More Related Content
What's hot
What's hot (20)
2012 topic 18 1 calculations involving acids and bases

2012 topic 18 1 calculations involving acids and bases
Recently uploaded
Driving Behavioral Change for Information Management through Data-Driven Gree...

Driving Behavioral Change for Information Management through Data-Driven Gree...Enterprise Knowledge
Recently uploaded (20)
The 7 Things I Know About Cyber Security After 25 Years | April 2024

The 7 Things I Know About Cyber Security After 25 Years | April 2024
2024: Domino Containers - The Next Step. News from the Domino Container commu...

2024: Domino Containers - The Next Step. News from the Domino Container commu...
Tech Trends Report 2024 Future Today Institute.pdf

Tech Trends Report 2024 Future Today Institute.pdf
Driving Behavioral Change for Information Management through Data-Driven Gree...

Driving Behavioral Change for Information Management through Data-Driven Gree...
IAC 2024 - IA Fast Track to Search Focused AI Solutions

IAC 2024 - IA Fast Track to Search Focused AI Solutions
What Are The Drone Anti-jamming Systems Technology?

What Are The Drone Anti-jamming Systems Technology?
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke

ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...

Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...
The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf

The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf
Automating Google Workspace (GWS) & more with Apps Script

Automating Google Workspace (GWS) & more with Apps Script
Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff

Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff
Powerful Google developer tools for immediate impact! (2023-24 C)

Powerful Google developer tools for immediate impact! (2023-24 C)
Raspberry Pi 5: Challenges and Solutions in Bringing up an OpenGL/Vulkan Driv...

Raspberry Pi 5: Challenges and Solutions in Bringing up an OpenGL/Vulkan Driv...
Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...

Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...
Boost PC performance: How more available memory can improve productivity

Boost PC performance: How more available memory can improve productivity
Unknown E
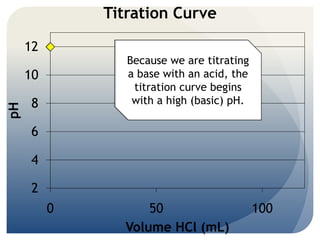
- 1. Titration Curve 12 Because we are titrating 10 a base with an acid, the titration curve begins 8 with a high (basic) pH. pH 6 4 2 0 50 100 Volume HCl (mL)
- 2. Titration Curve 12 The titration curve begins to flatten, hinting that 10 we are nearing a pKa 8 pH 6 4 2 0 50 100 Volume HCl (mL)
- 3. Titration Curve 12 The curve 10 begins to roll over, confirming 8 that we have a pH pKa≈9.5 6 4 2 0 50 100 Volume HCl (mL)
- 4. Titration Curve 12 10 The steep drop 8 in pH indicates pH that we are 6 passing the equivalence 4 point 2 0 50 100 Volume HCl (mL)
- 5. Titration Curve 12 pKa=9.5 Unknown E = Uracil 10 8 pH 6 4 2 0 50 100 Volume HCl (mL)