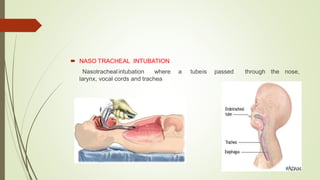
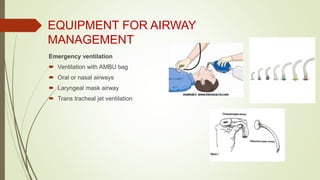
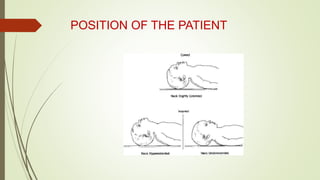
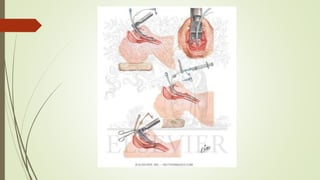
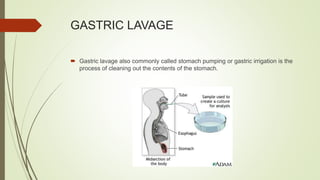
This document discusses various advanced neonatal procedures including endotracheal intubation, mechanical ventilation, and gastric gavage. It provides details on how to perform endotracheal intubation including indications, equipment, patient positioning, techniques, and complications. It also describes the purposes of mechanical ventilation including ventilation, oxygenation, and gas exchange. Various modes, settings, and indications for mechanical ventilation are outlined.