The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons
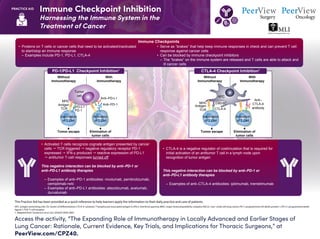
- 1. Immune Checkpoint Inhibition Harnessing the Immune System in the Treatment of Cancer Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. APC: antigen-presenting cell; CD: cluster of differentiation; CTLA-4: cytotoxic T-lymphocyte associated antigen 4; IFN-γ: interferon gamma; MHC: major histocompatibility complex; NSCLC: non–small cell lung cancer; PD-1: programmed cell death protein 1; PD-L1: programmed death ligand 1; TCR: T-cell receptor. 1. Adapted from: Soularue E et al. Gut. 2018;67:2056-2067. • Proteins on T cells or cancer cells that need to be activated/inactivated to start/stop an immune response – Examples include PD-1, PD-L1, CTLA-4 PD-1/PD-L1 Checkpoint Inhibition1 Without Immunotherapy With Immunotherapy MHC Antigen TCR PD-1 PD-L1 Anti–PD-L1 Anti–PD-1 Tumor cell Tumor escape Inactivation of T Cell Activation of T Cell Elimination of tumor cells CTLA-4 Checkpoint Inhibition1 Without Immunotherapy With Immunotherapy MHC CD80/86 CTLA-4 Anti– CTLA-4 antibody APC Antigen TCR Inactivation of T Cell Activation of T Cell Tumor escape Elimination of tumor cells • Serve as “brakes” that help keep immune responses in check and can prevent T cell response against cancer cells • Can be blocked by immune checkpoint inhibitors – The “brakes” on the immune system are released and T cells are able to attack and ill cancer cells Immune Checkpoints • Activated T cells recognize cognate antigen presented by cancer cells TCR triggered negative regulatory receptor PD-1 expressed IFN-γ produced reactive expression of PD-L1 antitumor T-cell responses turned off This negative interaction can be blocked by anti–PD-1 or anti–PD-L1 antibody therapies – Examples of anti–PD-1 antibodies: nivolumab, pembrolizumab, cemiplimab-rwlc – Examples of anti–PD-L1 antibodies: atezolizumab, avelumab, durvalumab • CTLA-4 is a negative regulator of costimulation that is required for initial activation of an antitumor T cell in a lymph node upon recognition of tumor antigen This negative interaction can be blocked by anti–PD-1 or anti–PD-L1 antibody therapies – Examples of anti–CTLA-4 antibodies: ipilimumab, tremelimumab
- 2. Current and Emerging Roles of Immunotherapy in NSCLC1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID Timing, Dose, and Duration of Durvalumab Treatment Dosing: 10 mg/kg IV infusion over 60 min Q2W Continue Q2W until unacceptable toxicity, disease progression, or a max of 12 mo Should be initiated within 42 days post-cCRT • Starting treatment within 14 days post-cCRT was associated with the best outcomes A Multimodal Treatment Approach Involving Surgery, Radiation, Chemotherapy, and Immunotherapy • Combining and/or sequencing PD-1/PD-L1 checkpoint inhibitors with chemotherapy and radiotherapy can produce synergistic treatment responses across the NSCLC disease spectrum • It is not yet known how, when, and where to integrate PD-1/PD-L1 checkpoint inhibitors into the treatment regimen to maximally benefit patients • It appears likely that optimal strategies for combining and sequencing vary depending on whether or not the disease is resectable • Significant unmet need in locally advanced NSCLC for novel therapies to improve outcomes beyond those achieved with cCRT • Durvalumab significantly improved PFS and OS vs placebo in PACIFIC → first major advance in decades for unresectable stage III NSCLC Indications Patients must have a CT scan to confirm no PD before initiating durvalumab Durvalumab is an anti–PD-L1 checkpoint inhibitor FDA approved and indicated for patients with unresectable stage III NSCLC whose disease has not progressed following cCRT Patients should NOT receive durvalumab if they are X Under 18 years of age X Being treated for an autoimmune disease or have received an organ transplant X Pregnant, planning to become pregnant, or breastfeeding Patients may be treated regardless of PD-L1 expression or EGFR mutation status
- 3. Current and Emerging Roles of Immunotherapy in NSCLC1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID Rationale for Neoadjuvant Immunotherapy in Resectable, Early Disease Neoadjuvant treatment with PD-1/PD-L1 CPIs before tumor resection may provide several benefits that improve prognosis in patients with early-stage, resectable NSCLC • Reduction of tumor size to allow for a potentially less morbid resection • Ability of patients to tolerate therapy better before surgery compared with afterward • Earlier eradication of nodal and micrometastatic disease • Introducing immunotherapy while the primary tumor is still in the body facilitates expansion and activation of tumor-specific T cells that engage in systemic surveillance to identify and attack future micrometastases • Availability of pre- and post-treatment specimens provides opportunity to rapidly assess treatment efficacy/pathologic response • Opportunity to evaluate potential biomarkers for response and toxicity • Several ongoing trials combine neoadjuvant CPI treatment with other therapies or strategies in an attempt to maximize the treatment response Selected Trials of Adjuvant Immunotherapy for NSCLC PEARLS (NCT02504372): Phase 3 IMpower010 (NCT02486718): Phase 3 Atezolizumab BSCvs 1° endpoint: DFS Active, Not Recruiting • Complete resection (stage IB-IIIA) • Resection 4-12 weeks prior to enrollment • Cisplatin eligible • Planned N = 1,280 Pembrolizumab Placebovs 1° endpoint: OS, LCSS Recruiting • Complete resection (stage IB-IIIA) • Specimen for PD-L1 expression testing • Any histology • Patients with 2 synchronous primary NSCLC cancers excluded • Planned N = 1,080 ANVIL (NCT02595944): Phase 3 Nivolumab Observationvs 1° endpoint: DFS, OS Recruiting • Complete resection (stage IB-IIIA) • CT ≤1 mo since randomization to confirm no PD • No prior checkpoint inhibitor treatment • Planned N = 903 BR31 (NCT02273375): Phase 3 PEARLS (NCT02504372): Phase 3 Durvalumab Placebovs 1° endpoint: DFS Recruiting • Complete resection (stage IB-IIIA) • No neoadjuvant chemotherapy • Prior postoperative adjuvant chemotherapy is permissible • Planned N = 1,360
- 4. Current and Emerging Roles of Immunotherapy in NSCLC1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID Selected Trials of Neoadjuvant Immunotherapy for NSCLC PEARLS (NCT02504372): Phase 3 J1772 (NCT03237377): Phase 2 Durvalumab ± tremelimumab + RT 1° endpoint: safety Active, Not Recruiting • Stage IIIA • No prior checkpoint inhibitor treatment or anticancer vaccine • Planned N = 32 Nivolumab ± ipilimumab 1° endpoint: safety Recruiting • Squamous or nonsquamous stage IB-IIIA • No preoperative chemotherapy or any other cancer therapy • CT or MRI prior to enrollment to confirm no brain metastases • Planned N = 30 AAAQ3153 (NCT02716038): Phase 2 Atezolizumab + nab-paclitaxel + carboplatin Historical chemotherapy responses vs 1° endpoint: MPR Recruiting • Squamous or nonsquamous stage IB-IIIA • Specimen for PD-L1 expression testing • Planned N = 30 J1414 (NCT02259621): Phase 2CheckMate -816 (NCT02998528): Phase 3 Nivolumab + platinum doublet or ipilimumab Platinum doublet chemotherapy vs 1° endpoint: OS, MPR, TTDM Recruiting • Stage IB-IIIA • Available primary lung tumor tissue • No prior checkpoint inhibitor treatment • Planned N = 350 Pembrolizumab 1° endpoint: safety, tumor response, pathologic response Recruiting • Stage II-IIIA • No anticancer treatment within 30 days prior to enrollment, including systemic therapy, RT, or major surgery • Planned N = 30 NEOMUN (NCT03197467): Phase 2
- 5. Current and Emerging Roles of Immunotherapy in NSCLC1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. BSC: best supportive care; cCRT: concurrent chemoradiotherapy; CPI: checkpoint inhibitor; CT: computed tomography; DFS: disease-free survival; EFS: event-free survival; EGFR: epidermal growth factor receptor; LCSS: lung cancer–specific survival; MPR: major pathologic response; MRI: magnetic resonance imaging; NSCLC: non–small cell lung cancer; PD: progressive disease; PD-1: programmed death protein 1; PD-L1: programmed death ligand 1; Q2W: every 2 weeks; RT: radiation therapy; SBRT: stereotactic body radiation therapy; TTDM: time to death or distant metastases. 1. Durvalumab (Imfinzi) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761069s002lbl.pdf. Accessed April 17, 2019. 2. Carbone DP et al. J Thorac Oncol. 2015;10:974-984. 3. Owen D et al. J Thoracic Dis. 2018;10:S404-S411. 4. https://clinicaltrials.gov. Accessed April 17, 2019. Selected Trials of Neoadjuvant + Adjuvant Immunotherapy for NSCLC LCMC3 (NCT02927301): Phase 2 Atezolizumab 1° endpoint: MPR Recruiting • Stage IB-IIIA or selected IIIB • Adequate primary tumor biopsy tissue • No prior therapy for lung cancer • Planned N = 180 J1414 (NCT02904954): Phase 2 Durvalumab ± SBRT 1° endpoint: DFS Recruiting • Stage I (>2 cm), II, and IIIA • No prior therapy for lung cancer • Planned N = 60 SAKK 16/14 (NCT02572843): Phase 2 Durvalumab 1° endpoint: PFS Active, Not Recruiting • Stage T1-3N2M0, stage IIIA (N2) • NSCLC irrespective of genomic aberrations or PD-L1 expression status • Planned N = 68 TOP 1501 (NCT02818920): Phase 2 Pembrolizumab 1° endpoint surgical feasibility Active, Not Recruiting • Stage IB (≥3 cm), IIA/IIB, or IIIA • No prior therapy for lung cancer • Planned N = 32 GECP 16/03_NADIM (NCT03081689): Phase 2 Nivolumab + paclitaxel + carboplatin 1° endpoint: PFS Active, Not Recruiting • Stage IIIA • No EGFR or ALK mutation • Planned N = 46 IMpower030 (NCT03456063): Phase 3 Atezolizumab + platinum-based chemo 1° endpoint: MPR, EFS Recruiting • Squamous or nonsquamous stage II-IIIB (T3N2 only) • No prior therapy for lung cancer • No EGFR or ALK mutation • Planned N = 374 Placebo + platinum-based chemo vs KEYNOTE-671 (NCT03425643): Phase 3 Pembrolizumab + platinum doublet chemo 1° endpoint: EFS, OS Recruiting • Stage IIB or IIIA • No prior therapy for lung cancer • Planned N = 786 Placebo + platinum doublet chemo vs
- 6. Guidance for Immune-Related Adverse Effects Associated With Immunotherapy1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID What Are irAEs? • Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of immune-related adverse effects • Any organ system can be affected, but more commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs Nervous system Cardiovascular Pulmonary Gastrointestinal Musculoskeletal Ocular Endocrine Dermatologic Hematologic Renal
- 7. Guidance for Immune-Related Adverse Effects Associated With Immunotherapy1-4 Access the activity, “The Expanding Role of Immunotherapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Current Evidence, Key Trials, and Implications for Thoracic Surgeons,” at PeerView.com/CPZ40. PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. a For irAE guidelines, please visit: https://www.asco.org/practice-guidelines/quality-guidelines/guidelines/supportive-careand-treatment-related-issues#/29866. AE: adverse effect; irAE: immune-related adverse effect. 1. Brahmer JR et al. J Clin Oncol. 2018;36:1714-1786. 2. Calabrese LH et al. Nat Rev Rheumatol. 2018;14:569-579. 3. Durvalumab (Imfinzi) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761069s002lbl.pdf. Accessed April 17, 2019. 4. Postow MA et al. N Engl J Med. 2018;378:158-168. How Should irAEs be Diagnosed and Managed? Minimal or No Symptoms; Diagnostic Changes Only • In general, immunotherapy should be continued with close monitoring, with the exception of some neurologic, hematologic, and cardiac toxicities Mild to Moderate Symptoms • Hold checkpoint inhibitor therapy for most grade 2 toxicities • Consider resuming immunotherapy when symptoms and/or lab values revert to grade 1 • Corticosteroids (initial dose of 0.5-1 mg/kg/day of prednisone or equivalent) may be administered Severe or Life-Threatening Symptoms Grade 3 toxicities • Hold checkpoint inhibitor therapy • Initiate high-dose corticosteroids (prednisone 1-2 mg/kg/day or methylprednisolone IV 1-2 mg/kg/day) • If symptoms do not improve with 48-72 hours of high-dose corticosteroid, infliximab may be offered for some toxicities • Taper corticosteroids over the course of at least 4-6 weeks • When symptoms and/or laboratory values revert to grade 1, rechallenging with immunotherapy may be considered; however, caution is advised, especially in those patients with early-onset irAEs; dose adjustments are not recommended Grade 4 toxicities • In general, permanent discontinuation of checkpoint inhibitor therapy is warranted, with the exception of endocrinopathies that have been controlled by hormone replacement irAEs are often diagnosed by exclusion; other causes should be ruled out (including AEs of other therapies used), but immunotherapy-related toxicity should always be included in the differential There should be a high level of suspicion that new symptoms are treatment related; early recognition, evaluation, and treatment of irAEs plus patient education are essential for the best outcome Depending on severity of irAE, management may require corticosteroid or other immunosuppressive treatment and interruption or discontinuation of therapy If appropriate immunosuppressive treatment is used, patients generally recover from irAEs Use of immunosuppressive therapy to manage irAEs does not appear to impact response to immunotherapy Grade 1 Grade 2 Grade 3/4 Additional resources are available on the ASCO website:a www.asco.org
