Cacoub p hcv meh 2014 - partie ii
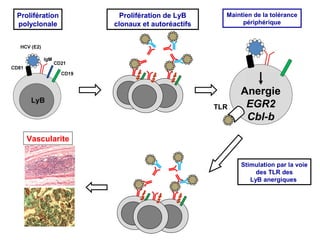
- 1. Prolifération polyclonale Prolifération de LyB clonaux et autoréactifs Maintien de la tolérance périphérique TLR Anergie EGR2 Cbl-b Vascularite Stimulation par la voie des TLR des LyB anergiques 2
- 2. A Major Role for T Cell Immunity in HCV-Vasculitis Abnormal T lymphocytes distribution Predominant T lymphocytes infiltration in vasculitis lesions Th1 cytokines profile in vasculitis lesions MHC-II polymorphism (DR11) Deficit in Treg lymphocytes 3
- 3. GWAS Study of HCV- and Cryoglobulin Related Vasculitis An association with rs9461776 (OR= 2.14, p=1.40E-07) between HLA-DRB1 and DQA1 was detected and further replicated (p=0.01) in additional samples. Zignego AL et al Washington AASLD 20013 5
- 4. Quantitative Deficit in Treg Lymphocytes (CD4+CD25+) in HCV-Vasculitis Boyer O, Saadoun D et al, Blood 2004 6
- 5. 7
- 6. Complete clinical response of HCV-vasculitis to anti-viral treatment is associated with an increase in CD4+CD25high Treg cells 6 6 ** -CR † ** † -NR/PR 5 5 4 4 4 3 Before treatment On treatment arly F/u E On Before treat. Treat. Early F/U Late F/U Late F/U. After Treat. C † * 40 CD 25high(cells /μl) CD25high (% of CD4+) A 30 20 10 0 N R C R CR NR/PR After Treat. B ef or e T x Before Treat. Landau DA et al, Arthritis Rheum 2008 8
- 7. Correlation between Immunological Response and Treg Lymphocytes in HCV MC Vasculitis 0.4 3 R²-0.16 , p<0.005 2 C 4 (g/l ) Cryoglobulins ( g/l ) R²-0.1, p< 0.005 1 0.2 0.0 0 0 20 40 60 CD 25high (cells /μl) Landau DA et al, Arthritis Rheum 2008 80 100 0 20 40 60 80 100 CD25 high (cells /μl) 9
- 8. HCV-related B-Cell Lymphoproliferative Disorders Antigen-sensitive B cell proliferation HCV (E2) DC Antigen-insensitive B cell proliferation DC CD81 Cytokines BAFF B cell Hyperγglobulinemia Polyclonal proliferation IgG IgH-bcl2? Cryoglobulinemia Vasculitis B-cell lymphoma Oligo/Monoclonal proliferation Anti-E2 IgM/Rheumatoid factor Other oncogenic events ? B-cell lymphoma Uncontrolled proliferation 10
- 9. HCV Cr yoglobulinemic Vasculitis Tr eatments
- 10. Chronic HCV infection HCV eradication Poly- oligoclonal B-cell expansion Immunosuppressors Autoantibodies RF - IC Mixed cryoglobulins Monoclonal B-cell proliferation Overt lymphoma Chemotherapy Plasma exchange Cryoglobulinemic vasculitis Steroids 12
- 11. Chronic HCV infection HCV eradication Poly- oligoclonal B-cell expansion Immunosuppressors Autoantibodies RF - IC Mixed cryoglobulins Monoclonal B-cell proliferation Overt lymphoma Chemotherapy Plasma exchange Cryoglobulinemic vasculitis 13
- 12. Clinical Remission of HCV-Related Vasculitis is Correlated to Sustained Virological Response (SVR) Progress in Anti-viral Therapy of HCV (1990-2011) Zuckerman, J Rheumatol 2000. Naarendorp, J Rheumatol 2001. Cacoub, Arthritis Rheum 2002, Zaja F, Blood 2003. Sansonno D, Blood 2003 , Cacoub, Arthritis Rheum 2005, Saadoun, Arthritis Rheum 2007, Saadoun, Ann Rheum Dis 2013 14
- 13. Predictive Factor s of Response to HCV T herapy in Cr yoglobulinemic Vasculitis Multivariate Analysis Odds ratio [95%CI] p • Renal involvement 0.27 [0.08-0.87] 0.02 • Renal insufficiency (GFR<70) 0.18 [0.05-0.67] 0.01 • Daily proteinuria > 1g 0.32 [0.09-1.11] 0.05 • Early virological response 3.53 [1.18-10.59] 0.02 Cacoub, Arthritis Rheum 2005, Saadoun, Arthritis Rheum 2007 15
- 14. Clinical Remission of HCV-Related Vasculitis is Correlated to Sustained Virological Response (SVR) Progress in Anti-viral Therapy of HCV (1990-2011) Zuckerman, J Rheumatol 2000. Naarendorp, J Rheumatol 2001. Cacoub, Arthritis Rheum 2002, Zaja F, Blood 2003. Sansonno D, Blood 2003 , Cacoub, Arthritis Rheum 2005, Saadoun, Arthritis Rheum 2007, Saadoun, Ann Rheum Dis 2013 16
- 15. Chronic HCV infection Poly- oligoclonal B-cell expansion Autoantibodies RF - IC Mixed cryoglobulins Immunosuppressors Monoclonal B-cell proliferation Overt lymphoma Chemotherapy Cryoglobulinemic vasculitis 17
- 16. Overall Survival of 151 HCV-Vasculitis Patients Overall survivall 32 deaths after a median follow-up of 54 months (IQR 26-89) Causes of death: - Infection (n=10) - Cirrhosis (n=10; 4 HCC) - Non-HCC neoplasia (n=4) - Cardiovascular (n=4) - Renal failure (n=2) - Vasculitis (n=2) - Unknown (n=2) Years Terrier B et al. Arthritis Rheum 2010 18
- 17. Prognostic Factors During follow-up Use of Peg-IFN/riba had a positive prognostic impact HR = 0.34 (0.16-0.67) After adjustment on vasculitis severity, immunosuppressants showed a negative impact HR = 4.05 (1.75-9.36) 19 Terrier B et al. Arthritis Rheum 2010
- 18. Chronic HCV infection Poly- oligoclonal B-cell expansion Autoantibodies RF - IC Mixed cryoglobulins Immuno-modulators Rituximab Monoclonal B-cell proliferation Overt lymphoma Cryoglobulinemic vasculitis 20
- 19. Rationale for Rituximab treatment in cryoglobulinemic vasculitis Roccatello, D. et al. Nephrol. Dial. Transplant. 2004 Rocatello D, Nephrol Dial Transplant, 2004 21
- 20. Treatment of Mixed Cryoglobulinemia Resistant to Interferonα with Rituximab Sansonno D et al, Zaja F et al, Blood 2003 22
- 21. Cryoglobulinemia Vasculitis: Poor Response Maintenance after Discontinuation of Rituximab 15 (93.7) 100 90 13 (81.2) 12 (75) 80 70 60 10 (62.5) 50 40 6 (37.5) 30 20 10 1 2 3 4 5 24 6 7 8 9 10 11 12 36 48 MONTHS Sansonno D et al, 2007 23
- 22. Rituximab for the Treatment of Severe Cryoglobulinemic Vasculitis RTX non-RTX De Vita S, Arthritis Rheum 2012 24
- 23. PegIFN plus Ribavirin Rituximab HCV Vasculitis: a TwoFaces Disease … Needs a Two Faces Treatment Strategy 27
- 24. 28
- 25. Outcome of HCV-MC according to treatment All PegIFNα-ribavirin n=93 Parameters n=55 RTX-PegIFNαribavirin n=38 P 8.4 ± 4.7 5.4 ± 4.0 0.004 68 (73.1) 40 (72.7) 28 (73.7) 0.98 22 (23.6) 3 (3.2) 17 (18.3) 13 (23.6) 2 (3.6) 10 (18.1) 9 (23.7) 1 (2.6) 7 (18.4) 49 (52.7) 24 (43.6) 26 (68.4) 35 (37.6) 8 (8.6) 17 (18.3) 25 (45.4) 6 (10.9) 10 (18.1) 10 (26.3) 2 (5.2) 7 (18.4) 55 (59.1) 33 (60) 22 (57.9) 5 (5.4) 2 (3.6) 3 (7.9) Time clinical response, months 6.8 ± 4.7 Clinical response CR PR NR Relapse Immunological response CR PR NR Relapse 0.001 Virological response SVR Death 0.94 0.70 29
- 26. Course of kidney parameters in HCV-MC according to the type of treatment PegIFNα-ribavirin n=10 Kidney inv. CR Creatininemia (µmol/l) Baseline EOF GFR (ml/min) Baseline EOF p 4 (40) 150 ± 30 169 ± 44 58 ± 7 59 ± 9 RTX-PegIFNαribavirin n=21 p 17 (80.9) 0.04 0.28 217 ± 47 136 ± 27 0.03 0.41 42 ± 5 57 ± 4 0.01 Daily Proteinuria (gr/d) Baseline 3.1 ± 0.9 EOF 1.2 ± 0.5 3±1 0.046 0.4 ± 0.1 <0.001 Hematuria (n,%) Baseline EOF 10 (100) 19 (90.5) 2 (20) 2 (10.5) <0.001 30
- 27. RTX/Peg-IFNα-Ribavirin vs. Peg-IFNα-Ribavirin in HCV Systemic Vasculitis Maintenance of Complete Response Dammacco F et al, Blood 2010 31
- 28. 32
- 29. Time Course of HCV Viral Load Terrier B et al. Arthritis Rheum 2009 33
- 30. 34
- 31. 35
- 32. 36
- 33. • If failure or contra-indication to PegINF/ribavirin, Rituximab may be used alone • If Geno1 HCV infection, combination of PegIFN/Ribavirin/Protease inhibitor. HCV: hepatitis C virus; PegIFN: pegylated interferon alpha; CNS: central nervous system; 37
- 34. Chronic HCV infection Poly- oligoclonal B-cell expansion Autoantibodies RF - IC Mixed cryoglobulins Immuno-modulators Low dose IL2 Monoclonal B-cell proliferation Overt lymphoma Cryoglobulinemic vasculitis 38
- 35. Reversible Quantitative Deficit in Treg Lymphocytes (CD4+CD25+) in HCV-Systemic Vasculitis A CD25high (% of CD4+) Boyer, Blood 2004. Landau Arthritis Rheum 2008 ** † -CR 6 6 5 5 4 4 4 3 Before treatmentOn On treatment arly F/u E Before treat. 3 Treat. Early F/U Late F/U Late F/U. After Treat. R²-0.1, p< 0.005 2 0.4 R²-0.16, p<0.005 C4 (g/l) Cryoglobulins ( g/l ) ** † -NR/PR 1 0 0 20 40 60 CD 25high (cells /μl) 80 100 0.2 0.0 0 20 40 60 80 CD25 high (cells /μl) 100 39
- 36. 40
- 37. Effects of Low-Dose Interleukin-2 on Levels of CD4Treg in Patients with HCV-Vasculitis, According to Treatment Course (C). 41
- 38. No Impact of Low-Dose Interleukin-2 on Levels Effector T Cells CD4+CD8+ in Patients with HCV-Vasculitis 42
- 39. Temporal Effects of Low-Dose Interleukin-2 on Clinical Features & Levels of Regulatory T Cells for Each Study Patient CD4+Treg (%) 30 30 30 30 20 20 20 20 10 10 10 10 Baseline C1 C2 C3 C4 Post IL-2 0 Baseline C1 C2 C3 C4 Post IL-2 0 Baseline C1 C2 C3 C4 Post IL-2 0 Baseline C1 C2 C3 C4 Post IL-2 Arthralgia Fatigue Kidney Involvement Neuropathy Purpura CLINICAL RESPONSE 0 Baseline C1 C2 C3 C4 Post IL-2 Baseline C1 C2 C3 C4 Post IL-2 Baseline C1 C2 C3 C4 Post IL-2 Baseline C1 C2 C3 C4 Post IL-2 Saadoun D et al, NEJM 2012
- 40. Anti-inflammatory Effects of Low-Dose Interleukin-2 Revealed through Unsupervised Transcriptome Analyses of PBMCs. BEFORE IL2 AFTER IL-2 CCL3 CCL3L1 CCL3L3 Up Down Khi2 test Inflammation 0 251 1,30E-40 Immune Response 16 684 3,40E-94 Lymphocyte 77 555 7,00E-49 Cell Cycle 1701 208 1,50E-138 Control 226 343 2,50E-01 Autoimmune & transplantation pathologies 0 46 7,60E-09 Inflammatory infectious diseases 6 242 7,60E-36 190 211 4,15E-02 IER3 CXCR7 OLR1 PDE48 PTGS2 IL1B IL1A CCL20 IL6 CLECL1 CD79A BLK CCL4L2 EBF1 CCL4L1 CXCR5 BAFFR 4-1BBL PLAUR NLRP3 RIPK2 ATF3 NAMPT-PBEF1 TNFRSF21DR6 ETS2 MAPK3K8-COT Other diseases GOS2 CD83 Saadoun D et al. NEJM 2011 45
- 41. Other Extra Hepatic Manifestations Associated with HCV Infection Fatigue Poor Health Related Quality of Life (HRQoL) Depression, cognitive impairment Major Cardiovascular Events (MACE) Insulin-resistance, T2DM Chronic kidney diseases
- 42. Fatigue, Depression and Extra Hepatic Manifestations (EM) in HCV Patients % of patients n=1614 % of controls n=412 Fatigue without depression 48 0.7 Fatigue with depression Depression without fatigue No fatigue and no depression 5 2 45 100 19 35 21 25 100 0 0 99.3 100 0.5 0.2 3.4 96 100 TOTAL Fatigue without EM Fatigue with EM EM without fatigue No fatigue and no EM TOTAL Poynard T et al. J Viral Hep, 2002
- 43. Fatigue, Depression and Extra Hepatic Manifestations (EM) in HCV Patients % of patients n=1614 % of controls n=412 Fatigue without depression 48 0.7 Fatigue with depression Depression without fatigue No fatigue and no depression 5 2 45 100 19 35 21 25 100 0 0 99.3 100 0.5 0.2 3.4 96 100 TOTAL Fatigue without EM Fatigue with EM EM without fatigue No fatigue and no EM TOTAL Poynard T et al. J Viral Hep, 2002
- 44. Association Fatigue, Depression and Extrahepatic manifestations (EM) in HCV Patients Multivariate analysis Fatigue (moderate or severe) in comparison to absence of fatigue was associated with: female gender, age > 50 years, Cirrhosis or many septa, purpura. Independently, fatigue was associated with: arthralgia, myalgia, paresthesia, sicca sd & pruritus. Poynard T et al. J Viral Hep, 2002
- 45. Fatigue Rate in Untreated HCV Patients 18 months vs baseline Baseline 18 months 39% 35% 26% 42% 39% 19% P=0.74 41% 37% 22% 69% 24% 7% P<0.001 40% 42% 18% 46% 40% 14% P=0.18 Non treated (n=72) No fatigue Moderate Severe Sustained responders (n=82) No fatigue Moderate Severe Non responders (n=224) No fatigue Moderate Severe Poynard T et al. J Viral Hep, 2002
- 46. Fatigue Rate in HCV Patients Non-Responders to IFN-RBV 18 months vs baseline Baseline 18 months 39% 35% 26% 42% 39% 19% P=0.74 41% 37% 22% 69% 24% 7% P<0.001 40% 42% 18% 46% 40% 14% P=0.18 Non treated (n=72) No fatigue Moderate Severe Sustained responders (n=82) No fatigue Moderate Severe Non responders (n=224) No fatigue Moderate Severe Poynard T et al. J Viral Hep, 2002
- 47. Decreased Fatigue Rate in HCV Patients Sustained Responders to IFN-RBV 18 months vs baseline Baseline 18 months 39% 35% 26% 42% 39% 19% P=0.74 41% 37% 22% 69% 24% 7% P<0.001 40% 42% 18% 46% 40% 14% P=0.18 Non treated (n=72) No fatigue Moderate Severe Sustained responders (n=82) No fatigue Moderate Severe Non responders (n=224) No fatigue Moderate Severe Poynard T et al. J Viral Hep, 2002
- 48. HCV Infection, Fatigue and Depression Fatigue prevalence ranges from 50 to 67% independently predicts poor HRQOL Depression documented in 28% of HCV patients prior to HCV therapy (DSM-IV). predictive of HRQOL during HCV therapy with peginterferon plus ribavirin. HCV may directly affect the CNS: through alterations in serotonergic and dopaminergic neurotransmission with resultant depressive symptoms.
- 49. Functional Assessment of Chronic Illness Fatigue (FACIT-F) in Patients Treated with SOF + PR FACIT-F Scores During and Post-Treatment in FUSION FACIT-F Scores During and Post-Treatment in NEUTRINO † † † 0.75 * * Normalized FACIT-F Normalized FACIT-F † † * 0.65 FACIT-F: All FACIT-F: No SVR FACIT-F: SVR 0.55 0.75 0.65 * FACIT-F: All FACIT-F: No SVR FACIT-F: SVR 0.55 0 4 8 12 16 Week 20 24 28 0 4 8 12 16 20 24 28 Week SVR12 associated with an improvement in fatigue scores Fatigue is worsened by PEG-IFN and/or RBV-related side effects and is less severely impacted by IFN-free regimens regardless of the length of active treatment FUSION & NEUTRINO Trials *P<0.05 decrement from patients’ own baseline † P<0.05 improvement from patients’ own baseline Younossi ZM, et al. AASLD 2013. Washington, DC. #2211
- 50. 2013
- 51. Actigraphy Parameters in HCV Patients Monday 18.04.2011 Tuesday 19.04.2011 Wednesday 20.04.2011 Thursday 21.04.2011 Friday 22.04.2011
- 52. 24 hours physical activity levels in HCV patients and controls Patients (n = 20) Controls (n = 19) P-value Daily sedentary time 654.7 (134.5) 642.3 (100.5) 0.747 Daily light time 227.3 (36.5) 227.3 (36.5) 260.0 (54.5) 260.0 (54.5) 0.038 0.038 Daily lifestyle time 255.6 (57.7) 245.7 (64.9) 0.616 Daily moderate time 219.8 (69.0) 211.6 (102.2) 0.773 Daily vigourous time 2.7 (9.8) 7.3 (19.7) 0.363 222.5 (72.9) 214.9 (104.4) 0.793 MVPA Total counts in bouts/24h 516043.2 (193788.9) 517989.0 (411773.6) 0.985 Total bouts 0.576 39.8 (13.6) 36.2 (24.7)
- 53. 2012
- 54. Cerebral MR Signal in HCV Patients and Spectral Analysis A C B
- 55. MR Signal in Basal Ganglia Myo-Inositol/Creatinine Ratio in HCV Patients According to Virological Response Significant reductionsT3 inbasal ganglia Cho/Cr and T1 vs. in SVR; p<0.05 MI/Cr in SVRs but not in NRs/relapsers. SVRs demonstrated improvements in verbal learning and visuo-spatial memory. Baseline (T1), week 12 t2), and for treatment candidates, 12 weeks post treatment with PEG-IFN and ribavirin (T3) T1 vs. T3 in SVR; p<0.05
- 56. Using PET, microglial activation positively correlated with HCV viraemia and altered cerebral metabolism in the brains of patients with mild hepatitis C. In vivo evidence for a neurotropic role for HCV.
- 57. HCV Infection and Poor HRQoL SF-36 questionnaire: patients with chronic HCV infection consistently show deficits in several domains. HRQOL worsens: with more advanced liver disease. with interferon- and ribavirin-based therapy, potentially leading to a reduction in adherence. Eradication of HCV: correlates positively with improvements in HRQoL.
- 58. 2013 2013
- 59. Health Related Quality of Life and HCV Therapy using Sofosbuvir physical functioning (PF), bodily pain (BP), general health (GH), vitality (VT), and mental health (MH).
- 60. HRQoL in FISSION GT 2 and GT 3 Treatment-Naïve: SOF+RBV vs Peg-IFN+RBV P<0.05 Significant difference between treatment arms at weeks 12 and 24 HRQoL Change in Summary Score 2 SOF+RBV EOT 0 SOF+RBV Physical Component Summary SOF+RBV Mental Component Summary SOF+RBV EOT -2 Peg-IFN+RBV Physical Component Summary -4 Peg-IFN+RBV EOT -6 Peg-IFN+RBV Mental Component Summary Peg-IFN+RBV EOT -8 Baseline 12 Weeks 24 Weeks Follow Up Week 12 Patients treated with SOF+RBV had better HRQoL scores at the end of treatment as compared to patients receiving PegIFN+RBV Younossi ZM, et al. EASL 2013. Amsterdam, The Netherlands. #1431
- 61. Other Extra Hepatic Manifestations Associated with HCV Infection Fatigue Poor Health Related Quality of Life (HRQoL) Depression, cognitive impairment Major Cardiovascular Events (MACE) Insulin-resistance, T2DM Chronic kidney diseases
- 62. Antiviral treatment for hepatitis C virus infection is associated with improved renal and cardiovascular outcomes in diabetic patients Short running title: Antiviral treatment for HCV and outcomes of DM Yao-chun Hsu1,2, Jaw-Town Lin 2,3,4, Hsiu J. Ho3, Yu-Hsi Kao5, Yen-Tsung Huang6, Nai-Wan Hsiao7, Ming-Shiang Wu8, Yi-Ya Liu9, Chun-Ying Wu1,9,12 Hepatology, 2013, in press
- 63. 2,267,270 patients with a diagnosis of diabetes mellitus (DM) between January 1, 1997 and December 31, 2011 1,630,156 DM patients without hepatitis B virus infection 746,280 patients with diabetes mellitus 3,957 HCV-infected patients ever treated with Peg-IFN+RBV 20,239 HCV-infected patients never treated with Peg-IFN+RBV 720,302 patients without HCV infection 1,411 patients in the treated cohort 1,411 patients in the untreated cohort 5,644 patients in the uninfected cohort
- 64. Baseline characteristics and follow-up status of the tree study cohorts Age (mean + SD) Gender, n (%) Female Male Comorbidity, n (%) Hyperlipidemia Hypertension Thyroid disorder Compensated cirrhosis COPD Peripheral arterial disease Diabetes medication, n(%) Metformin monotherapy Other oral monotherapy Two oral agents Three or more oral drugs Insulin dependence Uncategorized Treated cohort (n = 1411) 54.92 + 8.06 Untreated cohort (n = 1411) 55.01 + 7.99 Uninfected (n = 5644) 54.93 + 8.07 491 (34.8) 920 (65.2) 493 (34.9) 918 (65.1) 1964 (34.8) 3680 (65.2) 177 (12.5) 606 (42.9) 139 (9.9) 324 (23) 482 (34.2) 95 (6.7) 175 (12.4) 606 (42.9) 137 (9.7) 322 (22.8) 481 (34.1) 91 (6.4) 718 (12.7) 2444 (43.3) 544 (9.6) 1289 (22.8) 1924 (34.1) 373 (6.6) 96 (6.8) 533 (37.8) 302 (21.4) 174 (12.3) 138 (9.8) 168 (11.9) 80 (5.7) 569 (40.3) 311 (22) 157 (11.1) 117 (8.3) 177 (12.5) 346 (6.1) 2366 (41.9) 1093 (19.4) 682 (12.1) 474 (8.4) 683 (12.1) P* 0.932 0.995 0.952 0.953 0.96 0.994 0.999 0.954 0.107
- 65. Cumulative Incidence of End Stage Renal Disease (death adjusted as a competing risk event)
- 66. Cumulative Incidence of Stroke (death adjusted as a competing risk event)
- 67. Cumulative Incidence of Acute Coronary Events (death adjusted as a competing risk event)
- 68. Hepatology, 2012
- 69. Baseline Features in Gen1 HCV Patients and Gender, Age & BMI-Matched Controls Carotid intima-media 1.04 + 0.21 0.90 + 0.16 < 0.001 73 (41.9) 40 (23) < 0.001 Thickness, mm Carotid plaques
- 70. Risk Factors Associated with the Presence of Carotid Plaques in Gen1 HCV Patients Histology at biopsy Steatosis (%) 13 + 18.5 14.5 + 19 0.60 28/73 8/65 0.007 0.756 (0.327, 1.748) 0.51 73/28 37/36 0.003 2.177 (1.043, 4.542) 0.03 Grade of inflammation 1-2/3 Stage of fibrosis 1-2/3-4
- 71. Risk Factors Associated with the Presence of Carotid Plaques in Gen1 HCV Patients Carotideal Plaques (%) 100 80 P=0.008 P=0.51 60 40 20 0 N=67 CHC Age ≤55 yrs F0-2 N=21 CHC Age ≤55 yrs F3-4 N=43 CHC Age >55 yrs F0-2 N=43 CHC Age >55 yrs F3-4
- 72. 2013
- 73. LHID 2000 1,000,000 individuals 9220 Patients with a new diagnosis of HCV between January 1, 2004 and December 31, 2007 2453 Exclused Patients who were diagnosed as having HCV on only 1 occassion 6767 Patients with a new diagnosis of HCV 3654 Exclused 43 Age <20 2986 History of HCV before January 1, 2004 84 History of IBT before the first HCV diagnosis 1167 History of Stroke before the first HCV diagnosis 12452 Non-HCV cohort Matched by age and sex at a ratio of 1:4 3113 HCV cohort with a diagnosis of HCV on at least two occassions 30 208 IBT cohort received treatment for a period of >3 months 2875 Non-IBT cohort received no IBT between 2004 and 2008 Exclused Received IBT for a period of <3 months
- 74. Hepatitis C virus infection and stroke risk Cohorts Hazard ratio Non HCV P-value* 1.21 HCV 95% CI 1.05 - 1.40 0.011 1.06 - 1.43 0.006 1.06 - 1.42 0.008 1 1.23 HCV ‡ Non HCV 1 1.23 HCV † Non HCV 1 HCV invection was associated with a 23% increased risk of stroke, after adjusting for known prognostic factors. † Adjusted for age, sex, hyperlipidaemia, DM, IHD, hypertension, alcohol-related illness, COPD, aspirin use, clopidogrel use, warfarin use, dipyridamole use, ticlopidine use, statin use, ACE inhibitors use and influenza vaccination. ‡ Adjusted for age, DM, aspirin use and ACE inhibitors use.
- 75. Interferon-Based Therapy and Stroke-Free Survival in HCV Patients 100 Stroke-free survival rate IBT 0.95 Non-IBT Log-rank test, p = 0.003 0.90 0.85 IBT, interferon based therapy 0.80 0 1 2 3 Time (years) 4 5
- 76. Interferon-based therapy and stroke risk in patients with hepatitis C Hazard ratio Non IBT IBT ‡ Non IBT IBT † Non IBT P-value* 0.28 IBT 95% confidence interval 0.12 - 0.69 0.005 0.16 - 0.93 0.033 0.16 - 0.95 0.039 1 0.38 1 0.39 1 Interferon based therapy was associated with a 61% decreased risk of stroke in HCV patients after adjusting for known prognostic factors. † Adjusted for age, sex, hyperlipidaemia, DM, IHD, hypertension, alcohol-related illness, COPD, aspirin use, clopidogrel use, warfarin use, dipyridamole use, ticlopidine use, statin use, ACE inhibitors use and influenza vaccination. ‡ Adjusted for age, DM, aspirin use and ACE inhibitors use.
- 77. 2012
- 78. Myocardial injury in HCV patients Characteristics Age (yr) Sex Liver function Bilirubin (mg/dl) ALT (IU/L) Cardiac function Y-globulin (g/dl) Abnormal ECG (%) activity (%) Prothrombin percent IGC disappearance rate CPK (IU/L) HAI score (point) LDH (IU/L) Cardiac function Abnormal ECG (%) BNP (pg/ml) CPK (IU/L) LDH (IU/L) HANP (pg/ml) BNP (pg/ml) LVDd (mm) HANP (pg/ml) LVDd (mm) Ejection fraction (%) Ejection fraction (%) Severity score (point) Severity score (point) Severity score > 3 (%) Severity score > 3 (%) Chronic hepatitis C (n = 217) 57 + 9 104/113 0.7 + 0.3 77 + 61 1.6 + 0.3 90 + 16 0.172 + 0.041 8.9 + 3.3 9 94 + 46 172 + 38 22 + 18.8 94 9 46 + 19.6 + 12.5 172 + 38 22 + 18.8 48 + 5 19.6 + 12.5 48 + 5 66 + 7 66 + 7 4.3 + 1.6 4.3 + 1.6 87 87 Normal range 0.2 - 1.0 5 - 45 0.7- 1.2 80 - 100 300.158 - 0.232 - 190 107 - 230 Less than -18.4 30 190 107 230 Less than-43 Less than 18.4 39 - 55 43 Less than 55 - 39 - 55 80 55 - 80 Less Less than 3 than 3 HAI, histology activity index; BNP, brain natriuretic peptide; HANP, human atrial natriuretic peptide; LVDd, left ventricular end diastolic dimension
- 79. Myocardial SPECT Images in HCV Patients According to Virological Response SVR Before IFN therapy (M0; A), at the completion of IFN therapy (M6; B) and 6 months after the completion of IFN therapy (M12; C). The arrows show the regions of myocardial perfusion defects.
- 80. Myocardial SPECT Images in HCV Patients According to Virological Response SVR Relapse Before IFN therapy (M0; A), at the completion of IFN therapy (M6; B) and 6 months after the completion of IFN therapy (M12; C). The arrows show the regions of myocardial perfusion defects.
- 81. Myocardial SPECT Images in HCV Patients According to Virological Response SVR Relapse Non Response Before IFN therapy (M0; A), at the completion of IFN therapy (M6; B) and 6 months after the completion of IFN therapy (M12; C). The arrows show the regions of myocardial perfusion defects.
- 82. Severity Score of Myocardial Perfusion Defects in HCV Patients after 24 Weeks IFN IFN SVR group (n= 62) Relapse group (n= 48) NVR group (n= 45)
- 83. Severity Score of Myocardial Perfusion Defects in HCV Patients after 48 weeks PegIFN/Ribavirin IFN SVR group (n= 30) Relapse group (n= 9) NVR group (n= 6)
- 84. Other Extra Hepatic Manifestations Associated with HCV Infection Fatigue Poor Health Related Quality of Life (HRQoL) Depression, cognitive impairment Major Cardiovascular Events (MACE) Insulin-resistance, T2DM Chronic kidney diseases
- 85. Manifestation Prevalence certainly associated with HCV % -----------------------------------------------------5-40 • Vasculitis (PAN, cryoglobulinemia) 35-54 • Fatigue • Arthralgia-myalgia 25-35 10-25 • Sicca syndrome 10-40 • Autoantibodies • Thrombocytopenia 20-40 ? • Lymphoma
- 86. Auto-antibody production in chronic HCV infection. 70 60 A-nuclear A-phospholipid A-thyroglobulin A-smooth muscle ≥ one auto-Ab ≥ three auto-Ab 50 40 30 20 10 0 % Pawlotsky JM, Hepatology 1994. Pawlotsky JM, Ann Intern Med 1994. Prieto J, Hepatology 1996. Cacoub P, J Rheumatol 1997. Cacoub P, Medicine 2000.
- 87. Extrahepatic manifestations associated with HCV infection. (Prospective study in 321 HCV patients) Autoantibody Number % ----------------------------------------------------Antinuclear 124 41 • A-nucleosome 6 2 • A-DNA 8 3 • A-histone 9 3 • A-ENA 10 3 Cacoub P et al. Medicine 2000; 79: 47-56
- 89. Hepatitis C virus : extrahepatic manifestations, an update 2007 B-cell-Non Hodgin’s Lymphoma 2462 tested 13.5 % positive • vs 0-5 % in controls • vs 5 % in other malignant hemopathy Hepatitis C virus 469 tested 0 - 39 %
- 90. Hepatitis C virus : extrahepatic manifestations, an Effects of alpha-interferon on HCV+/SLVL course update 2007 HCV antibodies : B-NHL (< 3%) vs SLVL (15%) ----> Splenic lymphoma with villous lymphocytes may be associated with HCV infection After 6 months of IFN alpha treatment in SLVL/HCV+: Complete clinical hematologic response (spleen size < 12 cm, lymphocytosis <4500/mm3, No cytopenia ): ---> 7/9 HCV RNA negative Partial clinical hematologic response (spleen size or lymphocytosis decrease >50%) : ---> 2/9 HCV RNA + Hermine O. et al, N Engl J Med 2002; 347: 89-94
- 91. Extrahepatic Manifestations of HCV Infection Summary Extra-hepatic manifestations of chronic HCV infection include vasculitis, lymphomas, glucose related disorders, and rheumatologic conditions. Chronic HCV is associated with reduced health-related quality of life, in which depression, cognitive impairment and fatigue may be factors. Increased risk of major cardiovascular events and chronic kidney disease require attention. Clinicians should appreciate the extra-hepatic as well as the hepatic consequences of HCV infection and the potential of treatment strategies to reduce this overall impact. Healthcare costs imposed by these conditions must be considered in addition to those normally associated with chronic HCV infection.
- 92. Merci D. Saadoun, Paris D. Sene, Paris B. Terrier, Paris G. Géri, Paris P. Hausfater, Paris O. Lidove, Paris A. Gatel, St Brieuc J-M. Léger, Paris N. Limal, Paris T. Maisonobe, Paris JC Piette, Paris S. Caillat-Zucman, Paris P. Ghillani, Paris D. Klatzmann, Paris L. Musset, Paris M. Rosenzwajg, Paris L. Calabrese, Cleveland M. Casato, Roma C. Ferri, Modena G. Kerr, Washington E. Sasso, Seattle JA. Schifferli, Basel V. Soriano, Madrid L. Alric, Toulouse M. Bourlière, Marseille P. Halfon, Marseille S. Pol, Paris T. Poynard, Paris V. Thibault, Paris Les membres du GERMIVIC 107