Superior vena cava syndrome
- 1. SUPERIOR VENA CAVA SYNDROME
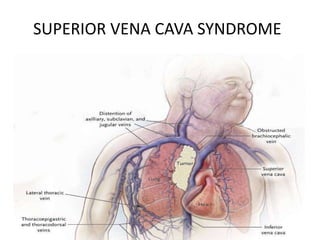
- 2. INTRODUCTION • Superior vena cava syndrome (SVCS) is obstruction of blood flow through the superior vena cava (SVC). • William Hunter first described the syndrome in 1757 in a patient with syphilitic aortic aneurysm.
- 4. Anatomy • Thin walled. • Low pressure vessel. • Drainage of venous blood from upper half of body. • 7cm in length. • 20-22mm in diameter.
- 5. COLLATERALS • There are four possible collateral systems which were first described in 1949 by McIntire and Sykes. 1.Azygos venous system. 2.Internal thoracic venous system. 3.Vertebral venous system. 4.External thoracic venous system.
- 6. Pathophysiology • Pathogenetic basis of SVCS is obstruction to the blood flow. • It can result from intrinsic or extrinsic obstacles. – Intrinsic-thrombosis or invading tissue(uncommon). – Extrinsic factors- compression or stricture of the vein. • When obstruction of the SVC occurs, – Vascular resistances rise and the venous return decreases. – SVC pressure increases consistently. – SVC with significant stenosis- collateral circulation opens up to bypass the obstruction and restore the venous return.
- 7. Obstruction of upper SVC, proximal to Azygos entry point.
- 8. Obstruction with Azygos involvement
- 9. Obstruction of lower SVC, distal to Azygos entry point.
- 10. • The timing of the obstruction development is important for its clinical implications. – In acute impairments, the blood flow is not rapidly distributed through the collateral network so symptoms arise markedly. – In the case of slow-growing diseases, the collateral venous network has enough time to expand in order to receive the circulating volume. • For this reason, long-lasting, severe SVC obstruction can sometimes be found without significant symptoms.
- 11. Etiology • Previously infectious diseases used to be common cause SVCS. • Infections, such as Tuberculosis, Syphilis, Histoplasmosis and Actinomycosis. • Consistent decrease because of: – Antibacterial therapies – Improved social conditions.
- 12. • Benign – Chronic mediastinitis. – Catheter-induced Thrombosis. – Aortic aneurysm – Retrosternal goiter – Pericardial, thymic, bronchogenic cysts. – Lymphadenopaties.
- 13. • Malignant – Non-small cell lung cancer(NSCLC):50% – Small cell lung cancer(SCLC): 22% – Lymphoma: 12% – Mets:9% – Germ cell tumors:3% – Thymoma:2% • Presently, Malignancies are predominant (95%) cause of SVCS. Wilson L et al., NEJM 2007
- 14. • Incidence of Iatrogenic SVCS is currently increasing. • Pacemaker and defibrillator placement . • Thrombosis related to the presence of central vein catheters McCurdy M et al., Crit Care Med 2012
- 15. Classification of SVCS There are three main classification proposals which follow different methods of categorization. • Doty and Standford’s classification (anatomical) – Type I: stenosis of up to 90% of the supra-azygos SVC – Type II: stenosis of more than 90% of the supraazygos SVC – Type III: complete occlusion of SVC with azygos reverse blood flow – Type IV: complete occlusion of SVC with the involvement of the major tributaries and azygos vein • Yu’s classification (clinical) – Grade 0: asymptomatic (imaging evidence of SVC obstruction) – Grade 1: mild (plethora, cyanosis, head and neck edema) – Grade 2: moderate (grade 1 evidence + functional impairment) – Grade 3: severe (mild/moderate cerebral or laryngeal edema, limited cardiac reserve) – Grade 4: life-threatening (significant cerebral or laryngeal edema, cardiac failure) – Grade 5: fatal • Bigsby’s classification (operative risk) – The low risk patients present: • No dyspnea at rest, • No facial cyanosis in the upright position, • No change of dyspnea and No worsening of facial edema and Cyanosis, during the supine position. – The high risk patients present facial cyanosis or dyspnea at rest in the sitting position.
- 16. Doty and Standford’s classification (anatomical) • Type I: Stenosis of up to 90% of the supra-azygos SVC • Type II: Stenosis of more than 90% of the supra- azygos SVC • Type III: Complete occlusion of SVC with azygos reverse blood flow • Type IV: Complete occlusion of SVC with the involvement of the major tributaries and azygos vein
- 17. • The clinical seriousness is related to: 1.Level of obstruction and rapidity of development, determining the effectiveness of collateral circulation. 2. Involvement of other mediastinal structures (compression or invasion of heart, pulmonary artery and central airways, phrenic nerve paralysis) Clinical presentation
- 18. • The clinical signs of this condition are mainly represented by – Cyanosis (due to venous stasis with normal arterial oxygenation) and – Edema of the upper chest, arms, neck and face (periorbital initially). • Swelling is usually more important on the right side, because of the better possibility of collateral circulation in the left brachiocephalic vein compared to the contralateral. • Other signs or symptoms are: 1. Coughing, 2. Epistaxis, 3. Hemoptysis, 4. Dysphagia, 5. Dysphonia and 6. Hoarseness (caused by vocal cord congestion), 7. Esophageal, Retinal and Conjuntival bleeding.
- 19. • In the case of significant cephalic venous stasis— 1.Headache- a common symptom and it is usually continuous and pressing, exacerbated by coughing. 2. Dizziness, 3. Buzzing, 4. Drowsiness, 5. Stupor, Lethargy and even coma may be encountered. • Dyspnea can be directly related to 1.Mediastinal mass or 2. Pleural effusion or 3. Cardiocirculatory impairment. • Supine position may worsen the symptoms.
- 20. • Yu’s classification (clinical) – Grade 0: Asymptomatic (Radiological SVC obstruction in the absence of symptoms) – Grade 1: Mild (Edema in head or neck, cyanosis, plethora) – Grade 2: Moderate (Edema in head or neck with functional impairment) – Grade 3: Severe (mild/moderate cerebral or laryngeal edema, limited cardiac reserve) – Grade 4: Life-threatening (significant cerebral or laryngeal edema, cardiac failure) – Grade 5: Fatal
- 21. Diagnosis • Physical examination is often crucial: – Presence of edema and superficial venous network of the upper chest may support the clinical diagnosis. • Imaging studies are however required.
- 22. • Chest X ray -The most common radiological findings are superior mediastinal widening and pleural effusion.
- 23. • CECT is the most useful tool in the evaluation of the mediastinal syndromes. – Assess level of obstruction. – Differentiate between thrombosis and tumor. – Differentiate between compression and infiltration. • The contrast flow can also help to distinguish the extent of the collateral network.
- 24. • Invasive venography – – Now rarely used due to the huge improvement in vascular CT imaging. – It is currently performed only as a preliminary to operative procedures such as stent placement. • Recently FDG-PETCT has gained an important role.
- 25. • The histological diagnosis remains the key factor for the causative treatment, in the case of neoplastic etiology. • It is necessary to have histological confirmation before starting treatment. • Pathologic confirmation of a thoracic malignancy can be obtained as a cytologic specimen via – Endobronchial fine needle aspiration – Mediastinoscopy – CT-guided needle biopsy
- 26. • Mediastinoscopy has a very high success rate and low complication rate.(5%) • Percutaneous transthoracic CT-guided fine needle biopsy is an effective and safe alternative to an open biopsy or mediastinoscopy. • Thoracocentesis-In presence of a pleural effusion, can establish the diagnosis of malignancy in 71% of patients. • Thoracoscopic biopsy or Thoracotomy is diagnostic if all other procedures have failed.
- 27. Treatment • Treatment depends on 1. Severity of symptoms. 2. Etiology. 3. Prognosis. • Goals – Relieve symptoms. – Attempt cure of the primary malignant process.
- 28. Supportive Care: – Elevation of head end of bed to decrease the hydrostatic pressure and thereby the edema. – Oxygen – Diuretics- reduce cerebral edema. – Glucocorticoids reduce the tumor burden in Lymphoma and Thymoma- likely to reduce the obstruction.
- 29. • Only a small percentage of patients with rapid-onset obstruction of the superior vena cava (SVC) are at risk for life-threatening complications. • Emergency treatment is indicated with – Central airway compromise. – Severe laryngeal edema. – Coma secondary to cerebral edema.
- 30. • Chemotherapy indicated in – Lymphomas. – Germ cell tumors. – Small cell lung cancer.
- 32. • A Cochrane systematic review concluded that chemotherapy and radiotherapy were equally effective at relieving SVCO secondary to lung cancer, where as stent insertion provided higher rates of response more rapidly. (Rowell and Gleeson, 2001).
- 34. Endovascular stents • The placement of an endovascular stent restores venous return and provides rapid and sustained symptomatic relief. • The technical success rate is in the range of 95 to 100%, and over 90% of patients report relief of symptoms. • Indications – – It is a useful procedure for pts with severe symptoms (eg, stridor) who require urgent intervention. – Can be placed before a tissue diagnosis is available – For rapid symptom palliation in pts with NSCLC and mesothelioma and for those with recurrent disease who have previously received systemic therapy or RT.
- 36. • Complications -Reported in 3 to 7% of patients. • Early complications include – Infection, – Pulmonary embolus, – Stent migration, – Hematoma at the insertion site, – Bleeding, and – Rarely, perforation or rupture of the SVC. • Late complications include – Bleeding (1 to 14 percent) and – Death (1 to 2 percent) from anticoagulation and – Stent failure with reocclusion.
- 37. • Stent failure is most often caused by thrombus or tumor ingrowth. • However, since most pts with malignancy related SVC syndrome have a short life expectancy, the stent usually remains patent until death. • If reocclusion does occur, it can be treated with a second stent or thrombolytic therapy, with good secondary patency rates. • Need for long-term anticoagulation — Short-term anticoagulation/antiplatelet therapy is often recommended.
- 38. Surgical intervention • Surgical bypass is now rarely performed because of the success of endovascular stenting. • The main proposal for SVC resection is direct infiltration in thymomas or in N0-N1 non-small cell lung cancer. • Armoured PTFE grafts and biologic material are the preferred choices. • Morbidity after SVC surgical procedures is high and the post-operative care must be intensive. • Long-term patency of a SVC by-pass graft is uncertain but, usually, the slow onset of the graft thrombosis favors the development of effective collateral circulation.
- 39. • RT provides considerable relief by reducing tumor burden. • Response time is 7-15 days but Symptomatic relief may be apparent as soon as 72 hours. (Wan J et al., Emerg Med Clin N Am 2009) • Palliative Schedules are: – 20 Gy in 5 fractions (4Gy/fraction,1 week) – 30 Gy in 10 fractions (3Gy/fraction,2week) Radiation therapy
- 40. • The fractionation schedule for radiotherapy – 2.5-4 Gy in 3 fractions, – followed by daily dose of 1.5-2 Gy, – Total dose of 35-60 Gy. • The radiation dose depends on tumor size and radioresponsiveness. • The radiation portal should include a 2-cm margin around the tumor. • Radiation therapy palliates SVC obstruction in 70% of patients with lung carcinoma and in more than 95% of those with lymphoma.
- 42. Management of Iatrogenic SVCS • Central venous catheter induced thrombosis – Thrombolytics (eg, streptokinase, urokinase, or Recombinant tissue-type plasminogen activator) – Anticoagulants (eg, heparin or oral anticoagulants). • Removal of the catheter, if possible, is another option, and it should be combined with anticoagulation to prevent embolization.
- 44. THANK YOU.
Editor's Notes
- In SVC obstruction, Azygos vein is important in formation of collateral circulation. With respect to collateral pathways, the SVC can be divided into 2 segments: Supra-azygos or pre-azygos and Infra-azygos or post-azygos SVC.
- In physiologic conditions, blood return to the right atrium is facilitated by the pressure gradient between the right atrium and venae . images. Sy WM, Lao RS. Collateral pathways in superior vena cava obstruction as seen on gamma Br J Radiol 1982; 55:294-300
- Once the thoracic imaging is obtained, the work-up should include brain, abdominal and bone studies in view of the probable malignant nature of the primary lesion. Blood tests blood gases FBC, U&E, LFT. Clotting screen. Serum calcium. Uric acid tumour markers: Beta HCG, AFP, LDH, CEA, CA15-3 •
- Treatment includes the following: Initial oxygen as required and steroids EG: dexamethasone 12-16 mg in divided doses, with proton pump inhibitor protection. A superior vena caval stent can be useful holding measure in patients who require urgent symptom relief, and this will often allow time to establish a tissue diagnosis. Thrombosis should be appropriate. Chemotherapy is indicated for patients with chemosensitive tumors such as lymphoma, germ cell tumors or SCLC. Radiotherapy is the mainstay of treatment for patients with other solid tumours. Patients with clinical SVCS often gain significant symptomatic improvement from conservative treatment measures, including elevation of the head of the bed and supplemental oxygen. Corticosteroids and diuretics are often used to relieve laryngeal or cerebral edema, although documentation of their efficacy is questionable.
- It seems appropriate that highly responsive tumours (NHL,HL,SCLC) should be treated with chemotherapy first, whereas patients with NSCLC and severe SVCO should be offered a stent first.
- Chemotherapy and Radiotherapy are effective in relieving SVCO in a proportion of patients whilst stent insertion appears to provide relief in a higher proportion and more rapidly.
- Surgical resection of mediastinal tumor and reconstruction of the SVC is rarely considered in view of its morbidity and mortality and the limited life expectancy of most pts who present with this complication. Surgery indicated in malignant thymoma and thymic carcinoma, which are relatively resistant to chemotherapy and radiation.
- with many patients developing recurrent symptoms before dying of the underlying disease particularly if symptoms are severe, more rapid palliation can be achieved through the use of an intraluminal stent, followed by RT for disease control. SVCO is effectively palliated by EBRT in 60 per cent of patients with NSCLC and 80 per cent with SCLC. Stent placement is also effective in relieving symptoms in patients who fail to respond to RT.
- These agents are most effective when patients are treated within 5 days after the onset of symptoms.
