Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM
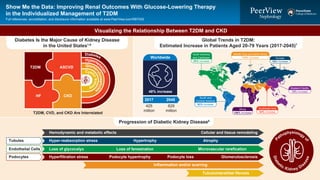
- 1. Diabetes Hyperten sion Cyst ickidn ey Glome rulonephritis Visualizing the Relationship Between T2DM and CKD Global Trends in T2DM: Estimated Increase in Patients Aged 20-79 Years (2017-2045)7 48% increase 2017 Diabetes Is the Major Cause of Kidney Disease in the United States1-6 T2DM, CVD, and CKD Are Interrelated T2DM ASCVD HF CKD 425 million 629 million North America and Caribbean 35% increase South and Central America 62% increase Middle East and North Africa 110% increase Africa 156% increase Southeast Asia 84% increase Western Pacific 15% increase Europe 16% increase 2045 Worldwide Progression of Diabetic Kidney Disease8 P athophysiology of Diabe tic Kidney Dis ease Hemodynamic and metabolic effects Hyper-reabsorption stress Hypertrophy Atrophy Loss of glycocalyx Loss of fenestration Microvascular rarefication Hyperfiltration stress Tubules Endothelial Cells Podocytes Podocyte hypertrophy Podocyte loss Inflammation and/or scarring Tubulointerstitial fibrosis Glomerulosclerosis Cellular and tissue remodeling Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full references, accreditation, and disclosure information available at www.PeerView.com/RBT930
- 2. Exploring the Science: Improving Renal Outcomes in Patients With T2DM Multidimensional Effects at GFR >60 mL/min/1.73 m2 14-15 Outcome Trials With SGLT2 Inhibitors16-28 ADA Standards of Medical Care in Diabetes9 FDA-Approved SGLT2 Inhibitors10-13 Cardiovascular CKD Staging Albuminuria Stages, Range (mg/g), and Description A1: <30 Normoalbuminuria ≥90 (Stage 1) 60-90 (Stage 2) 45-59 (Stage 3a) GFRCategories,mL/min/1.73m2 30-44 (Stage 3b) 15-29 (Stage 4) <15 (ESKD 6) A2: 30-300 Microalbuminuria A3: >300 Macroalbuminuria Renal CANVAS CREDENCE DECLARE-TIMI 58 DAPA-CKD EMPA-REG OUTCOME EMPA-KIDNEY VERTIS CV VERTIS MET and SU Canagliflozin Dapagliflozin Empagliflozin Ertugliflozin SGLT2 inhibition Cardiac and renal protection Negative caloric balance ↓ A1C ↑ Uricosuria ↓ Blood pressure ↓ Plasma volume ↑ Tubuloglo- merular feedback ↓ Total body fat mass ↓ Inflammation ↓ Glucose toxicity ↓ Plasma uric acid ↓ Arterial stiffness ↓ Myocardial stretch Afferent arteriole constriction Glycosuria Natriuresis C D E V C D E V eGFR ≥30-<90 mL/min/1.73 m2 and UACR ≥300 mg/g eGFR ≥25-<75 mL/min/1.73 m2 and UACR ≥200 mg/g eGFR ≥45-<75 mL/min/1.73 m2 and UACR ≥200 mg/g or eGFR ≥20-<45 mL/min/1.73 m2 Post-hoc analysis; mean (SD) BL eGFR 88.2 mL/min/1.73 m2 and BL UACR 1.31 mg/mmol ↓ Epicardial fat ↓ Atherosclerosis ↓ Ventricular arrhythmias Activation of ACE2-Ang1/7 ↓ Inflammation ↓ Fibrosis ↑ Cardiac contractility ↓ Intraglomerular hypertension ↓ Hyperfiltration Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full references, accreditation, and disclosure information available at www.PeerView.com/RBT930
- 3. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Brenner B et al. N Engl J Med. 2001;345:861-869. 2. Lewis EJ et al. N Eng J Med. 2001;345:851-860. 3. Zelniker TA et al. Circulation. 2019;139:2022-2031. 50 40 30 20 10 0 0.6 0.5 0.4 0.3 0.2 0.1 0 0 12 24 36 48 Placebo Losartan RESIDUAL RISK RENAAL Risk reduction: 16% P = .02 Time of Study, mo Follow-Up, mo PrimaryCompositeEndpoint,% Risk reduction: 20% P = .02 Irbesartan Amlodipine Placebo PrimaryCompositeEndpoint,% IDNT 0 6 12 18 24 30 36 42 48 54 RESIDUAL RISK Doubling of Serum Creatinine, ESKD, or Death1,2 SGLT2i CVOTs: Meta-Analysis3 a Meta-analysis of three CVOTs with a total of 34,322 participants (EMPA-REG OUTCOME, CANVAS, DECLARE). 0 -10 -20 -30 -40 -50 RelativeRisk Reduction,%a HR (95% CI) P 0.89 (0.83-0.96) .001 0.84 (0.75-0.94) .002 0.89 (0.80-0.98) .018 0.97 (0.86-1.10) NS 0.69 (0.61-0.79) <.001 0.62 (0.58-0.67) <.001 0.55 (0.48-0.64) <.001 -11% -16% -11% -3% -31% -38% -45% MACE CV Death Fatal or Nonfatal MI Fatal or Nonfatal Stroke HHF Pure Renal Endpoints Worsening of Kidney Function
- 4. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Jardine MJ et al. Am J Nephrol. 2017;46:462-472. 2. Perkovic V et al. N Engl J Med. 2019;380:2295-2306. 3. Invokana (canagliflozin) Prescribing Information. http://www.janssenlabels.com/package- insert/product-monograph/prescribing-information/INVOKANA-pi.pdf. 4. Jardine MJ et al. J Am Soc Nephrol. 2020;31:1128-1139. CREDENCE Study Design The CREDENCE Study1-4 Inclusion Criteria ü ≥30 years ü A1C ≥6.5% and ≤12.0% ü eGFR ≥30 and <90 mL/min/1.73 m2 ü UACR >300 and ≤5,000 mg/g • Canagliflozin 100 mg • Placebo • 1:1 randomization, double-blind • 2.62 years (median) • Event-driven (405 events) • Composite renal endpoint • ESKD • Doubling of sCr • Renal death • CV death û Dialysis û Kidney transplant û Recent CV event û NYHA class IV û HF Exclusion Criteria Interventions Follow-Up Primary Outcome Population: • Multicenter (34 countries); target N = 4,401 • Patients with T2DM and albuminuria using ACEi or ARB -20 -18 -16 -14 -12 -10 -8 -6 -4 -2 0 Least-SquareMeanChange, mL/min/1.73m2 Time Since Randomization, mo 0 200 400 600 800 1,000 1,200 0 6 12 18 24 30 36 '" GeometricMean Time Since Randomization, mo UACR Median Baseline Canagliflozin 913.5 Placebo 918.0 Baseline, mL/min/1.73 m2 Canagliflozin 56.4 Placebo 56.0 Placebo Canagliflozin Canagliflozin Placebo No. of Patients Placebo 2,113 2,061 1,986 1,865 1,714 1,158 685 251 Canagliflozin 2,114 2,070 2,019 1,917 1,819 1,245 730 271 0 6 12 18 24 30 36 42 No. of Patients Placebo 2,178 1,985 1,882 1,720 1,536 1,006 583 210 Canagliflozin 2,179 2,005 1,919 1,782 1,648 1,116 652 241 Change From Baseline in eGFR 32% reduction
- 5. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Jardine MJ et al. Am J Nephrol. 2017;46:462-472. 2. Perkovic V et al. N Engl J Med. 2019;380:2295-2306. 3. Invokana (canagliflozin) Prescribing Information. http://www.janssenlabels.com/package- insert/product-monograph/prescribing-information/INVOKANA-pi.pdf. 4. Jardine MJ et al. J Am Soc Nephrol. 2020;31:1128-1139. The CREDENCE Study1-4 Renal-Specific Composite Outcome of ESKD, Doubling of Creatinine, or Renal Deatha a Indicated to reduce the risk of ESKD, doubling of serum creatinine, CV death, and hospitalization for HF in adults with T2DM and diabetic nephropathy with albuminuria. PatientsWithanEvent,% Time Since Randomization, mo 0 5 10 15 20 0 6 12 18 24 30 36 42 HR = 0.66 (95% Cl, 0.53-0.81) P < .001 Placebo Canagliflozin No. at Risk Placebo 2,199 2,178 2,131 2,046 1,724 1,129 621 170 Canagliflozin 2,202 2,181 2,144 2,080 1,786 1,211 646 196 34% risk reduction Effects of Canagliflozin on eGFR Slope by Screening eGFR 0 10 20 30 40 50 60 70 80 90 0 6 12 18 24 30 36 42 AdjustedMean(SE)eGFR, mL/min/1.73m2 Time Since Randomization, mo Screening eGFR 60-<90 mL/min/1.73 m2 Screening eGFR 45-<60 mL/min/1.73 m2 Canagliflozin Placebo Screening eGFR 30-<45 mL/min/1.73 m2
- 6. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Jardine MJ et al. J Am Soc Nephrol. 2020;31:1128-1139. 2. Invokana (canagliflozin) Prescribing Information. http://www.janssenlabels.com/package-insert/product-monograph/prescribing- information/INVOKANA-pi.pdf. CREDENCE: AEs Stratified by Degree of Renal Impairment1 FDA Label Update: Canagliflozin2 a Based on confirmed and adjudicated results. Screening eGFR, mL/min/1.73 m2 HR (95% CI) P HHF All 30-<45 45-<60 60-<90 0.61 (0.47-0.80) 0.70 (0.46-1.06) 0.43 (0.26-0.72) 0.72 (0.44-1.16) .27 All SAEs All 30-<45 45-<60 60-<90 0.87 (0.79-0.97) 0.85 (0.71-1.00) 0.78 (0.65-0.93) 0.99 (0.84-1.17) .15 Fracturea All 30-<45 45-<60 60-<90 0.98 (0.70-1.37) 1.04 (0.58-1.86) 0.82 (0.44-1.50) 1.08 (0.61-1.91) .77 Amputation All 30-<45 45-<60 60-<90 1.11 (0.79-1.56) 1.36 (0.73-2.54) 0.64 (0.33-1.22) 1.40 (0.82-2.39) .14 Favors canagliflozin Favors placebo 0.25 0.5 1.0 2.0 4.0 1. Indications and usage updated • Adults with T2DM and eGFR ≥30 mL/min/1.73 m2 , with or without albuminuria, may start canagliflozin 100 mg Continuation of canagliflozin is not recommended in patients with eGFR <30 mL/min/1.73 m2 unless albuminuria is >300 mg/d 2. Boxed warnings removed • Lower extremity amputations 3. Warnings and precautions removed • AKI as a stand-alone warning • Warning that symptomatic hypotension can occur in patients on ACEi/ARBs FDA Label Update: Canagliflozin
- 7. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Invokana (canagliflozin) Prescribing Information. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/INVOKANA-pi.pdf. 2. Farxiga (dapagliflozin) Prescribing Information. https://www.azpicentral.com/farxiga/farxiga.pdf#page=1. 3. Jardiance (empagliflozin) Prescribing Information. https://docs.boehringer-ingelheim.com/Prescribing%20 Information/PIs/Jardiance/jardiance.pdf. 4. Steglatro (ertugliflozin) Prescribing Information. https://www.merck.com/product/usa/pi_circulars/s/steglatro/steglatro_pi.pdf. 5. Dekkers CCJ et al. Nephrol Dial Transplant. 2020;35:i33-i42. SGLT2i Prescribing Information: Contraindications, Warnings, and Precautions1-4 CANVAS, DECLARE, EMPA-REG, and CREDENCE: Renal Outcomes by eGFR Stratum5 a eGFR <30 mL/min/1.73 m2 in patients who are being treated for glycemic control without established CVD or CV risk factors. Contraindications Do not use in patients with a history of hypersensitivity to this agent Severe renal impairment, ESKD, or dialysis Warnings/Precautions Lower limb amputation in patients with CVD, limb infections, or leg ulcers Hypotension, volume depletion Ketoacidosis AKI or renal impairment Urosepsis, pyelonephritis Hypoglycemia risk increases when used with insulin or SU Fournier’s gangrene Genital mycotic infections DAPA X Xa DAPA X X X X X X Increased LDL-C Hypersensitivity reactions Bone fracture CANA X X CANA X X X X X X X X X EMPA X X EMPA X X X X X X X X X ERTU X X ERTU X X X X X X X X X SGLT2i, n/N Trials Placebo, n/N Estimated NNT per 5 y Renal outcomes per eGFR stratum, mL/min/1.732 eGFR <45 21/374 14/189 33 Canagliflozin (CANVAS) NA NA 41 Canagliflozin (CREDENCE) 122/678 166/687 8 13 21/606 38/659 31 Empagliflozin (EMPA-REG) 24/822 26/416 18 Canagliflozin (CANVAS) NA NA 61 Canagliflozin (CREDENCE) 58/630 97/636 8 30 Dapagliflozin (DECLARE) 65/3,838 121/3,894 58 Empagliflozin (EMPA-REG) 37/2,406 29/1,232 72 Canagliflozin (CANVAS) NA NA 71 Canagliflozin (CREDENCE) 65/893 77/876 38 Subgroup mean (I2 = 11.7%) 62 Dapagliflozin (DECLARE) 41/4,137 79/4,025 90 Empagliflozin (EMPA-REG) 18/1,043 17/486 39 Canagliflozin (CANVAS) NA NA 47 Subgroup mean (I2 = 0.0%) 79 0.4 0.6 0.8 1.0 1.4 1.61.2 SGLT2i better Placebo better HR (95% Cl) 0.70 (0.36-1.39) 0.65 (0.29-1.48) 0.71 (0.56-1.89) 0.70 (0.57-0.88) 0.60 (0.35-1.02) 0.42 (0.24-0.73) 0.78 (0.46-1.13) 0.59 (0.43-0.82) 0.60 (0.48-0.74) 0.54 (0.40-0.73) 0.62 (0.38-1.01) 0.58 (0.41-0.84) 0.81 (0.58-1.13) 0.63 (0.52-0.76) 0.50 (0.34-0.73) 0.49 (0.25-0.95) 0.44 (0.25-0.78) 0.48 (0.36-0.64) Empagliflozin (EMPA-REG) Subgroup mean (I2 = 0.0%) eGFR 45-60 eGFR 60-90 eGFR >90 Dapagliflozin (DECLARE) Subgroup mean (I2 = 0.0%)
- 8. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Cherney D et al. Diabetologia. 2020;63:1128-1140. 2. Silverthorn DU. Human Physiology: An Integrated Approach. 5th ed. Prentice Hall; 2009. LSM Changes From Baseline in eGFR1 SGLT2 Inhibitors and Turboglomerular Feedback2 3.0 2.0 1.0 0 -1.0 -2.0 -3.0 -4.0 -5.0 6 12 18 26 39 52 65 78 91 104 LSMChangeFromBaseline ineGFR,mL/min/1.73m2 LSMChangeFromBaseline ineGFR,mL/min/1.73m2 Time, wk 3.0 2.0 1.0 0 -1.0 -2.0 -3.0 -4.0 -5.0 6 12 18 26 39 52 65 78 91 104 Patients With Baseline UACR <3.39 mg/mmol Patients With Baseline UACR ≥3.39 mg/mmol BL BL Non-ertugliflozin Ertugliflozin 5 mg Ertugliflozin 15 mg Ertugliflozin 5 mg Time, wk Patients, n Non-ertugliflozin 489 490 502Ertugliflozin 15 mg 477 475 487 461 467 473 451 464 459 442 436 398 468 426 395 462 444 413 385 358 382 353 341 332 316 408 373 312 359 336 Ertugliflozin 5 mg Patients, n Non-ertugliflozin 142 147 126Ertugliflozin 15 mg 140 143 118 136 136 114 134 136 131 134 128 119 112 132 122 111 109 101 114 109 112 106 100 104 95 98 91 97 85 336 1 2 3 4 5 2 3 4 5 GFR increases Flow through tubule increases Flow past macula densa increases Paracrine from macula densa to afferent arteriole Afferent arteriole constricts 1 Efferent arteriole Glomerulus Distal tubule Collecting Proximal tubule duct Loop of Henle Bowman’s capsule Macula densa Granular cells Afferent arteriole 25 Seconds <40 >45 GFR
- 9. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. American Diabetes Association. Diabetes Care. 2020;43(suppl 1):S1-S212. ADA Treatment Algorithm1 First-line therapy is metformin and comprehensive lifestyle modifications (including weight management and physical activity) Consider independently of baseline A1C or individualized A1C target ASCVD predominates If A1C above target HF or CKD predominates If A1C above target Indicators of high risk or established ASCVD, CKD, or HF Compelling need to minimize hypoglycemia If A1C above target If A1C above target If A1C above target If A1C above target If A1C above target Cost is a major issue If A1C above target If A1C above target To avoid therapeutic inertia, reassess and modify treatment regularlyNO • Established ASCVD • Indicators of high ASCVD risk (age >55 years with coronary, carotid, or lower extremity artery stenosis >50% or LVH) Preferably GLP-1 RA with proven CVD benefit or SGLT2i with proven CVD benefit if eGFR adequate If further intensification is required or patient is now unable to tolerate GLP-1 RA and/or SGLT2i, choose agents demonstrating CV safety: • For patients on GLP-1 RA, considering adding SGLT2i with proven CVD benefit; DPP-4i if not on GLP-1 RA; basal insulin; TZD; or SU • Particularly HFrEF (LVEF <45%) • CKD: specifically eGFR 30-60 mL/min/1.73 m2 or UACR >30 mg/g, particularly UACR >300 mg/g Preferably SGLT2i with evidence of reducing HF/CKD progression in CVOTs if eGFR adequate or If SGLT2i not tolerated/ contraindicated or if eGFR less than adequate add GLP-1 RA with proven CVD benefit Avoid TZD in the setting of HF; choose agents demonstrating CV safety: for patients on SGLT2i, consider adding GLP-1 RA with proven CVD benefit; DPP-4i (not saxagliptin) in the setting of HF (not on GLP-1 RA); basal insulin; or SU DPP-4i GLP-1 RA SGLT2i TZD SGLT2i or TZD SGLT2i or TZD GLP-1 RA or DPP-4i or TZD SGLT2i or DPP-4i or GLP-1 RA Continue with addition of other agents as outlined above Consider the addition of SU or basal insulin: • Choose later-generation SU with lower risk of hypoglycemia • Consider basal insulin with lower risk of hypoglycemia GLP-1 RA with good efficacy for weight loss SGLT2i SGLT2i GLP-1 RA with good efficacy for weight loss If quadruple therapy required, or SGLT2i and/or GLP-1 RA not tolerated/ contraindicated, use regimen with lowest risk of weight gain Preferably DPP-4i (if not on GLP-1 RAs) based on weight neutrality If DPP-4i not tolerated/contraindicated or patient already on GLP-1 RA, cautious addition of SU, TZD, or basal insulin SU TZD TZD SU • Insulin therapy: basal insulin with lowest acquisition cost or • Consider DPP-4i or SGLT2i with lowest acquisition cost YES Compelling need to minimize weight gain or promote weight loss If A1C above individualized target proceed as below
- 10. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Heerspink HJL et al. Nephrol Dial Transplant. 2020;35:274-282. 2. Wheeler DC et al. Nephrol Dial Transplant. 2020 Aug 30 [Epub ahead of print]. 3. Heerspink HJL et al. Presented at European Society of Cardiology Congress 2020 (ESC 2020). The DAPA-CKD Trial1-3 DAPA-CKD Study Design Inclusion Criteria ü ≥18 years ü eGFR ≥25-75 mL/min/1.73 m2 ü UACR 200 to 5,000 mg/g ü Stable MTD of ACEi or ARB for ≥4 weeks (if not contraindicated) ü Multicenter (~400 sites); target N = 4,300 ü Patients with and without T2DM • Dapagliflozin 10 mg • Placebo • 1:1 randomization • ~45 months (median) • Event-driven (681 events) • ≥50% decline in eGFR • ESKD • Renal or CV death û Polycystic kidney disease û Lupus nephritis û ANCA vasculitis û T1DM Exclusion Criteria Interventions Follow-Up Primary Outcome Stopped Early for Efficacy Primary Outcome Sustained ≥50% eGFR Decline, ESKD, Renal or CV Death 0 0 2 4 6 8 10 12 14 16 18 20 22 24 4 8 12 16 Time Since Randomization, mo Hazard ratio, 0.61 (95% CI, 0.51-0.72) P = .000000028 NNT = 19 Placebo Dapagliflozin 197 events 312 events CumulativeIncidence,% 20 24 28 32 2,152 2,001 1,955 1,898 1,841 1,701 1,288 831 309 2,152 Dapagliflozin No. at Risk Placebo 1,993 1,936 1,858 1,791 1,664 1,232 774 270 2,152 2,001 1,955 1,898 1,841 1,701 1,288 831 309 2,152 Dapagliflozin No. at Risk Placebo 1,993 1,936 1,858 1,791 1,664 1,232 774 270 Secondary Outcome Sustained ≥50% eGFR Decline, ESKD, Renal Death 0 0 2 4 6 8 10 12 14 16 18 20 22 24 4 8 12 16 Time Since Randomization, mo Hazard ratio, 0.56 (95% CI, 0.45-68) P = .000000018 Placebo Dapagliflozin 142 events 243 events CumulativeIncidence,% 20 24 28 32
- 11. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. Heerspink HJL et al. Nephrol Dial Transplant. 2020;35:274-282. 2. Wheeler DC et al. Nephrol Dial Transplant. 2020 Aug 30 [Epub ahead of print]. 3. Heerspink HJL et al. Presented at European Society of Cardiology Congress 2020 (ESC 2020). The DAPA-CKD Trial1-3 Summary of the Primary Outcome and Its Components Placebo events Well tolerated; DKA was not reported in any patients receiving DAPA (but in 2 receiving PBO) Hazard Ratio (95% CI) P Dapagliflozin events NC83Transplantation NC62Renal death Favors dapagliflozin Hazard Ratio (95% CI) Favors placebo 0.3 0.6 1.0 1.4 .20290.81 (0.58-1.12)8065CV death .00800.66 (0.48-0.90)9968Chronic dialysis .00450.67 (0.51-0.88)12084eGFR <15 mL/min/1.73 m2 .00040.64 (0.50-0.82)161109ESKD <.00010.53 (0.42-0.67)201112≥50% eGFR decline .0000000280.61 (0.51-0.72)312192Primary composite endpoint
- 12. Show Me the Data: Improving Renal Outcomes With Glucose-Lowering Therapy in the Individualized Management of T2DM Full abbreviations, accreditation, and disclosure information available at PeerView.com/RBT930 1. https://clinicaltrials.gov/ct2/show/NCT03594110. EMPA-KIDNEY Study Design1 Inclusion Criteria ü ≥18 years ü eGFR ≥25-<45 mL/min/1.73 m2 or ≥45-<90 mL/min/1.73 m2 and UACR ≥200 mg/g (or protein:creatinine ≥300 mg/d) ü Multicenter (~7 countries); target N = 6,000 ü Patients with and without T2DM using an ACEi or ARB ü Estimated completion date is June 2022 • Empagliflozin • Placebo • 1:1 randomization, double-blind • ~3.1 years (median) • Renal endpoint – Sustained ↓eGFR to <10 mL/min/ 1.73 m2 – Sustained decline of ≥40% in eGFR from randomization – ESKD – Renal death -- or -- • CV death û Polycystic kidney disease û T2DM and ASCVD with eGFR >60 mL/min/1.73 m2 û Bariatric surgery Exclusion Criteria Interventions Follow-Up Primary Outcome Currently Recruiting
