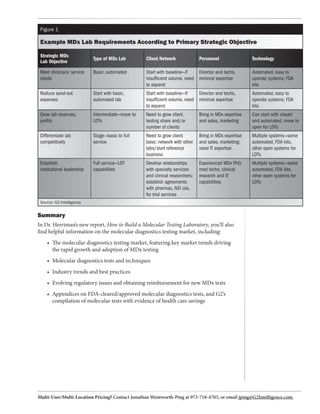
This document provides a guide for developing or expanding a molecular diagnostic testing laboratory. It includes strategic and operational considerations for basic or full-service molecular diagnostic (MDx) labs. The guide covers developing an MDx strategy, selecting instrument platforms, facility planning, personnel requirements, financial planning, quality systems, and regulatory compliance. It also provides an overview of the large and growing MDx testing market and outlines industry best practices based on a survey of 90 MDx laboratories.