Kartik Juris QR.pptx
•Download as PPTX, PDF•
0 likes•7 views
Jurisprudence
Report
Share
Report
Share
Recommended
More Related Content
Similar to Kartik Juris QR.pptx
Similar to Kartik Juris QR.pptx (15)
National pharmaceutical pricing authority with drugs price control order (dpc...

National pharmaceutical pricing authority with drugs price control order (dpc...
More from YashThorat20
More from YashThorat20 (15)
Recently uploaded
Recently uploaded (20)
Call US 📞 9892124323 ✅ Kurla Call Girls In Kurla ( Mumbai ) secure service

Call US 📞 9892124323 ✅ Kurla Call Girls In Kurla ( Mumbai ) secure service
Russian Call Girls In Gtb Nagar (Delhi) 9711199012 💋✔💕😘 Naughty Call Girls Se...

Russian Call Girls In Gtb Nagar (Delhi) 9711199012 💋✔💕😘 Naughty Call Girls Se...
High Class Call Girls Nagpur Grishma Call 7001035870 Meet With Nagpur Escorts

High Class Call Girls Nagpur Grishma Call 7001035870 Meet With Nagpur Escorts
20240429 Calibre April 2024 Investor Presentation.pdf

20240429 Calibre April 2024 Investor Presentation.pdf
VIP Kolkata Call Girl Jodhpur Park 👉 8250192130 Available With Room

VIP Kolkata Call Girl Jodhpur Park 👉 8250192130 Available With Room
Solution Manual for Principles of Corporate Finance 14th Edition by Richard B...

Solution Manual for Principles of Corporate Finance 14th Edition by Richard B...
Call Girls Service Nagpur Maya Call 7001035870 Meet With Nagpur Escorts

Call Girls Service Nagpur Maya Call 7001035870 Meet With Nagpur Escorts
Solution Manual for Financial Accounting, 11th Edition by Robert Libby, Patri...

Solution Manual for Financial Accounting, 11th Edition by Robert Libby, Patri...
Log your LOA pain with Pension Lab's brilliant campaign

Log your LOA pain with Pension Lab's brilliant campaign
Dharavi Russian callg Girls, { 09892124323 } || Call Girl In Mumbai ...

Dharavi Russian callg Girls, { 09892124323 } || Call Girl In Mumbai ...
Dividend Policy and Dividend Decision Theories.pptx

Dividend Policy and Dividend Decision Theories.pptx
Independent Call Girl Number in Kurla Mumbai📲 Pooja Nehwal 9892124323 💞 Full ...

Independent Call Girl Number in Kurla Mumbai📲 Pooja Nehwal 9892124323 💞 Full ...
VIP Kolkata Call Girl Serampore 👉 8250192130 Available With Room

VIP Kolkata Call Girl Serampore 👉 8250192130 Available With Room
06_Joeri Van Speybroek_Dell_MeetupDora&Cybersecurity.pdf

06_Joeri Van Speybroek_Dell_MeetupDora&Cybersecurity.pdf
Independent Lucknow Call Girls 8923113531WhatsApp Lucknow Call Girls make you...

Independent Lucknow Call Girls 8923113531WhatsApp Lucknow Call Girls make you...
Monthly Market Risk Update: April 2024 [SlideShare]![Monthly Market Risk Update: April 2024 [SlideShare]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Monthly Market Risk Update: April 2024 [SlideShare]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Monthly Market Risk Update: April 2024 [SlideShare]
Interimreport1 January–31 March2024 Elo Mutual Pension Insurance Company

Interimreport1 January–31 March2024 Elo Mutual Pension Insurance Company
Kartik Juris QR.pptx
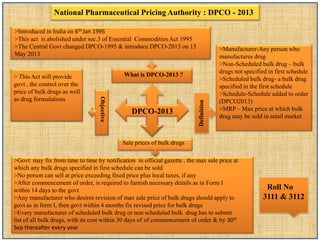
- 1. DPCO-2013 >Introduced in India on 6th Jan 1995 >This act is abolished under sec.3 of Essential Commodities Act 1995 >The Central Govt changed DPCO-1995 & introduce DPCO-2013 on 15 May 2013 > This Act will provide govt , the control over the price of bulk drugs as well as drug formulations >Govt may fix from time to time by notification in official gazette , the max sale price at which any bulk drugs specified in first schedule can be sold >No person can sell at price exceeding fixed price plus local taxes, if any >After commencement of order, is required to furnish necessary details as in Form I within 14 days to the govt >Any manufacturer who desires revision of max sale price of bulk drugs should apply to govt as in form I, then govt within 4 months fix revised price for bulk drugs >Every manufacturer of scheduled bulk drug or non scheduled bulk drug has to submit list of all bulk drugs, with its cost within 30 days of of commencement of order & by 30th Sep thereafter every year >Manufacturer-Any person who manufactures drug >Non-Scheduled bulk drug – bulk drugs not specified in first schedule >Scheduled bulk drug- a bulk drug specified in the first schedule >Schedule-Schedule added to order (DPCO2013) >MRP – Max price at which bulk drug may be sold in retail market National Pharmaceutical Pricing Authority : DPCO - 2013 Definition Sale prices of bulk drugs Objective What is DPCO-2013 ? Roll No 3111 & 3112