Papilomavirus.ppt
•Download as PPT, PDF•
0 likes•3 views
Presentación que resalta datos importantes relacionados con el agente causal del cancer causado por papilomavirus. Mostrando los procesos de replicación del virus, la patogénesis del patógeno.
Report
Share
Report
Share
Recommended
Recommended
More Related Content
Similar to Papilomavirus.ppt
Similar to Papilomavirus.ppt (20)
The effects of circumcision on the penis microbiome

The effects of circumcision on the penis microbiome
Introduction to Viruses of Medical Importance-1.ppt

Introduction to Viruses of Medical Importance-1.ppt
Analysis in to the Epidemiology and Pathophysiology of Respiratory Syncytial ...

Analysis in to the Epidemiology and Pathophysiology of Respiratory Syncytial ...
Molecular localization of epstein barr virus and rb tumor suppressor gene exp...

Molecular localization of epstein barr virus and rb tumor suppressor gene exp...
molecular mechanism of viral diseases and biotechnological interventions for ...

molecular mechanism of viral diseases and biotechnological interventions for ...
More from LibardoCaraballo1
More from LibardoCaraballo1 (7)
Recently uploaded
Recently uploaded (20)
HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx

HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx
dusjagr & nano talk on open tools for agriculture research and learning

dusjagr & nano talk on open tools for agriculture research and learning
Sensory_Experience_and_Emotional_Resonance_in_Gabriel_Okaras_The_Piano_and_Th...

Sensory_Experience_and_Emotional_Resonance_in_Gabriel_Okaras_The_Piano_and_Th...
Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx

Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Simple, Complex, and Compound Sentences Exercises.pdf

Simple, Complex, and Compound Sentences Exercises.pdf
QUATER-1-PE-HEALTH-LC2- this is just a sample of unpacked lesson

QUATER-1-PE-HEALTH-LC2- this is just a sample of unpacked lesson
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Papilomavirus.ppt
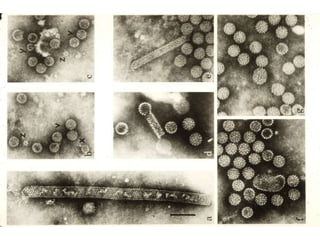
- 3. HPV and skin warts (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Table 66-3.)
- 4. From Medical Microbiology, 5th ed., Murray, Rosenthal & Pfaller, Mosby Inc., 2005, Fig. 52-4. Papillomavirus disease
- 5. Clinical genital tract and mucosal HPV’s (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Table 66-4.)
- 6. HPV16 genome Genomic map of HPV-16. The genome is a double-stranded circular DNA molecule of 7904 base pairs. Transcription occurs in a clockwise manner; the only transcriptional promoter presently mapped for HPV-16 is designated P97. The open reading frames deduced from the DNA sequence are designed E1 to E7, L1, and L2 and are indicated outside of the circular genome. AE and AL representthe early and late polyadenylation sites. The viral long control region (LCR) contains transcriptional and replication regulatory elements. (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Fig. 66-2.)
- 7. Phylogenetic tree of papillomaviruses
- 8. Papilloma virus gene function (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Table 66-1)
- 9. Differentiation of normal cutaneous squamous epithelium and papillomaviral activities in productively infected benign lesions. The various epithelial strata and the host-differentiation, stage-specific, gene-expression profile are indicated in the left and center panels. (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Fig. 65-4.) Papillomavirus replication and differentiation of the epidermis
- 10. Replication cycle of a papillomavirus. To establish a wart or papilloma, the virus must infect a basal epithelial cell. Our knowledge is limited about the initial steps in the replication cycle such as attachment (1), uptake (2), endocytosis (3), and transport to the nucleus and uncoating of the viral DNA (4). Early-region transcription (5), translation of the early proteins (6), and steady-state viral DNA replication (7) all occur in the basal cell and in the infected suprabasal epithelial cell. Events in the viral life cycle leading to the production of virion particles occur in the differentiated keratinocyte: vegetative viral DNA replication (8), transcription of the late region (9), production of the capsid proteins L1 and L2 (10), assembly of the virion particles (11), nuclear breakdown (12), and release of virus (13). (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Fig. 65-6.) Papillomavirus replication
- 11. From Medical Microbiology, 5th ed., Murray, Rosenthal & Pfaller, Mosby Inc., 2005, Fig. 52-3. Papillomavirus pathogenesis
- 12. Cervical squamous carcinoma precursors. Schematic representation of cervical cancer precursors and the different terminologies that have been used to refer to them. The risk for microinvasion from different states of squamous intraepithelial lesions (cervical intraepithelial neoplasia) is arbitrarily represented and is not necessarily proportional to that illustrated in this scheme. (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Fig. 66-4.) Cervical dysplasia
- 13. Low-grade squamous intraepithelial lesions of the cervix. A: Histologic section showing koilocytes with vacuolated cytoplasm and parakeratosis (H&E, *234). B: The darkly stained nuclei contain papillomavirus capsid antigen. [Immunoperoxidase stained with genus-specific antiserum (H&E, *234)]. C: Cervical smear. The squamous epithelial cells are rounded. They occur in clumps and display koilocytic changes (Papanicolaou stain; H&E *375). D: Cervical smear. The darkly stained nuclei contain papilloamavirus capsid antigen. (Immunoperoxidase staind with genus-specific antiserum; H&E, *750). (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001, Fig. 66-5.) Cervical dysplasia
- 14. From Medical Microbiology, 5th ed., Murray, Rosenthal & Pfaller, Mosby Inc., 2005, Fig. 52-5. Papillomavirus cytology
- 15. Correlation between HPV type, integration, and histology (From Cullen, A.P., Reid, R., Campion, M., Lorincz, A.T (1991), "Analysis of the physical state of different human papillomavirus DNAs in intraepithelial and invasive cervical neoplasm", Journal of Virology, vol. 65, p. 606-612, Fig. 4)
- 16. Structure of the single copy of HPV-16 DNA integrated into the SiHa cell line derived from a cervical carcinoma. The jagged line represents human chromosomal sequences; the nucleotide numbers pertain to the integrated HPV-16 genome. The open boxes indicate the early and late open reading frames (ORFs). Integration has occurred in the E2 ORF, and a portion of the E2 ORF has been deleted. (From Fields Virology, 4th ed, Knipe & Howley, eds, Lippincott Williams & Wilkins, 2001Fig. 66-9.) HPV DNA integration into a human chromosome
- 17. Summary: human papillomavirus • Structure – Small (8 kb) circular dsDNA genome, naked capsid • Pathogenesis – transmission by direct contact or sexual; skin, mucosa – replication in nucleus of basal cells of epithelium; very host dependent; coupled to epithelial differentiation – no spread – primarily cellular immune response – transforming infection; warts are tumors; cervical carcinoma • Diagnosis – cytology (PAP smear; koilocytosis) – immunohistochemistry – nucleic acid – electron microscopy • Treatment/prevention – surgery – recombinant subunit (VLP) vaccine