Double Displacment Reactions
•Download as DOC, PDF•
0 likes•173 views
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (11)
Construction of Ionic Equations through the Continuous Variation Method

Construction of Ionic Equations through the Continuous Variation Method
Nitrogen cycle in aquatic ecosystem...................................

Nitrogen cycle in aquatic ecosystem...................................
Isotopic analysis of soluble organic matter in meteorites

Isotopic analysis of soluble organic matter in meteorites
Similar to Double Displacment Reactions
Similar to Double Displacment Reactions (20)
Chemistry 2 experiment 1 - types of chemical reactions

Chemistry 2 experiment 1 - types of chemical reactions
Lab 9 Chemical Reactions IIPre-lab Questions1. Michelangelo u.docx

Lab 9 Chemical Reactions IIPre-lab Questions1. Michelangelo u.docx
More from Frederick High School
More from Frederick High School (20)
Recently uploaded
(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7

(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7Call Girls in Nagpur High Profile Call Girls
Recently uploaded (20)
Eluru Call Girls Service ☎ ️93326-06886 ❤️🔥 Enjoy 24/7 Escort Service

Eluru Call Girls Service ☎ ️93326-06886 ❤️🔥 Enjoy 24/7 Escort Service
MONA 98765-12871 CALL GIRLS IN LUDHIANA LUDHIANA CALL GIRL

MONA 98765-12871 CALL GIRLS IN LUDHIANA LUDHIANA CALL GIRL
Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...

Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...
(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7

(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7
Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...

Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...
Falcon's Invoice Discounting: Your Path to Prosperity

Falcon's Invoice Discounting: Your Path to Prosperity
Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...

Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...
Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available

Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available
FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756

FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756
👉Chandigarh Call Girls 👉9878799926👉Just Call👉Chandigarh Call Girl In Chandiga...

👉Chandigarh Call Girls 👉9878799926👉Just Call👉Chandigarh Call Girl In Chandiga...
Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...

Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...
Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...

Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...
Value Proposition canvas- Customer needs and pains

Value Proposition canvas- Customer needs and pains
Enhancing and Restoring Safety & Quality Cultures - Dave Litwiller - May 2024...

Enhancing and Restoring Safety & Quality Cultures - Dave Litwiller - May 2024...
Double Displacment Reactions
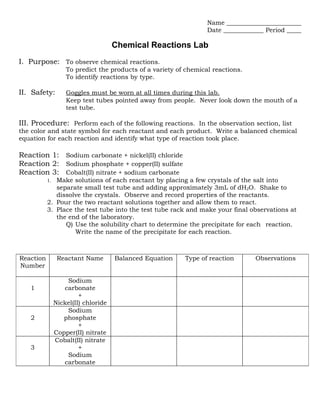
- 1. Name Date Period Chemical Reactions Lab I. Purpose: To observe chemical reactions. To predict the products of a variety of chemical reactions. To identify reactions by type. II. Safety: Goggles must be worn at all times during this lab. Keep test tubes pointed away from people. Never look down the mouth of a test tube. III. Procedure: Perform each of the following reactions. In the observation section, list the color and state symbol for each reactant and each product. Write a balanced chemical equation for each reaction and identify what type of reaction took place. Reaction 1: Sodium carbonate + nickel(II) chloride Reaction 2: Sodium phosphate + copper(II) sulfate Reaction 3: Cobalt(II) nitrate + sodium carbonate 1. Make solutions of each reactant by placing a few crystals of the salt into separate small test tube and adding approximately 3mL of dH2O. Shake to dissolve the crystals. Observe and record properties of the reactants. 2. Pour the two reactant solutions together and allow them to react. 3. Place the test tube into the test tube rack and make your final observations at the end of the laboratory. Q) Use the solubility chart to determine the precipitate for each reaction. Write the name of the precipitate for each reaction. Reaction Reactant Name Balanced Equation Type of reaction Observations Number Sodium 1 carbonate + Nickel(II) chloride Sodium 2 phosphate + Copper(II) nitrate Cobalt(II) nitrate 3 + Sodium carbonate