6 Urea MoC.pptx
•Download as PPTX, PDF•
0 likes•6 views
Materials used in Urea Plants
Report
Share
Report
Share
Recommended
More Related Content
Similar to 6 Urea MoC.pptx
Similar to 6 Urea MoC.pptx (20)
Materials used in restorations/ orthodontic course by indian dental academy

Materials used in restorations/ orthodontic course by indian dental academy
Effects of Continuous Cooling On Impact and Micro Structural Properties of Lo...

Effects of Continuous Cooling On Impact and Micro Structural Properties of Lo...
Corrosion and Environmental Degradation of Materials-2.pdf

Corrosion and Environmental Degradation of Materials-2.pdf
ENGINEERING CHEMISTRY: Module 2-corrosion & its control - metal finishing

ENGINEERING CHEMISTRY: Module 2-corrosion & its control - metal finishing
Metallurgical difficulties in welding of ferritic martensitic and duplex sta...

Metallurgical difficulties in welding of ferritic martensitic and duplex sta...
Recently uploaded
Recently uploaded (20)
From Event to Action: Accelerate Your Decision Making with Real-Time Automation

From Event to Action: Accelerate Your Decision Making with Real-Time Automation
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf

The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf
Understanding Discord NSFW Servers A Guide for Responsible Users.pdf

Understanding Discord NSFW Servers A Guide for Responsible Users.pdf
Exploring the Future Potential of AI-Enabled Smartphone Processors

Exploring the Future Potential of AI-Enabled Smartphone Processors
Workshop - Best of Both Worlds_ Combine KG and Vector search for enhanced R...

Workshop - Best of Both Worlds_ Combine KG and Vector search for enhanced R...
Powerful Google developer tools for immediate impact! (2023-24 C)

Powerful Google developer tools for immediate impact! (2023-24 C)
08448380779 Call Girls In Greater Kailash - I Women Seeking Men

08448380779 Call Girls In Greater Kailash - I Women Seeking Men
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Advantages of Hiring UIUX Design Service Providers for Your Business

Advantages of Hiring UIUX Design Service Providers for Your Business
Tata AIG General Insurance Company - Insurer Innovation Award 2024

Tata AIG General Insurance Company - Insurer Innovation Award 2024
6 Urea MoC.pptx
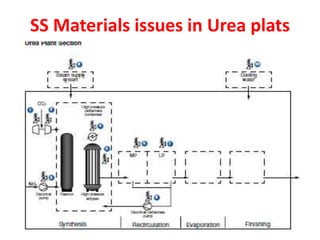
- 1. SS Materials issues in Urea plats
- 3. Amino formic acid can form at low pressure zone (3)- destroy passive film on SS & corrodes Urea Ammonium carbonate
- 5. Influence of oxygen partial pressure on corrosion reaction Reaction(3) is amino formic acid- can form ammonia and CO2 at low pressure itself. This acid is responsible for high corrosion rate urea MoC. (1) ammonia carbamate
- 6. Field SS Corrosion Summary a. Stainless steels (UG) can corrode at HAZ and ferrite rich regions; c. b. Welding start and finish points are defect prone and corrode c. Temper bead type welding procedure improves d. High ferrite forms if welding is done at high heat input (J) e. Urea reactor’s SS lining and tray support bracket lugs corrodes more f. Top dome SS petal type weld lining also corrodes & may even leaks g. In carbamate condenser tube to tube sheet weld area may corrode. TS lining also get affected
- 7. Lab Test results & Conclusions • HPHT expt. with NH3 & CO2 as Bi-molecular reaction indicated the possibility of forming amino-formic acid. • Electrochemical reaction rates are influenced by O2 and the type of passive films on metal surface. • Thus any region which possesses weak passive films- --tend to corrode at high rate selectively. • Passive film formed on Delta ferrite region of SS weld is comparatively weak than the base and can corrode at rates of 2-3mm/year or more. • Contd.-----
- 8. • However, ferrite present in duplex stainless steels is likely to behave differently. • Delta ferrite in weld forms at high temperature and would contain lot of Si, P and other trump/impurity elements act as ‘scavenger’ so the passive film would be much weaker than that formed on ferrite phase in DSS. • Urea grade 316L SS is refined and contain much less Si, P etc., and form least amount delta ferrite and thus has low corrosion rate.