Tracing the Arc of Immunotherapy in Melanoma: Insights on the Current Role and Cutting-Edge Uses of Checkpoint Inhibitors for Patient Care
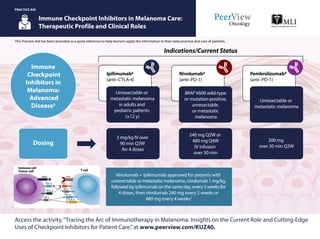
- 1. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Immune Checkpoint Inhibitors in Melanoma Care: Therapeutic Profile and Clinical Roles PRACTICE AID Access the activity,“Tracing the Arc of Immunotherapy in Melanoma: Insights on the Current Role and Cutting-Edge Uses of Checkpoint Inhibitors for Patient Care,”at www.peerview.com/KUZ40. Ipilimumab2 (anti–CTLA-4) Unresectable or metastatic melanoma in adults and pediatric patients (≥12 y) Nivolumab3 (anti–PD-1) BRAF V600 wild-type or mutation-positive, unresectable, or metastatic melanoma Pembrolizumab4 (anti–PD-1) Unresectable or metastatic melanoma 3 mg/kg IV over 90 min Q3W for 4 doses Nivolumab + ipilimumab approved for patients with unresectable or metastatic melanoma, nivolumab 1 mg/kg, followed by ipilimumab on the same day, every 3 weeks for 4 doses, then nivolumab 240 mg every 2 weeks or 480 mg every 4 weeks3 240 mg Q2W or 480 mg Q4W IV infusion over 30 min 200 mg over 30 min Q3W Indications/Current Status Immune Checkpoint Inhibitors in Melanoma: Advanced Disease1 Dosing MHC TCR PD-L1 PD-L1 CTLA-4 PD-1 PD-L2 B7.1 B7.1 T cell Immune cell Tumor cell
- 2. Immune Checkpoint Inhibitors in Melanoma Care: Therapeutic Profile and Clinical Roles CTLA-4: cytotoxic T-lymphocyte–associated antigen-4; PD-1: programmed death 1; Q2W: every 2 wk; Q3W: every 3 wk; Q4W: every 4 wk; Q12W: every 12 wk. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology: Melanoma. v2.2018. https://www.nccn.org/professionals/physician_gls/pdf/melanoma.pdf. Accessed May 14, 2018. 2. Yervoy (nivolumab) Prescribing Information. https://packageinserts.bms. com/pi/pi_yervoy.pdf. Accessed May 14, 2018. 3. Opdivo (nivolumab) Prescribing Information. https://packageinserts.bms.com/pi/pi_opdivo.pdf. Accessed May 14, 2018. 4. Keytruda (pembrolizumab) Prescribing Information. http://www.merck.com/product/usa/pi_circulars/k/keytruda/ keytruda_pi.pdf. Accessed May 14, 2018. 5. Eggermont AMM et al. N Engl J Med. 2018;378:1789-1801. PRACTICE AID Access the activity,“Tracing the Arc of Immunotherapy in Melanoma: Insights on the Current Role and Cutting-Edge Uses of Checkpoint Inhibitors for Patient Care,”at www.peerview.com/KUZ40. Ipilimumab2 (anti–CTLA-4) Cutaneous melanoma with pathologic involvement of regional lymph nodes of >1 mm after complete resection, including total lymphadenectomy Nivolumab3 (anti–PD-1) Pembrolizumab5 (anti–PD-1) Indications/Current Status Immune Checkpoint Inhibitors in Melanoma: Adjuvant Therapy1 Dosing Phase 3 data in patients with completely resected stage III disease; 43% reduction in risk of recurrence or death (HR = 0.57; P < .001) Melanoma with lymph node involvement or metastatic disease after complete resection 10 mg/kg IV over 90 min Q3W for 4 doses, then 10 mg/kg Q12W ≤3 y or documented disease recurrence or unacceptable toxicity 240 mg over 30 min Q2W or 480 mg over 30 min Q4W 200 mg over 30 min Q3W (used in phase 3 trial)
- 3. Immune Checkpoint Inhibitors in Melanoma Care: ManagementofImmune-MediatedAdverseReactions1 ICPi: immune-checkpoint inhibitor; IMAR: immune-mediated adverse reaction. 1. Brahmer JR et al. J Oncol Pract. 2018;14:247-249. PRACTICE AID Access the activity,“Tracing the Arc of Immunotherapy in Melanoma: Insights on the Current Role and Cutting-Edge Uses of Checkpoint Inhibitors for Patient Care,”at www.peerview.com/KUZ40. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Educate patients and caregivers on immunotherapies, their MOA, and any potential IMARs prior to and throughout treatment Hepatic (autoimmune hepatitis ALT/AST increases) Endocrine (hypophysitis, thyroiditis, type 1 diabetes) Respiratory (pneumonitis) Renal (nephritis, renal failure) Neuromuscular (peripheral sensory neuropathy; rarely) Gastrointestinal (colitis/diarrhea) Skin (maculopapular rash, pruritus) Continue ICPi therapy with close monitoring in most cases Instruct patients to report any new symptoms, as they could be IMARs and may require immediate management Talking Points Overview of IMARs General Guidelines: Management by Grade 1 Hold ICPis and consider resuming when symptoms/laboratory values grade ≤1; low-dose corticosteroids may be administered 2 Hold ICPis and initiate high-dose corticosteroids; consider infliximab if symptoms do not improve with 48-72 h of corticosteroid treatment 3 Permanently discontinue ICPis in most cases 4
