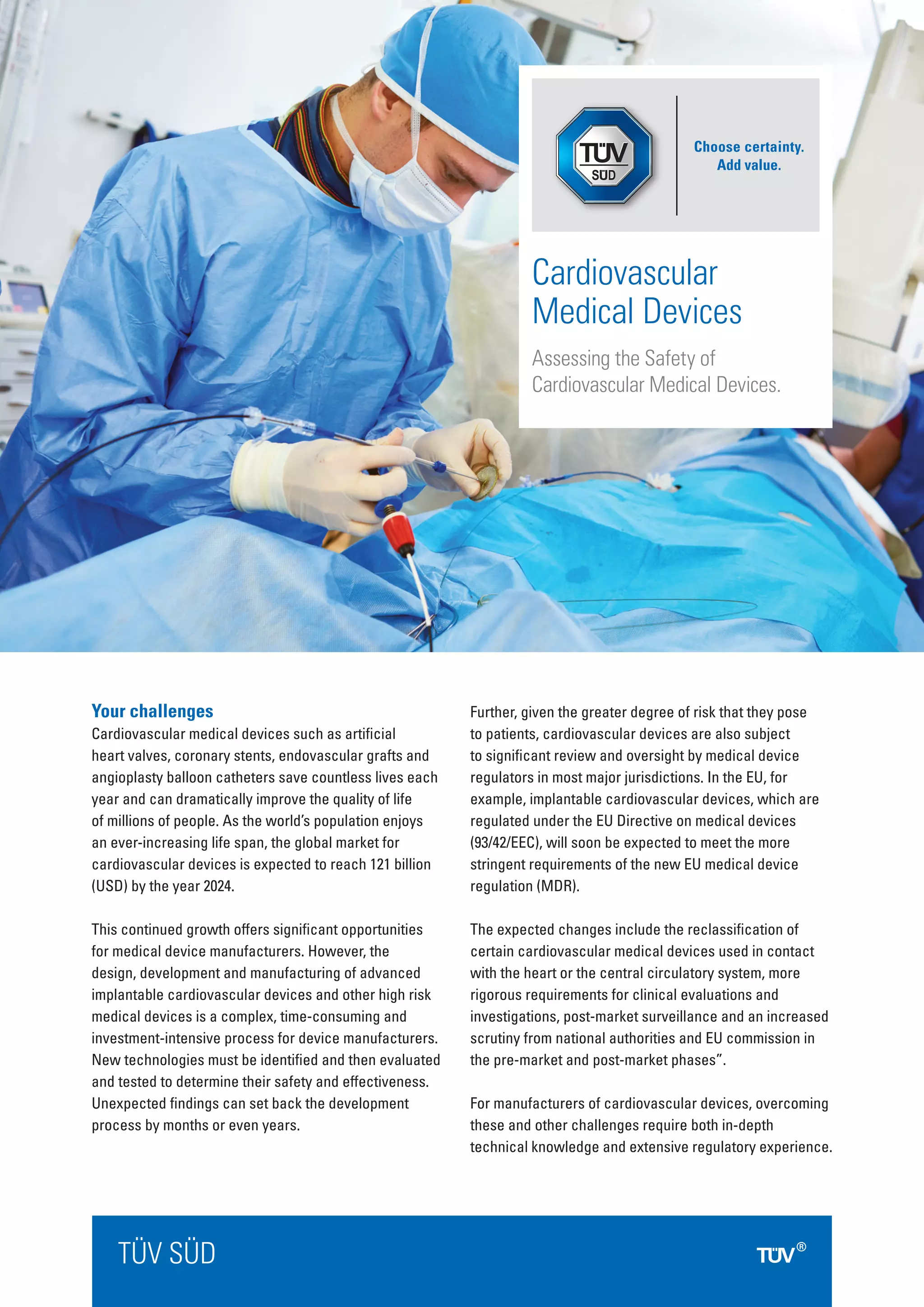
This document discusses the challenges facing manufacturers of cardiovascular medical devices, including the complex design and testing processes required, stringent regulatory oversight, and changing regulatory requirements. It summarizes the services offered by TUV SUD to help manufacturers address these challenges, including technical documentation review, quality auditing, testing, clinical services, and obtaining necessary market approvals. TUV SUD has extensive expertise in cardiovascular devices and regulatory requirements globally to support manufacturers through the compliance process.