Recommended
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
More Related Content
Featured
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
Featured (20)
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...

Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike Routes
Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...

Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...
Handout 3.22.16
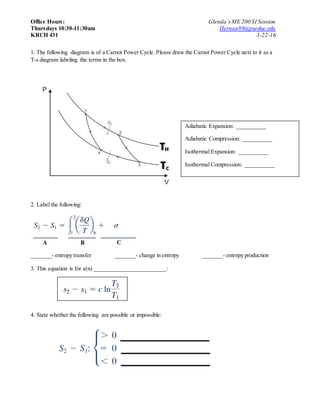
- 1. Office Hours: Glenda’sME 200 SI Session Thursdays 10:30-11:30am Hernan89@purdue.edu KRCH 431 3-22-16 1. The following diagram is of a Carnot Power Cycle. Please draw the Carnot Power Cycle next to it as a T-s diagram labeling the terms in the box. 2. Label the following: _______- entropy transfer _______- change in entropy _______- entropy production 3. This equation is for a(n) _________________________: 4. State whether the following are possible or impossible: Adiabatic Expansion: __________ Adiabatic Compression: __________ Isothermal Expansion: __________ Isothermal Compression: __________
- 2. Office Hours: Glenda’sME 200 SI Session Thursdays 10:30-11:30am Hernan89@purdue.edu KRCH 431 3-22-16 5. Water,initially a saturated liquid at 150°C (423.15 K), is contained in a piston–cylinder assembly. The water undergoes a process to the corresponding saturated vapor state,during which the piston moves freely in the cylinder. If the change of state is brought about by heating the water as it undergoes an internally reversible process at constant pressure and temperature, determine the work and heat transfer per unit of mass,each in kJ/kg. NEXT SESSION: Thursday, March 24 6:30-7:20pm UNIV 019