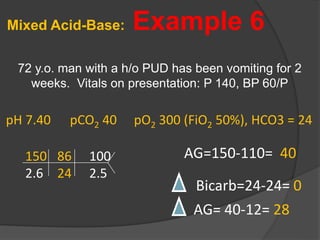
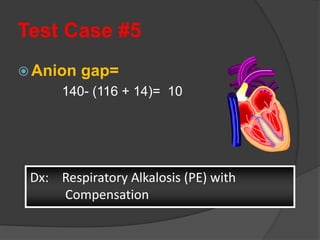
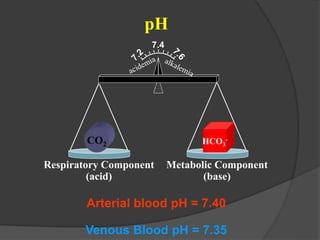
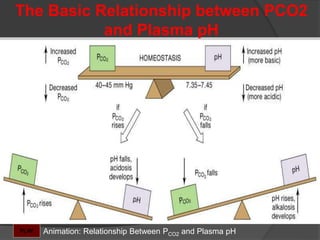
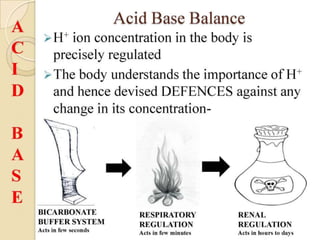
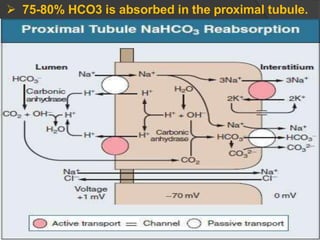
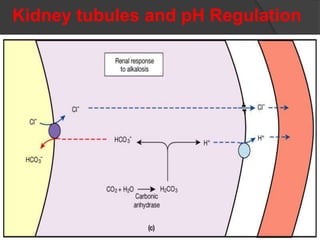
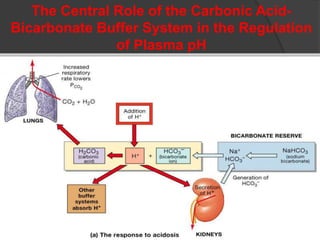
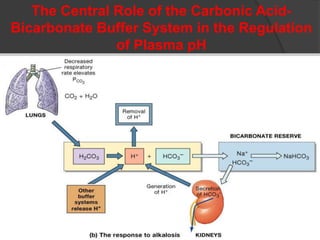
This document discusses arterial blood gas analysis and acid-base physiology. It provides indications for obtaining an ABG such as respiratory or metabolic disorders, hypoxia, shock, sepsis, and decreased cardiac output. It then defines the components of an ABG - pH, PaCO2, PaO2, HCO3, and base excess - and their normal ranges. It explains the Henderson-Hasselbalch equation and how the kidneys and respiratory system work to regulate pH levels and compensate for acid-base imbalances through bicarbonate and CO2 elimination. Various acid-base disorders like respiratory acidosis, metabolic acidosis, and mixed disorders are covered.


![ PaO2; the partial pressure of oxygen in arterial blood
normal range breathing air > 75 mmHg on air (increases with FiO2)
pH; the acidity or alkalinity of the blood, determined by the
concentration of hydrogen ions [H+]
normal range 7.35 – 7.45
PaCO2; the partial pressure of carbon dioxide in arterial blood
normal range 35 – 45 mmHg
Bicarbonate; a buffer, neutralises the effects of excess acid
normal range 22 – 26 mmol L-1
Base excess; a measure of the degree of excess acid or alkali
(base) in the blood
normal range +2 to -2 mmol L-1
Terms used in arterial blood gas analysis](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-3-320.jpg)


![CENTRAL EQUATION OF ACID-BASE
PHYSIOLOGY
Henderson Hasselbach Equation:
where [ H+] is related to pH by
To maintain a constant pH, PCO2/HCO3- ratio should
be constant.
When one component of the PCO2/ [HCO3-] ratio is
altered, the compensatory response alters the other
component in the same direction to keep the
PCO2/[HCO3- ] ratio constant.
[H+] in nEq/L = 24 x (PCO2 / [HCO3 -]
[ H+] in nEq/L = 10 (9-pH)](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-6-320.jpg)

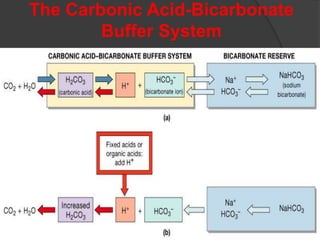
![Bicarbonate-Carbonic Acid Buffer System
CO2 + H2O H2CO3 HCO3 + H
(carbonic anhydrase)
at pH = 7.40 and pCO2 = 40: [HCO3] = 24
Henderson-Hasselbalch Equation:
pH = pK + log 10 [(HCO3)/(H2CO3 + CO2)]
(at normal) = 6.1 + log 10 (24/pCO2 x .03)
= 6.1 + log (24/1.2) = 6.1 + log 20 =
6.1 + 1.3 = 7.40](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-8-320.jpg)




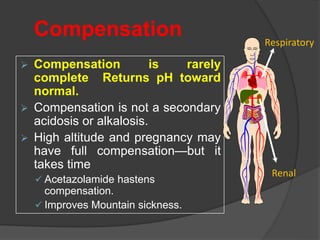
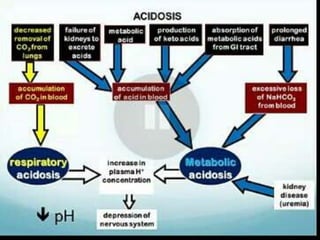
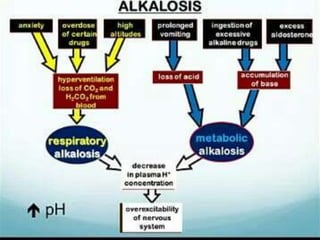
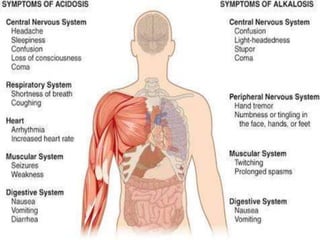

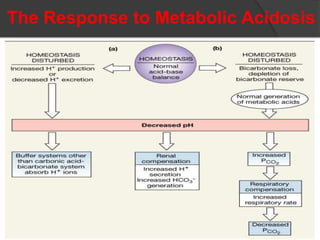



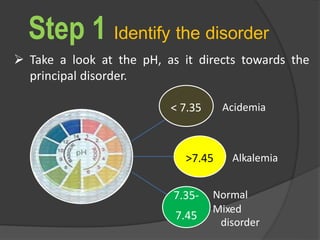
![Acidemia and Alkalemia vs.
acidosis and alkalosis
pH < 7.36 ([H+] > 44) is acidemia.
pH > 7.44 ([H+] < 36) is alkalemia.
mixed disorders of acidosis and
alkalosis may be neither acidemic
nor alkalemic.
e.g. 7.40/ 25/ pCO2/ 15](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-32-320.jpg)






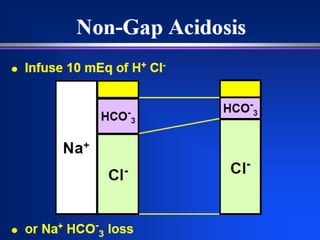
![Metabolic Acidosis: Normal
AG
Loss of HCO3:
Severe diarrhea.
Post-hypocapnia.
Ureteroileostomy.
Acetazolamide.
Failure to excrete [H+]
Renal Tubular Acidosis
○ Types 1-4.
○ Toluene.
Administration of [H+]
Ammonium chloride.
1. Loss of HCO3
2. Failure to excrete [H+]
3. Administration of [H+]](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-45-320.jpg)

















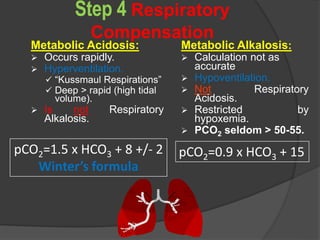
![Six Steps for Acid-Base Analysis
Step 5:
Is the respiratory compensation adequate?
Expected pCO2 range =
[1.5(measured HCO3
-)]+8+/- 2
[1.5 (5) +8] +/- 2 = [13.5 – 17.5]
pCO2 = 10
Therefore it IS a respiratory alkalosis.](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-65-320.jpg)






![Six Steps for Acid-Base Analysis
Step 5:
Is the respiratory compensation
adequate?
Expected pCO2 range =
[1.5(measured HCO3
-)]+8+/- 2
[1.5 (10) +8] +/- 2 = [21 - 25]
E.pCO2 = 25, therefore this is normal
respiratory compensation](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-72-320.jpg)






![Six Steps for Acid-Base Analysis
Step 5:
Is the respiratory compensation adequate?
Expected pCO2 range =
[1.5(measured HCO3-)]+8+/- 2
[1.5 (8) +8] +/- 2 = [18-22]
pCO2 = 28, therefore this is a respiratory acidosis
even though the value is below 40!!](https://image.slidesharecdn.com/abgcardiologyboard15-1-2024-240123195236-d012606f/85/Acid-base-and-ABG-interpretation-in-ICU-79-320.jpg)