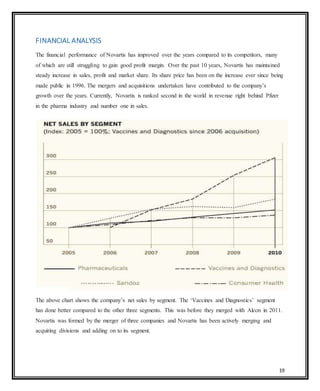
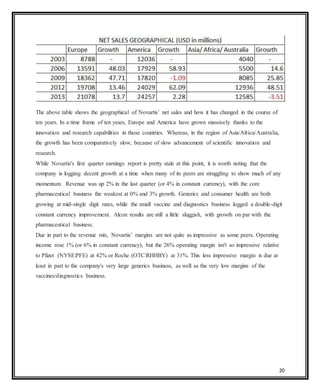
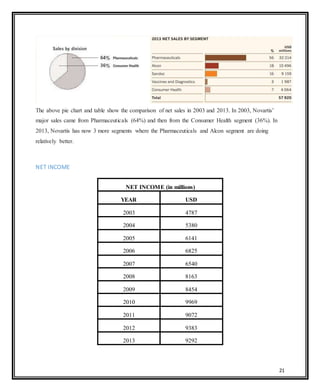
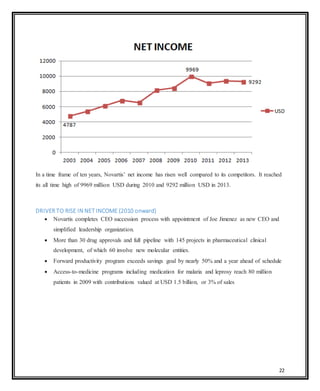
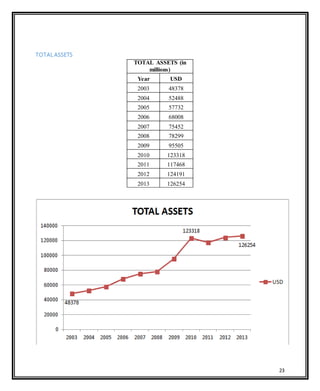
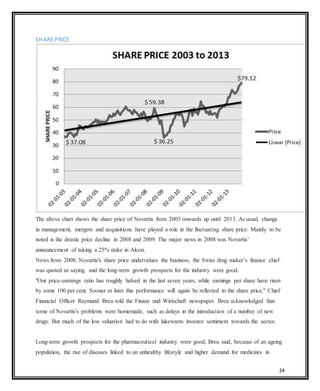
Novartis is a large pharmaceutical company headquartered in Switzerland that was formed in 1996 through the merger of Ciba-Geigy and Sandoz. Over the years, Novartis has expanded through acquisitions of companies like Alcon, Hexal, and Fougera to become a leader in pharmaceuticals, eye care, generics, and vaccines. The document provides details on Novartis' products, history, acquisitions, organizational structure, marketing issues, financial performance, and strategy for further growth.