bronchioles, accompanied by destruction of their walls and without obvious fibrosis.
Emphysema of lung is defined as hyper inflation of the lung ais spaces due to obstruction of non respiratory bronchioles as due to loss of elasticity of alveoli.
It is a type of chronic obstructive
pulmonary disease.
It is a progressive disease of lungs.
Friendly Massage in Ajman - Malayali Kerala Spa Ajman

At Malayali Kerala Spa Ajman, Full Service includes individualized care for every client. We specifically design each massage session for the individual needs of the client. Our therapists are always willing to adjust the treatments based on the client's instruction and feedback. This guarantees that every client receives the treatment they expect.
By offering a variety of massage services, our Ajman Spa Massage Center can tackle physical, mental, and emotional illnesses. In addition, efficient identification of specific health conditions and designing treatment plans accordingly can significantly enhance the quality of massaging.
At Malayali Kerala Spa Ajman, we firmly believe that everyone should have the option to experience top-quality massage services regularly. To achieve that goal we offer cheap massage services in Ajman.
If you are interested in experiencing transformative massage treatment at Malayali Kerala Spa Ajman, you can use our Ajman Massage Center WhatsApp Number to schedule your next massage session.
Contact @ +971 529818279
Visit @ https://malayalikeralaspaajman.com/
Luxury Massage Experience at Affordable Rate - Malayali Kerala Spa Ajman

At Malayali Kerala Spa Ajman we providing the top quality massage services for our customers.
Our massage center prioritizes efficiency to ensure a quality massage experience for our clients at Malayali Kerala Spa Ajman. We offer a convenient appointment system and precise massage services.
Reach us at Villa No 7, Near Ammar Bin Yasir Street Al Rashidiya 2 - Ajman - United Arab Emirates.
Phone : +971 529818279
Satisfying Spa Massage Experience at Just 99 AED - Malayali Kerala Spa Ajman

Satisfying Spa Massage Experience at Just 99 AED - Malayali Kerala Spa AjmanMalayali Kerala Spa Ajman
Our Spa Massage Center Ajman prioritizes efficiency to ensure a satisfying massage experience for our clients at Malayali Kerala Spa Ajman. We offer a hassle-free appointment system, effective health issue identification, and precise massage techniques.
Our Spa in Ajman stands out for its effectiveness in enhancing wellness. Our therapists focus on treating the root cause of issues, providing tailored treatments for each client. We take pride in offering the most satisfying Pakistani Spa service, adjusting treatment plans based on client feedback.
For the most result-oriented Russian Spa treatment in Ajman, visit our Massage Center. Our Russian therapists are skilled in various techniques to address health concerns. Our body-to-body massage is efficient due to individualized care and high-grade massage oils.Recently uploaded (20)
HEALTH ASSESSMENT IN NURSING USING THE NURSING PROCESSpptx

HEALTH ASSESSMENT IN NURSING USING THE NURSING PROCESSpptx
2024 Media Preferences of Older Adults: Consumer Survey and Marketing Implica...

2024 Media Preferences of Older Adults: Consumer Survey and Marketing Implica...
Emotional and Behavioural Problems in Children - Counselling and Family Thera...

Emotional and Behavioural Problems in Children - Counselling and Family Thera...
English Drug and Alcohol Commissioners June 2024.pptx

English Drug and Alcohol Commissioners June 2024.pptx
THE SPECIAL SENCES- Unlocking the Wonders of the Special Senses: Sight, Sound...

THE SPECIAL SENCES- Unlocking the Wonders of the Special Senses: Sight, Sound...
Test bank clinical nursing skills a concept based approach 4e pearson educati...

Test bank clinical nursing skills a concept based approach 4e pearson educati...
Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot

Faridkot ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 7742996321 ℂ𝕒𝕝𝕝 𝔾𝕚𝕣𝕝𝕤 Faridkot
Fit to Fly PCR Covid Testing at our Clinic Near You

Fit to Fly PCR Covid Testing at our Clinic Near You
Friendly Massage in Ajman - Malayali Kerala Spa Ajman

Friendly Massage in Ajman - Malayali Kerala Spa Ajman
3. User Guide Activity Budget Tracking App Steps to apply.pptx

3. User Guide Activity Budget Tracking App Steps to apply.pptx
Luxury Massage Experience at Affordable Rate - Malayali Kerala Spa Ajman

Luxury Massage Experience at Affordable Rate - Malayali Kerala Spa Ajman
Satisfying Spa Massage Experience at Just 99 AED - Malayali Kerala Spa Ajman

Satisfying Spa Massage Experience at Just 99 AED - Malayali Kerala Spa Ajman
Changes and Modifications
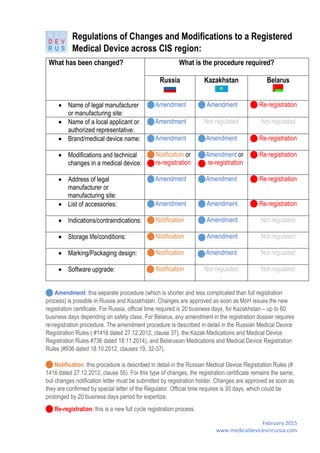
- 1. February 2015 www.medicaldevicesinrussia.com Regulations of Changes and Modifications to a Registered Medical Device across CIS region: Amendment: this separate procedure (which is shorter and less complicated than full registration process) is possible in Russia and Kazakhstan. Changes are approved as soon as MoH issues the new registration certificate. For Russia, official time required is 20 business days, for Kazakhstan – up to 60 business days depending on safety class. For Belarus, any amendment in the registration dossier requires re-registration procedure. The amendment procedure is described in detail in the Russian Medical Device Registration Rules ( #1416 dated 27.12.2012, clause 37), the Kazak Medications and Medical Device Registration Rules #736 dated 18.11.2014), and Belarusian Medications and Medical Device Registration Rules (#936 dated 18.10.2012, clauses 19, 32-37). Notification: this procedure is described in detail in the Russian Medical Device Registration Rules (# 1416 dated 27.12.2012, clause 55). For this type of changes, the registration certificate remains the same, but changes notification letter must be submitted by registration holder. Changes are approved as soon as they are confirmed by special letter of the Regulator. Official time requires is 30 days, which could be prolonged by 20 business days period for expertize. Re-registration: this is a new full cycle registration process. What has been changed? What is the procedure required? Russia Kazakhstan Belarus Name of legal manufacturer or manufacturing site: Amendment Amendment Re-registration Name of a local applicant or authorized representative: Amendment Not regulated Not regulated Brand/medical device name: Amendment Amendment Re-registration Modifications and technical changes in a medical device: Notification or re-registration Amendment or re-registration Re-registration Address of legal manufacturer or manufacturing site: Amendment Amendment Re-registration List of accessories: Amendment Amendment Re-registration Indications/contraindications: Notification Amendment Not regulated Storage life/conditions: Notification Amendment Not regulated Marking/Packaging design: Notification Amendment Not regulated Software upgrade: Notification Not regulated Not regulated
