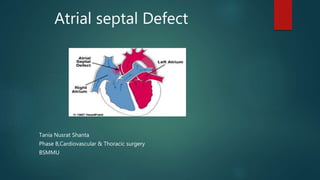
Atrial septal defect .by shanta
- 1. Atrial septal Defect Tania Nusrat Shanta Phase B,Cardiovascular & Thoracic surgery BSMMU
- 2. OBJECTIVES Introduction Historical consideration Incidence Etiology Embryology Types of ASD Natural History Haemodynamics Clinical Manifestation Evaluation and Management
- 3. Introduction Atrial septal defect (ASD) is a form of congenital heart defect that enables blood flow between the left and right atria via the interatrial septum. Severity depends on : - size of defect - size of shunt - associated anomalies Resulting in spectrum from : asymptomatic to right sided overload, PAH , even atrial arrhythmias.
- 4. Historical consideration Roesler in 1934, analyzed ASD among the 62 recorded autopsy cases only one had been correctly diagnosed during life. By 1941, Bedford and colleagues were able to make the diagnosis clinically in a number of patients. In 1948 in Toronto, Murray reported closing an ASD in a child by external suturing. Lewis and Taufic reported the first successful open repair of an ASD in 1953. Gibbon started the era of open heart surgery in 1953 when he successfully repaired an ASD in a young woman using a pump-oxygenator. By the late 1960s almost all surgeons used CPB exclusively for these repairs. Percutaneous catheter techniques for closing a fossa ovalis ASD using a polyester double umbrella device were introduced by King and Mills in 1974. It is not certain who first repaired the sinus venosus syndrome, but the malformation was clearly illustrated by Bedford and colleagues in 1957. Repair of PAPVC to the inferior vena cava was performed by Kirklin and colleagues at Mayo Clinic in 1960 and was also subsequently reported by Zubiate and Kay in 1962.
- 5. INCIDENCE Atrial septal defect occurs as an isolated anomaly in 8% to 10% of all congenital heart defects (CHDs). Incidence = 56 per 100,000 live births. Recent estimates are much higher (100 per 100,000 live births), likely due to increased recognition in the era of common use of echocardiography. Male:Female = 1:2, For sinus venosus ASD= 1:1 About 30% to 50% of children with CHDs have an ASD as part of the cardiac defect. Excluding bicuspid aortic valve and mitral valve prolapse, ASD is the most common form of congenital heart defect found among adults and is the most common acyanotic shunt lesion in adults as well.
- 6. ETIOLOGY Actual etiology of this congenital defect is unknown. Some factors may play role as there are some evidences of being association with ASD. Factors include: - Genetic factor - Environmental factor including antenatal use of teratogenic drugs, congenital infection. Genetic factor: - A familial predisposition to ASD is well documented. -Numerous genetic conditions and syndromes known for their extracardiac manifestations are also associated with ASD. -ASD is the most common congenital cardiac defect associated with VACTERL. -Genetic syndromes with associated ASD include Holt-Oram syndrome. - Trisomy 21 is associated with ASD. -Noonan syndrome is associated with ASD and pulmonary valve stenosis. -DiGeorge syndrome (22q11.4 deletion) and Ellis–Van Creveld syndromes are associated with primum ASD.
- 7. EMBRYOLOGY The primitive atrium is first partitioned into right and left atria by growth of the septum primum—a thin,crescent- shaped membrane that grows from the roof of the primitive atrium toward the endocardial cushions. Foramen primum, composed of the free edge of the septum primum and the endocardial cushions.
- 8. Fenestrations develop in the septum primum that coalesce to form the ostium secundum. As the septum primum then fuses with the endocardial cushions, the ostium secundum maintains a right-to- left atrial flow that is important in the fetal circulation. Failure of this fusion results in the development of a primum ASD.
- 9. A second septum, the septum secundum, then forms to the right of the septum primum, growing toward the endocardial cushions and usually closing the ostium secundum. Failure to close the ostium secundum results in the formation of a secundum ASD.
- 10. ANATOMY When viewed from its right aspect, the atrial septum is composed of interatrial and atrioventricular regions. The interatrial portion is characterized by the fossa ovalis, which is the anatomic hallmark of a morphologic right atrium. When viewed from the left atrium, the atrial septum is entirely interatrial because the atrioventricular component lies below the mitral annuls between the left ventricle and right atrium.
- 11. Anatomic Types Ostium Secundum defect Posterior defect Ostium Primum defect Sinus Venosus defect Coronary Sinus septal defect Confluent defect
- 12. Ostium secundum defects or secundumASDs The most common type, 70% to 75% of ASDs. Location: in the midportion of the atrial septum, within or including the fossa ovalis. Defects result from a deficient septum primum or an abnormally large foramen secundum. Two times more common in female patients. Association: -Mitral valve prolapse and other forms of congenital heart disease. -It may also be associated with rheumatic mitral stenosis (i.e.Lutembacher syndrome).
- 13. Ostium primum defects or primumASDs 15% to 20% of ASDs and are part of the spectrum of atrioventricular (AV) septal defects (also known as AV canal defects or endocardial cushion defects). Location: These defects occur in the inferior–anterior portion of the atrial septum. Association: Cleft in the anterior leaflet of the mitral valve, leading to varying degrees of mitral regurgitation. In their complete form, they include a large ventricular septal defect and a common AV valve. Most common ASD type associated with Down’s syndrome.
- 14. Sinus venosus defects Constitute the remaining 5% to 10% of septal defects. These lesions involve the portion of the atrial wall derived from the sinus venosus Location: Sinus venosus defects are typically at the orifice of the superior vena cava (SVC) at the junction of the right atrium or, less frequently, in the region of the inferior vena cava (IVC). Association: with partial anomalous pulmonary venous drainage of the right pulmonary veins
- 17. ACCORDING TO SIZE: In younger children – In older children Small defect: <3 mm Small defect: <6 mm Moderate defect: 3-8mm Moderate defect:6 – 12mm Large defect: >8 mm Large defect: >12 mm
- 18. Natural History In patients with an ASD smaller than 3 mm in size diagnosed before 3 months of age, spontaneous closure occurs in 100% of patients at 1½ years of age. Spontaneous closure occurs more than 80% of the time in patients with defects between 3 and 8 mm before 1½ years of age. An ASD with a diameter larger than 8 mm rarely closes spontaneously. Spontaneous closure is not likely to occur after 4 years of age Most children with an ASD remain active and asymptomatic. Rarely, congestive heart failure (CHF) can develop in infancy.
- 19. If a large defect is untreated, CHF and pulmonary hypertension begin to develop in adults who are in their 20s and 30s, and it becomes common after 40 years of age. With or without surgery, atrial arrhythmias (flutter or fibrillation) may occur in adults. The incidence of atrial arrhythmias increases to as high as 13% in patients older than 40 years of age. Infective endocarditis does not occur in patients with isolated ASDs. Cerebrovascular accident, resulting from paradoxical embolization through an ASD, is a rare complication.
- 20. HEMODYNAMICS LEFT-TO-RIGHT SHUNT In early infancy, when pulmonary resistance is high, left and right ventricular compliances are similar, and net shunting through an ASD is typically slight. As the left ventricle matures, it becomes less compliant in diastole than the right, and left atrial pressure rises. This drives a left-to-right shunt at the atrial level in the presence of an ASD. With age, the disparity between systemic and pulmonary resistance, and in turn between left and right ventricular compliance, results in increased left-to-right shunting and advancing right ventricular volume loading. Over time, right ventricular volume load results in dilation and hypertrophy, eventually affecting the function of both ventricles. Atrial enlargement may contribute to the late incidence of atrial fibrillation. Right ventricular volume overload is noted to occur as a rule when ASDs are larger than 6 mm in diameter.
- 21. HEMODYNAMICS
- 22. Volume-induced hypertrophy of the right ventricle produces a loss of coronary reserve and eventual impairment of right ventricular systolic and diastolic function. Left ventricular functional reserve is diminished by adulthood in most patients with ASD. Although left ventricular systolic function may be normal at rest, the left ventricle exhibits a subnormal diastolic dimension, and a loss of functional reserve at exercise. Mechanisms that account for left ventricular dysfunction include (1) septal displacement secondary to right ventricular dilation and hypertrophy and (2) systolic anterior movement of the mitral valve. In general, the functional loss in the left and right ventricles is normalized 6 months following ASD closure in children and young adults.
- 23. CARDIAC CHAMBERS IN ATRIAL SEPTAL DEFECT Typically in ASD and related conditions, the RIGHT ATRIUM is greatly enlarged (at least grade 3 or 4 on a scale of 1 to 6) and thick walled. The LEFT ATRIUM is not enlarged. RIGHT VENTRICULAR (RV) diastolic size is increased, often greatly, because of volume overload imposed by the left-to- right shunt. Whereas normal RV diastolic dimensions are between 0.6 and 1.4 cm · m2, in patients with large left-to- right shunts at atrial level they average 2.66 cm · m2 and may be as large as 4 cm · m2. Morphologically, the LEFT VENTRICLE (LV) is normal or slightly decreased in size.However, important LV dynamic abnormalities are present in most patients.
- 24. Mitral Prolapse Mitral valve prolapse occurs in association with fossa ovalis ASD, sinus venosus and probably other types of ASDs and related conditions that result in left-to- right shunts at the atrial level. Prevalence of true prolapse is about 20% . Mitral Regurgitation Mitral prolapse in ASD can lead to mitral regurgitation, as does ordinary mitral prolapse. True prevalence of regurgitation in unselected patients varies because older patients and those with larger pulmonary blood flows have a higher prevalence of this abnormality and prolapse.
- 26. SYMPTOMS AND SIGNS Vary with the size of defect. Small defect: Asymptomatic and is usually,diagnosed during a routine health check up. Large defect: Symptomatic and patients usually present with Breathlessness Paltitation Recurrent Pulmonary infections Failure to thrive. Easy fatigability. Tachycardia Tachypnea Paroxysmal atrial tachycardia Atrial fibrillation Older patient with CHF : elevated JVP ,edema, hepatomegaly,cardiac cachexia. Patient with severe PHTN: may present with cyanosis ,hemoptysis. Platypnea Orthodeoxia
- 27. General examination Appearance: Usually normal Heart rate: Normal Respiratory rate: Normal Weight and height: may be less than 10th centile. Precordium Inspection: Normal or Slight prominence of precordium Palpation: Apex beat may be shifted to left P2 may be palpable Left parasternal heave may be present
- 28. Auscultation: S1 is normal S2 is widely splitted and fixed Ejection systolic murmur,medium pitched, soft, grade 1-3/6 & best heard at left 2nd & 3rd ICS. A mid-diastolic tricuspid flow murmur (in fourth and fifth left intercostal spaces) present in borderline situations only on inspiration.
- 29. INVESTIGATIONS Diagnostic Investigations includes -X-ray -ECG -Echocardiography - cardiac catheterization -Cardiac MRI
- 30. Xray Findings Cardiomegaly RA enlargement RV enlargement Full pulmonary conus Increased pulmonary vascular markings Plethoric lung fields
- 31. ECG Secundum ASD: RSR’ pattern in lead V1 QRS duration < 0.11 seconds (incomplete right bundle branch block) Right-axis deviation RV hypertrophy First-degree AV block (20%) RA enlargement (about 50%) with a prominent P wave in lead II Primum ASD: RSR’ pattern in lead V1 Left-axis deviation First-degree AV block, classically seen with right bundle branch block and left anterior fascicular block
- 32. ECG Enlarged ‘p’ wave indicating Right atrial hypertroph y rsR’ seen and tall R wave Indicating RBBB and RVH
- 33. Echocardiography Primary diagnostic imaging modality for ASD. Provides: - exact localization of ASD - size of ASD - measurement of septal rims - Confirmation of the shunt - Abnormal motion of ventricular septum. - Associated lesions can be identified
- 34. Echocardiography A two-dimensional echocardiographic study is diagnostic. The study shows the position as well as the size of the defect, which can best be seen in the subcostal four chamber view. A. The SVC type of sinus venosus defect shows a defect in the posterosuperior atrial septum. B. In secundum ASD, a dropout can be seen in the midatrial septum. C. The primum type shows a defect in the lower atrial septum.
- 35. Indirect signs of a significant left-to-right atrial shunt include RV enlargement and RA enlargement, as well as dilated PA, which often accompanies an increased flow velocity across the pulmonary valve. Pulsed Doppler examination reveals a characteristic flow pattern with the maximum left-to-right shunt occurring in diastole. Color-flow mapping enhances the evaluation of the hemodynamic status of the ASD.
- 36. In older children and adolescents, especially in those with overweight, adequate imaging of the atrial septum may not be possible with the ordinary transthoracic echocardiographic study. Transesophageal echocardiography (TEE) may be used as an alternative.
- 37. Cardiac catheterization Typically not required for diagnostic purposes except to assess pulmonary pressures and resistance or as part of a planned transcatheter device closure. Hemodynamic assessment: An important assessment is comparison of pulmonary artery pressure with systemic pressure and measurement of pulmonary vascular resistance. If pulmonary pressures are elevated, the response to oxygen or other vasodilators should be assessed. Alternatively, the ASD can be balloon occluded with assessment of hemodynamics to ensure that closure is safe. Cardiac MRI : Can be helpful, as it can provide additional information beyond echocardiography.
- 38. MANAGEMENT Patients with small shunts and normal RV size are generally asymptomatic and require no therapy but need longtime follow up for spontaneous closure. Moderate to large shunt and/or symptomatic ASD should be managed with following strategies: - Medical therapy - Interventional therapy - Surgical therapy
- 39. Medical management Aim to reduce volume overload and to strengthen functions of heart muscles. Symptomatic children : Diuretics: -These agents relieve ventricular overload, peripheral and pulmonary congestion Digoxin: -Helps to strengthen the heart muscle, enabling it to pump more efficiently. Afterload reducers: - Enalapril - Captopril Exercise restriction is no necessary. Prophylaxis for infective endocarditis is not indicated Atrial arrythmias : Appropriate Antiarrhythmic drugs. Atrial fibrillation : Antiarrhythmic drugs +anticoagulants. Irreversible PAH :dobutamine, calcium channel blockers (high dose), diuretics, prostacycline, sildenafil or oxygen therapy. Treatment of Other complications, like pulmonary infections, thrombo- embolic events or heart failure should also be treated accordingly.
- 40. Interventional therapy Closure of ASD : In patients with small secundum ASDs and minimal left-to-right shunts without right ventricular enlargement, closure is not required Indications of ASD closure- -All symptomatic patients -Asymptomatic patients with- • Qp : Qs ratio of at least 2 : 1 • Right ventricular enlargement Presence of sufficient rim of tissue( at least 5 mm) Time of closure- usually after the 1st yr and before entry into school
- 41. Contraindication: Sinus venosus, coronary sinus or primum ASD Extensive congenital cardiac anomaly. Known sepsis within one month prior to implantation or any untreated systemic infection prior to device placement. Bleeding disorder, untreated ulcer or any other contraindications to aspirin therapy. Demonstrated intracardiac thrombi on echo. Any patient whose size or condition would cause to be a poor candidate for cardiac catheterization.
- 42. Transcatheter device closure of secundum type ASD was first performed in 1976 by Mills and King. Different ASD closure devices: Clamshell(TM) device Buttoned device Angel wings(TM) device Atrial septal defect occluder system device In the United States, currently only the Amplatzer septal occluder and Helex septal occluder are approved by FDA for secundum ASD closure.
- 43. Complications of Device Closure: Device misalignment/embolization Device erosion of atrial wall or aorta Device impingement on adjacent structures AV valve, Coronary sinus, SVC, Pulmonary veins, Aorta Infection including endocarditis Thromboembolic Complication Allergic reaction Residual shunt
- 44. Postdevice Closure Follow-up • After closure, antiplatelet therapy, frequently aspirin and clopidogrel, is prescribed for a minimum of 6 months, after which time the device is generally believed to have endothelialized. • Postprocedure echocardiographic studies check for a residual atrial shunt and unobstructed flow of pulmonary veins, coronary sinus, and venae cavae, and proper function of the mitral and tricuspid valves. • If 1-month and 1-year follow-up echocardiography findings are normal then yearly or biennial follow-up will suffice.
- 45. Surgical management Surgical closure has been the “gold standard” form of treatment of ASD Surgeons need proper training and expertise in performing operations. The surgical approach can be by right thoracotomy or sternotomy ,partial or full submammary skin incision,hemisternotomy, and more limited incisions are feasible with either approach. Procedure- Simple suture or patch closure Timing- Surgery is usually delayed until the patient is 2 to 4 years of age because the possibility of spontaneous closure exists. In infancy- If CCF not respond to medical management.
- 47. Indication: ASD with RA and RV enlargement with / without symptoms. ASD minimum diameter > 10 mm on echocardiography A sinus venosus, coronary sinus or primum ASD Chronic atrial arrythmia with ASD Contraindication: Patients with severe irreversible PAH & reverse shunt SPO2 < 90%
- 48. Preoperative risk factors: • Older age at operation, • Presence of atrial fibrillation, and • Elevated pulmonary pressure and resistance. Mortality: < 0.5% Complications: • Cerebrovascular accident • Postoperative arrhythmias may develop in the immediate postoperative period. • Postpericardiotomy syndrome
- 49. Advantages of Surgery- Can be performed in any type of ASD Associated anatomical abnormality can be corrected concurrently. Excellent late outcome. Disadvantages of Surgery- Costly Needs expertise hands Long Hospital stay pain and residual thoracotomy scars
- 50. Postoperative Follow-up • Cardiomegaly on chest radiographs and enlarged RV dimension on echo as well as the wide splitting of the S2 may persist for 1 or 2 years after surgery. • The ECG typically demonstrates RBBB (or RV conduction disturbance). • Atrial or nodal arrhythmias occur in 7% to 20% of postoperative patients. • Occasionally, sick sinus syndrome, which occurs especially after the repair of a sinus venosus defect, may require antiarrhythmic drugs, pacemaker therapy, or both.
- 51. PROGNOSIS: Patients generally survive up to adulthood without surgical or percutaneous intervention mainly with small to moderate size ASD and many patients live to advanced age. The results after surgical or device closure in children with moderate to large shunts are excellent. Mortality is less than 2% after surgical closure of uncomplicated ASD. Mortality and morbidity increase with pulmonary vascular disease.
- 52. TAKE –HOME MESSAGES Atrial septal defects are relatively common CHD Early symptoms are usually rare except very large deffect. Any kind of closure is safe and effective and associated with improved life expectancy A comprehensive treatment plan should include input from the primary care provider, the Paediatric Cardiologist and the Paediatric Cardiovascular surgeon.