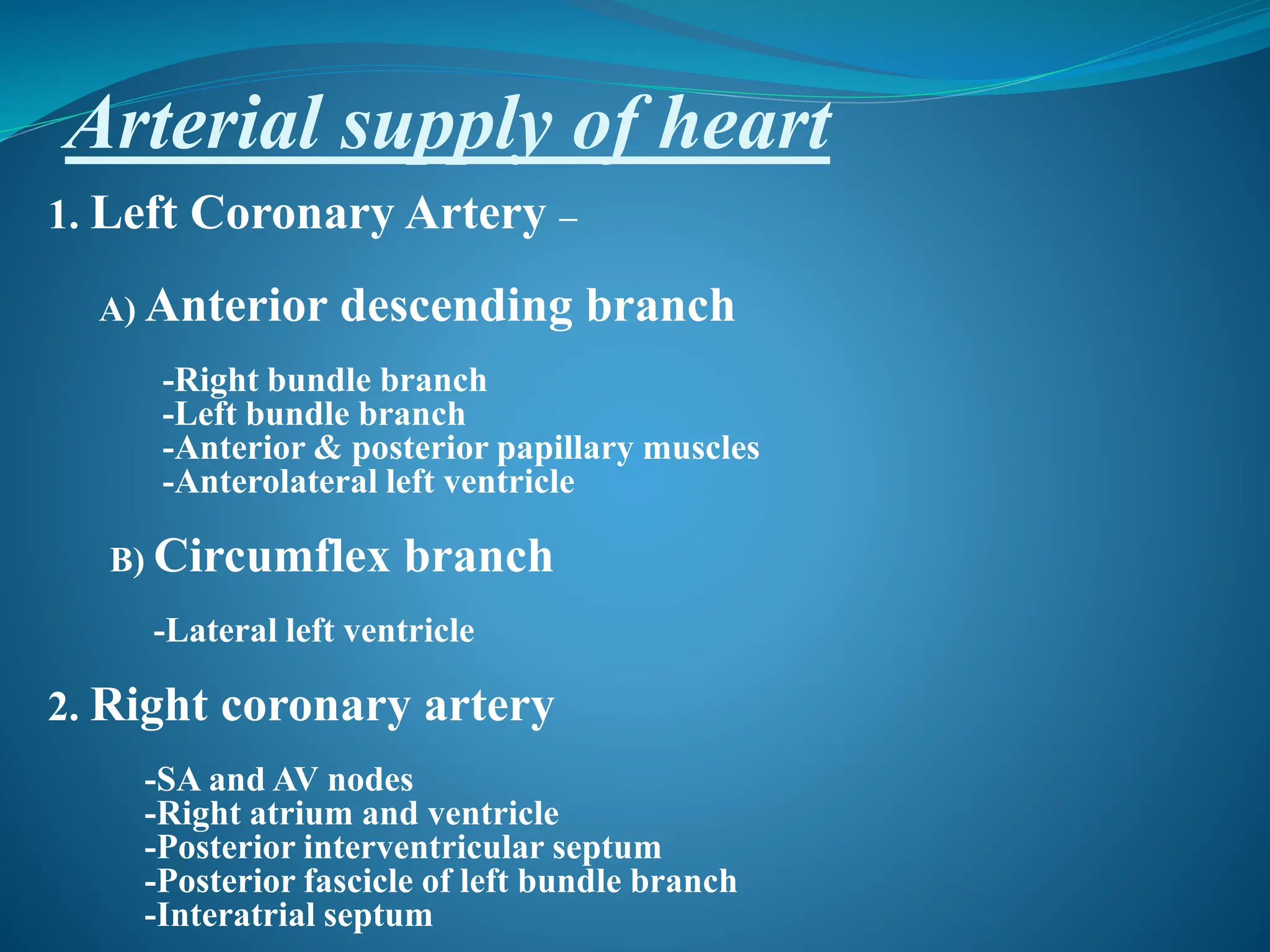
This document discusses the arterial supply of the heart, control of coronary circulation, determinants of myocardial oxygen supply and demand, ischemic heart disease, risk factors and pathophysiology of IHD, and evaluation and management of cardiac patients undergoing non-cardiac surgery. Key points include:
- The left coronary artery supplies the left side of the heart and the right coronary artery supplies the right side.
- Coronary blood flow is regulated by metabolic demands and weakly by the autonomic nervous system.
- Ischemic heart disease results from an imbalance between myocardial oxygen supply and demand.
- Risk factors for IHD include age, smoking, hypertension, diabetes, and family history.
- Evaluation of cardiac patients for