Viruses and vaccine production
- 1. Viral vaccine and production
- 2. CONTENT 1. What is a vaccine ? 2. Classical vaccines & engineered vaccines 3. Replicating versus inactivated vaccines 4. Purpose of vaccination 5. Crucial aspect defining the process choice 6. Host cell characteristics & growth requirements 7. Advantages & disadvantages of insect cells 8. Virus/antigen stability 9. Vaccine production 10. Vaccine production in bioreactors 11. Process intensification in bioreactors 12. Manufacturing basics
- 3. 1. WHAT IS A VACCINE ? • Biological preparation providing Active Acquired Immunity • Goal of vaccination: to stimulate the body to develop a specialized, protective, immune response in the absence of disease. • Immune responses target viral proteins which can be delivered as : i. Weakened (attenuated) viruses that replicate in the host without causing disease. ii. Inactivated (killed) virions. iii. Purified protein products. iv. Nucleic acids to direct synthesis of desired proteins.
- 4. 2. CLASSICAL VACCINES & ENGINEERED VACCINES CLASSICAL VACCINES ENGINEERED VACCINES 1. Attenuated viruses usually obtained by repeated passage in animals or cultured cells. 1. provide a safe source of antigen, representative of a particular pathogen. 2. Unpredictable results and inadequately attenuated viruses. 2.1 Virulent viruses weakened by deletion/modifications of specific genes 2.2 Genes for capsid or envelope proteins can be transferred to a safer virus. 2.3 genes of virulent virus moved into bacteria, yeast, or cultured cells to produce large quantities of a particular viral protein. 2.4 administer DNA (cloned genes) directly into the recipient. 3. possibility of virus reverting to virulence. 3. use of adjuvants
- 5. 3. REPLICATING VERSUS INACTIVATED VACCINES LIVING VACCINE INACTIVATED VACCINE 1. Fewer, smaller doses and adjuvant unnecessary 1. Do not replicate in recipient 2. Cheap to produce 2. Lower cost to develop 3. Longer protection and less chance of hypersensitivity. 3. Unlikely to cause disease through residual virulence 4. Stable on storage
- 6. 4.Purpose of vaccination • Stimulate the production of neutralizing antibodies for the purpose of blocking infection • Neutralizing antibodies might - i. prevent a virus from binding to a receptor ii. prevent membrane fusion iii. prevent un-coating of the viral genome.
- 7. 5.Crucial aspect defining the process choice 5.1 VACCINE DEMAND: i. Spread of a virus and its mutation rate. ii. Time period Ex: seasonal influenza a time period as short as 5–6 months. iii. Polio or measles require a more or less constant supply of the same vaccine strain for worldwide application. iv. Vaccines against dengue or yellow fever are only needed in certain regions of the world.
- 8. 5.2 VACCINE TYPE i. Live attenuated viruses, inactivated viruses, virus subunits, viral vectors or recombinant virus-like particles/proteins ii. Viruses with a high mutation rate are unsuitable candidates for live attenuated vaccine type as reversion may occur during vaccine production. iii. Attenuated virus strains in cell culture gives lower process yield iv. Wild-type live viruses require biosafety level 3 v. Recombinant vaccines may require higher antigen concentrations per dose
- 11. 5.3 VIRUS/ANTIGEN REQUIREMENTS FOR GROWTH/EXPRESSION i. Chemically defined protein-free media ii. Hepatitis A (strain HM175) propagated in MRC-5 human diploid cells iii. Recombinant hepatitis B surface antigen (HBsAg) produced in yeast cells grown in a complex medium of extract of yeast, soy peptone, dextrose, amino acids, and mineral salts iv. Measles virus propagated in chick embryo cell culture
- 12. 6. HOST CELL CHARACTERISTICS & GROWTH REQUIREMENTS : i. Right cell line & optimal process parameter conditions e.g., temperature, pH value, dissolved oxygen concentration, medium composition, etc. ii. Human/higher animal cell substrates for whole virus replication. iii. Insect cell substrates for recombinant antigen and virus-like particle (VLP) production. iv. Continuous cell lines preferred over primary cells : Vero, MDCK, MRC-5, WI-38, HEK293, PER.C6, AGE1.CR ,EB66 v. Media development- serum free
- 13. 7. ADVANTAGES & DISADVANTAGES OF INSECT CELLS : i. Suspension cultures using serum-free media ii. Insect cell-baculovirus expression system iii. Difficultly to express membrane proteins , secreted glycoproteins at appropriate levels or complex glycan structures 8. VIRUS/ANTIGEN STABILITY : i. Hollow-fiber-based perfusion systems enables selective separation and further processing of the product-containing medium. ii. To avoid virus/antigen degradation in upstream processing, which is mainly caused by the release of cellular proteases after cell lysis low thermo-stabilities.
- 14. 9. VACCINE PRODUCTION Roller bottles i. A practical and low-cost option for cell culture at laboratory scale and for large-scale manufacturing of products ii. Automated handling of up to 1000 RBs per batch (IDT Biologika)
- 16. 10. VACCINE PRODUCTION IN BIOREACTORS DISCONTINUOUS BATCH CULTIVATION MODE i. Low instrumental and operational intervention ii. Good virus yield coefficients and high nutrient consumption iii. Fed-batch or perfusion systems aims on higher cell concentrations & increased volumetric virus yields
- 17. 11. PROCESS INTENSIFICATION IN BIOREACTORS i. Microcarriers ii. Fixed-bed systems or packed-bed systems iii. Shake flasks , wave bioreactors or STRs iv. Fed-batch mode v. Perfusion systems (external hollow-fiber) vi. Continuous bioreactor vii. Use of ‘Single-use bioreactors’ in vaccine production
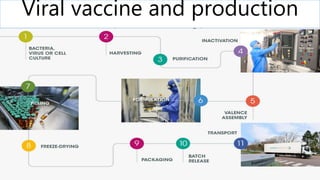
- 18. 12. MANUFACTURING BASICS GENERATION of the pathogen recombinant protein derived from the pathogen RELEASE the antigen from the substrate and isolate it from the bulk of the environment PURIFICATION of the antigen unit operations of column chromatography & ultrafiltration inactivation of isolated virus. FORMULATION of the vaccine include an adjuvant QUALITY CONTROL (QC) testing safety, potency, purity, sterility, and other assays specific to the product STORAGE addition of stabilizers or lyophilization , very low temperatures