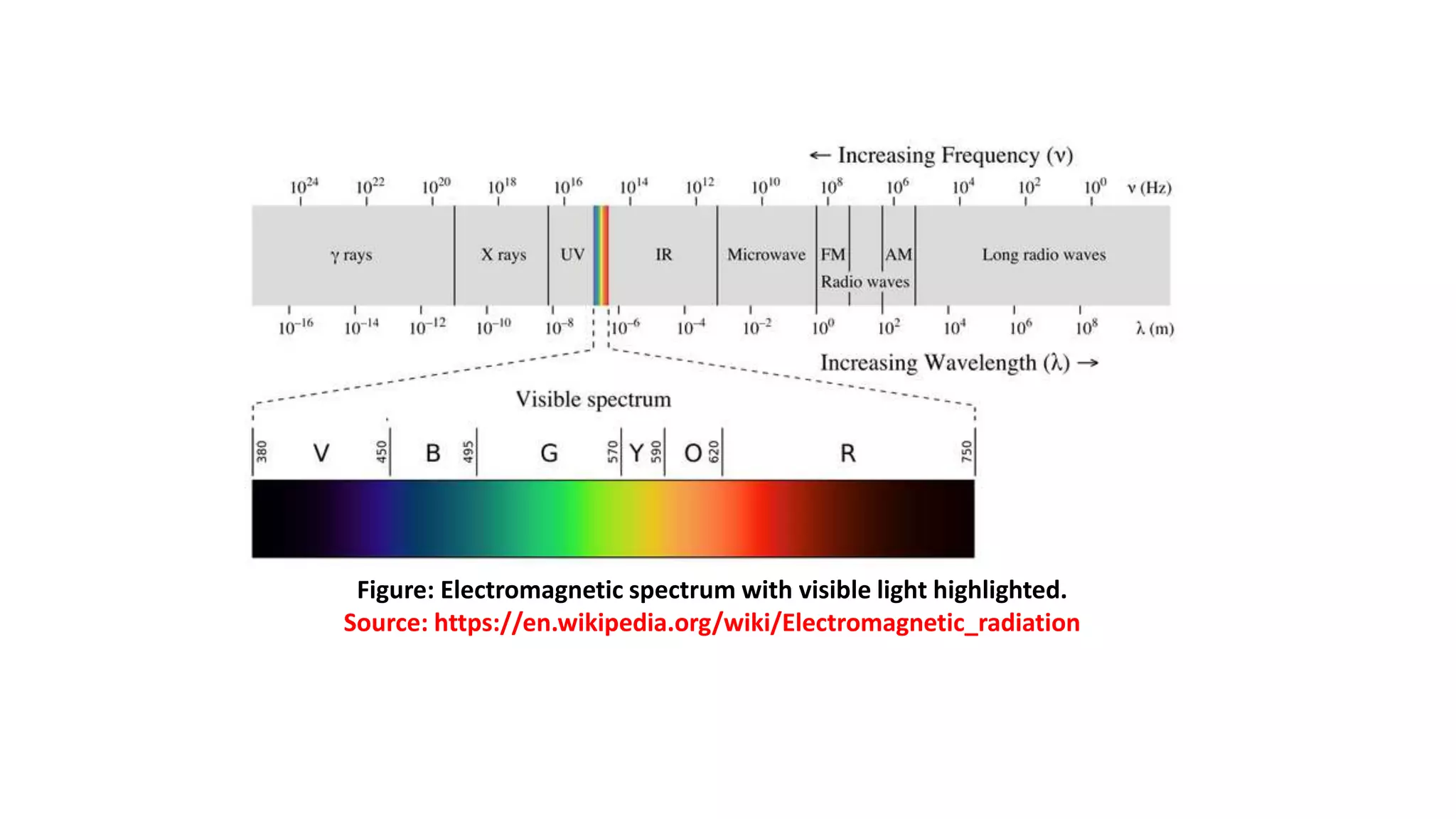
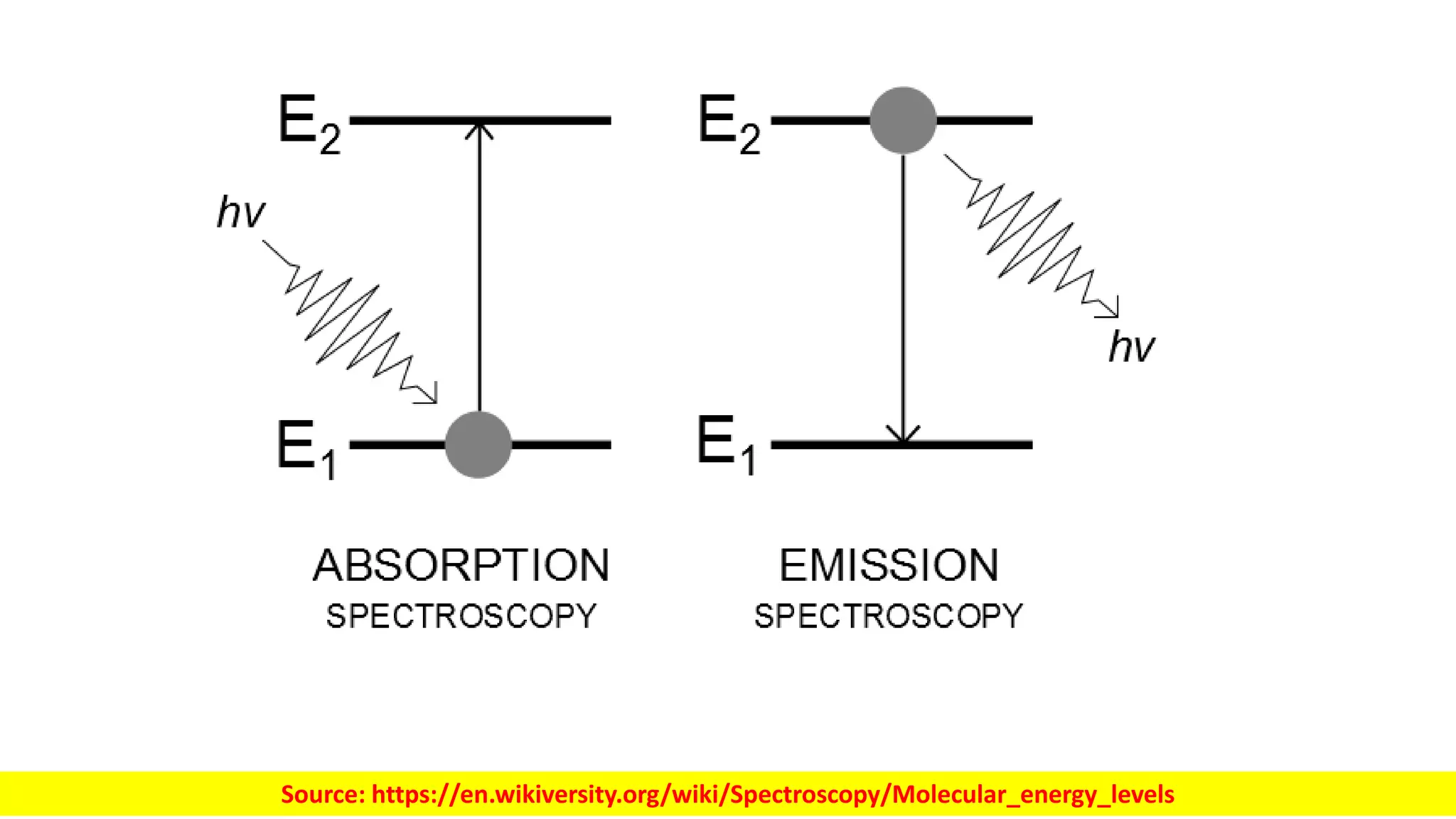
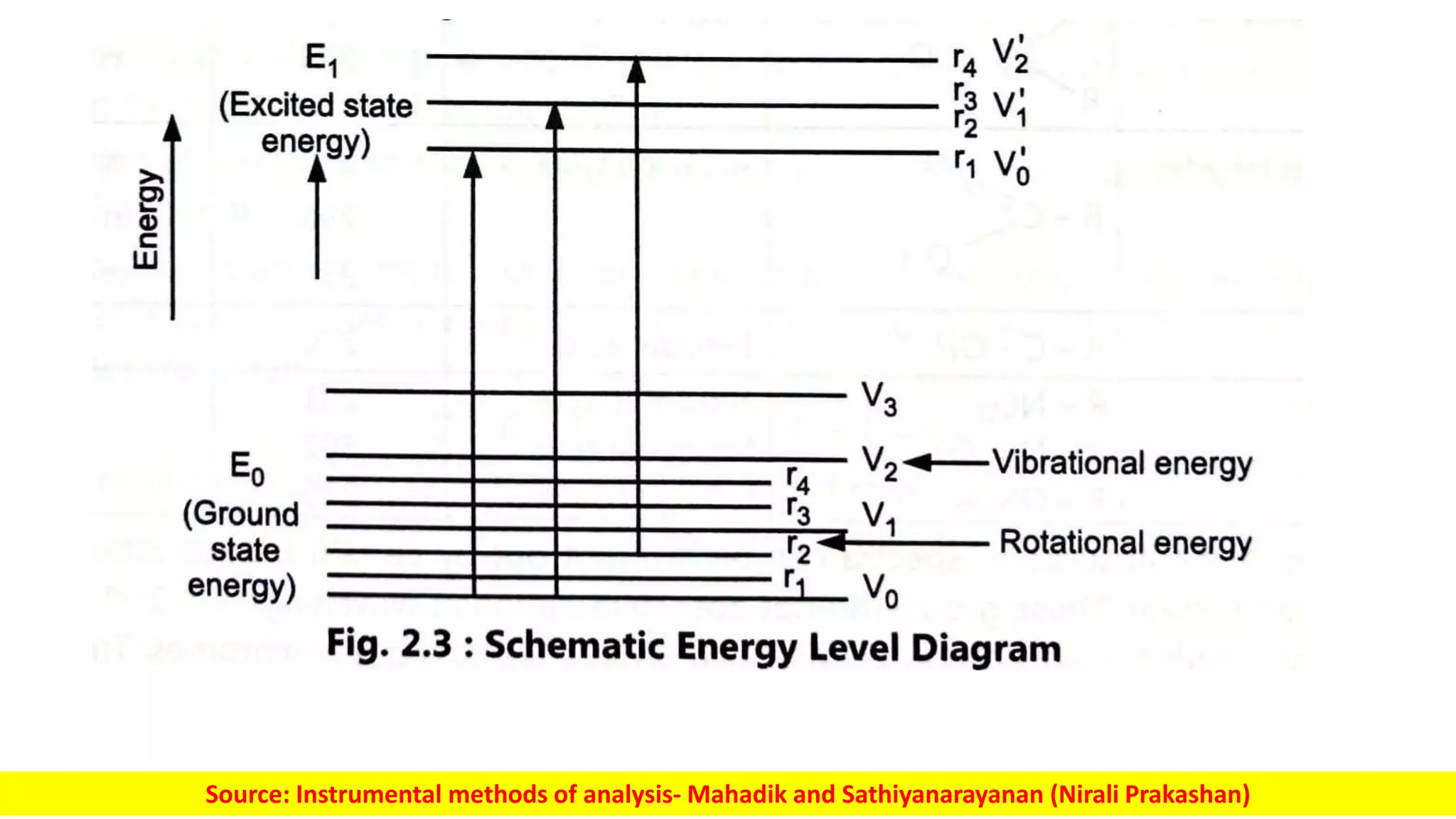
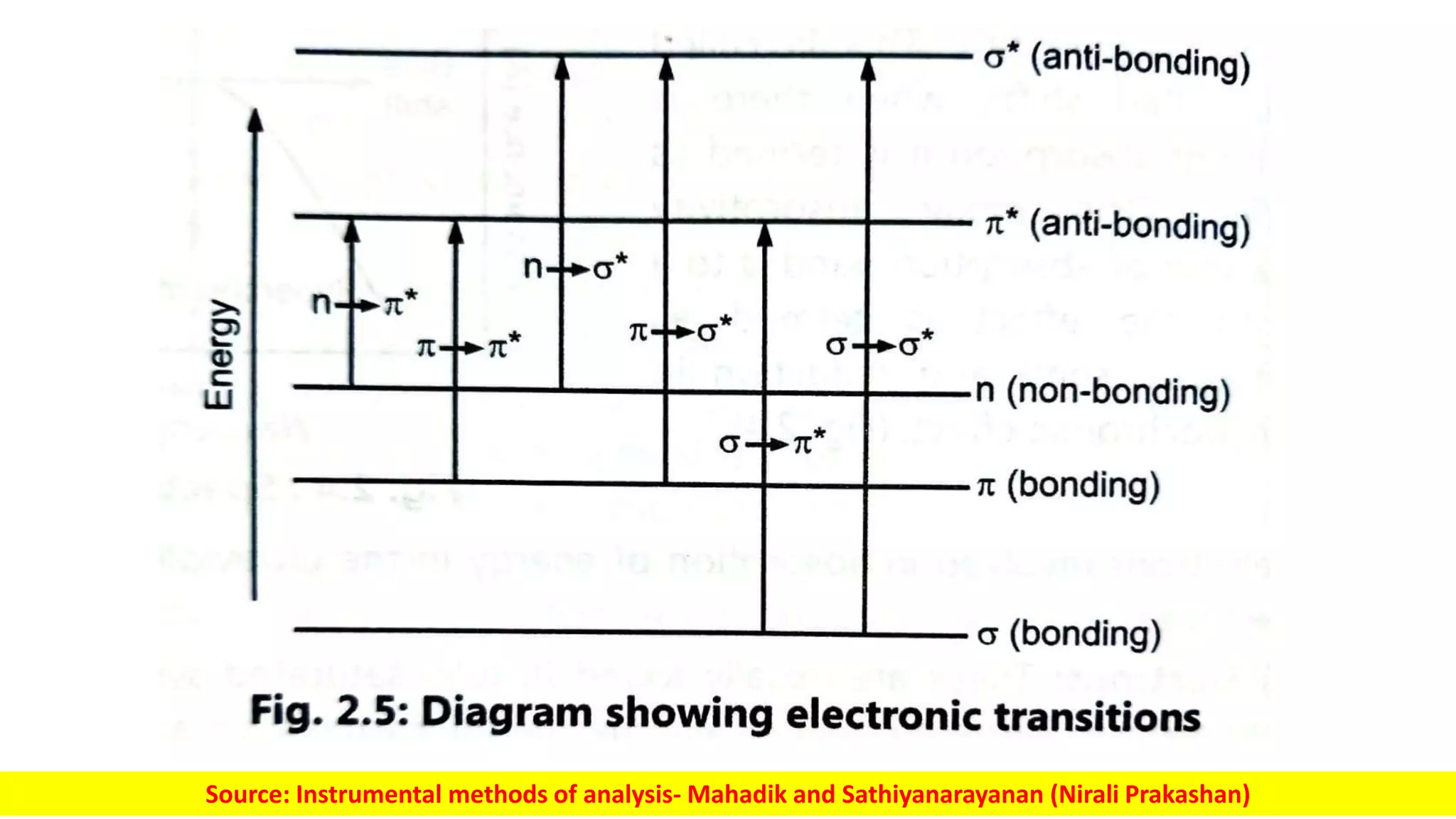
Electromagnetic radiation spans a wide spectrum of frequencies and wavelengths that interact with matter. Spectroscopy techniques analyze these interactions by measuring absorption or emission of electromagnetic radiation. Different regions of the electromagnetic spectrum are used in techniques like UV-vis spectroscopy, infrared spectroscopy, and microwave spectroscopy to study molecular structure and identify functional groups. Absorption occurs when radiation excites molecules to higher energy states, while emission spectroscopy analyzes radiation released as molecules return to lower states.